2. 上海呼吸物联网医学工程技术研究中心 上海 200032;
3. 上海市呼吸病研究所 上海 200032;
4. 青岛大学青岛医学院附属烟台毓璜顶医院病理科 烟台 264000;
5. 同济大学附属第十人民医院肿瘤科-同济大学癌症中心 上海 200072
2. Shanghai Engineering Research Center of Internet of Things for Respiratory Medicine, Shanghai 200032, China;
3. Shanghai Respiratory Research Institute, Shanghai 200032, China;
4. Department of Pathology, Yantai Yuhuangding Hospital, Qingdao Medical College, Qingdao University, Yantai 264000, Shandong Province, China;
5. Department of Oncology, Tenth People's Hospital of Tongji University-Tongji University Cancer Center, Shanghai 200072, China
肺癌是发病率高、预后差的恶性肿瘤,在2022年最常见的癌症中,前列腺癌、肺癌和大肠癌占男性新增病例的48%,乳腺癌、肺癌和大肠癌占女性新增病例的51%[1]。在肺癌的两种类型中,小细胞肺癌(small cell lung cancer,SCLC)的发病率低于非小细胞肺癌(non-small cell lung cancer,NSCLC)。NSCLC进一步划分为肺腺癌(lung adenocarcinoma,LUAD)和肺鳞癌(lung squamous cell carcinoma,LUSC),其中LUAD是最常见的亚型。NSCLC早期症状不明显,大多患者在晚期才被诊断出来,因此迫切需要可靠的生物标志物来评估诊断和监测进展[2]。
氨基酰-tRNA合成酶(aminoacyl-tRNA synthetase,AARS)在生物体内普遍存在,其主要功能是催化氨基酸与相应的tRNA之间发生酯化反应形成氨基酰-tRNA,为蛋白质生物合成提供原料,确保遗传信息准确传递[3]。线粒体天冬氨酰-tRNA合成酶2(aspartyl-tRNA synthetase 2,mitochondrial,DARS2)负责翻译启动及蛋白质合成[4],其突变多与线粒体疾病的发生发展密切相关。已有文献报道DARS2在肺癌、胰腺癌和膀胱癌的预后评估模型中表现出良好预后预测意义[5-8],这些模型以RNA结合蛋白(RNA binding protein,RBP)为基础构建。RBP对转录后调控至关重要,其表达异常是促癌网络的重要一环[9]。本研究选取NSCLC的两个不同亚型,采用多种数据库比较和细胞功能实验相结合的方法,以期揭示DARS2在NSCLC发生发展中的作用。
材料和方法细胞培养和转染 复旦大学附属中山医院实验研究中心负责培养细胞,培养基和血清以9∶1制备成完全培养基,在25 T培养瓶中正常培养,并储存于标准培养箱。取40%的H1299细胞铺于6孔板上,次日每孔添加0.5 mL感染增强液、0.5 mL培养基、1 μL聚凝胺和14 μL shDARS2慢病毒液进行转染。
Western blot实验 取适当RIPA裂解液,使用前添加蛋白酶抑制剂,6孔板每孔加入80~100 μL裂解液,吹打数下,促进细胞裂解。在冰上裂解15 min后枪头刮至EP管,4 ℃下18 407×g离心10 min,取上清。BCA法定量蛋白浓度,5×上样缓冲液按4∶1稀释,后续以电泳转膜曝光等标准流程进行。
细胞实验 增殖实验:将慢病毒转染的H1299细胞以5×103/孔的密度接种至96孔板,在培养箱孵育72 h,每孔加入100 μL含有10 μL CCK-8试剂的培养基,继续孵育4 h,用多功能酶标仪检测吸光度,并评估对细胞增殖能力的影响。划痕实验:将慢病毒转染的H1299细胞接种于6孔板中直至长满,枪头垂直于6孔板底部划一条直线,PBS清洗后悬浮细胞,倒置显微镜在12 h和24 h时拍照记录划痕宽度,使用ImageJ软件分析划痕的相对宽度,并评估对细胞迁移能力的影响。
qRT-PCR实验 使用Trizol提取H1299细胞RNA,以10 μL为逆转录体系制备cDNA,加DEPC水5倍稀释。按照TAKARA试剂盒说明书,qRT-PCR按照10 μL体系预混并加样。DARS2正向引物序列:5'-AAGATGTGGTCCTACTAACTGC-3',反向引物序列:5'-TGTTTCTAGAAGGTCAG-CACAT-3'。
数据库分析 通过数据库分析DARS2在LUAD及LUSC中的相关生物信息学(表 1)。
| Database | Function | Website |
| GEPIA | Gene expression profiling interactive analysis | http://gepia.cancer-pku.cn |
| GEO | Query and download experiments and curated gene expression profiles. | https://www.ncbi.nlm.nih.gov/geo/ |
| HPA | Open access resource for human proteins | https://www.proteinatlas.org |
| UALCAN | Web resource for analyzing cancer OMICS data | https://ualcan.path.uab.edu |
| cBioPortal | Interactive exploration of multidimensional cancer genomics data sets | https://www.cbioportal.org |
| Linkedomics | Analyze and compare cancer multi-omics data within and across tumor types | http://www.linkedomics.org/login.php |
| Metascape | A gene annotation and analysis resource | https://metascape.org/gp/index.html |
统计学分析 使用GraphPadPrism Version 9.00软件进行统计分析,两组数据之间差异分析采用独立样本t检验,P < 0.05为差异有统计学意义。
结果DARS2在NSCLC中表达及与NSCLC患者生存预后的关系 通过GEO (GSE19804、GSE18842和GSE21933)数据库分析发现,在NSCLC组织中DARS2 mRNA的表达水平均明显高于正常组织(P < 0.001,图 1A~C);GEPIA数据库结果进一步证实,在LUAD和LUSC组织中DARS2 mRNA的表达水平均显著上调(P < 0.001,图 1D)。在HPA数据库检索界面输入“DARS2”,并在“Tissue Atlas”和“Pathology Atlas”模块分别选择“Lung”和“Lung cancer”,生成正常肺组织、LUAD和LUSC组织中DARS2蛋白表达水平的免疫组化图(图 1E~G),结果显示DARS2蛋白在LUAD与LUSC组织中均高表达。GEPIA数据库病理分期分析结果显示,DARS2 mRNA表达水平在不同病理分期的LUAD组织中明显不同(P < 0.001,图 1H),且与LUAD的病理分期呈正相关。预后分析模块结果显示,以中位数为截断值,DARS2表达水平仅与LUAD患者总生存期呈负相关(P < 0.001,图 1J)。
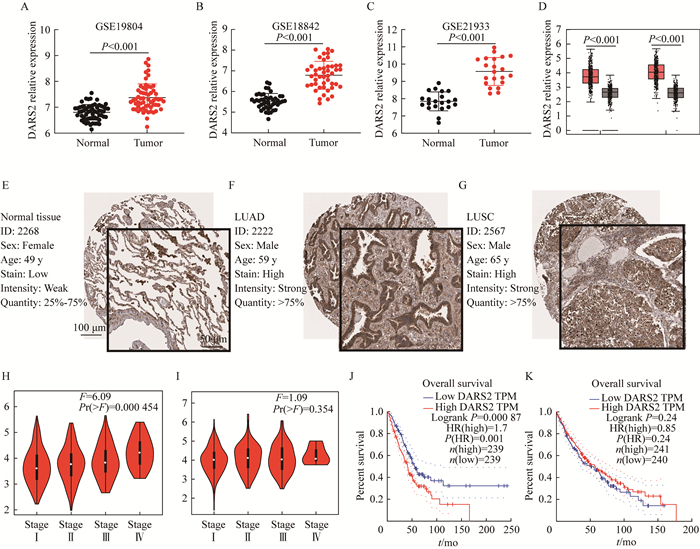
|
| A-D: DARS2 mRNA expression analysis in NSCLC and normal tissues in GEO (GSE19804, GSE18842 and GSE21933) and GEPIA; E-G: Representative IHC images of DARS2 expression of normal, LUAD and LUSC in HPA (HPA026506) database; H-I: Expression of DARS2 mRNA in different stages of LUAD and LUSC in GEPIA; J-K: Correlation between DARS2 expression and prognosis of LUAD and LUSC in GEPIA. 图 1 DARS2在NSCLC和正常组织中的表达与预后 Fig 1 Expression and prognosis of DARS2 in NSCLC and normal tissues |
DARS2在NSCLC中的基因变异及甲基化水平分析 利用cBioPortal数据库分析DARS2在NSCLC中基因变异情况,结果显示:在503例LUAD患者中有81例(16%)发生基因变异,变异类型主要包括错义突变、基因扩增和mRNA高表达;在466例LUSC患者中有83例(18%)发生基因变异,变异类型主要包括错义突变、剪接突变、截短突变、结构变异、基因扩增和mRNA高表达(图 2A)。UALCAN数据库甲基化分析结果显示:在LUAD组织中,DARS2基因启动子甲基化水平明显降低(P=0.004 9,图 2B),而在LUSC组织中,DARS2基因启动子甲基化水平明显升高(P=0.020 6,图 2B)。

|
| A: Alteration of DARS2 gene in LUAD and LUSC (cBioPortal); B: Promoter methylation level of DARS2 in LUAD and LUSC (UALCAN). 图 2 NSCLC中DARS2的基因变异和启动子甲基化改变 Fig 2 Genetic alteration and promoter methylation of DARS2 in NSCLC |
DARS2敲低对H1299细胞增殖和迁移的影响 qRT-PCR实验结果表明,DARS2在LUAD细胞(H1299)中的表达量明显高于正常支气管上皮细胞(BEAS-2B和HBE)(P=0.02,图 3A),故选择在H1299细胞中敲低DARS2基因的表达并用qRT-PCR与Western blot检测转染效率(图 3B)。CCK-8实验结果显示,DARS2基因敲低组在培养72 h后OD值明显降低(P < 0.001,图 3C),与正常组相比增殖能力明显削弱。细胞划痕实验结果显示,敲低DARS2基因后12 h和24 h,H1299细胞的迁移能力均显著低于正常组(P < 0.001,图 3D)。这些结果表明DARS2表达水平下降有助于抑制LUAD细胞的增殖和迁移。

|
| A: Expression level of DARS2 in cell lines identified by qRT-PCR; B: Transfection rate identified by qRT-PCR and Western blot; C-D: CCK-8 and wound healing were performed to identify the proliferation and migration rate after DARS2 knockdown in H1299 cells. 图 3 下调DARS2对NSCLC细胞增殖和迁移影响 Fig 3 Interference of DARS2 expression impaired proliferation and migration of NSCLC cells |
DARS2在LUAD与LUSC中共表达基因分析 通过LinkedOmics数据库对DARS2在LUAD与LUSC中相关基因进行分析,结果显示:LUAD中有4 180和6 135个基因分别与DARS2表达呈正相关和负相关(图 4A),而LUSC中有3 767个和5 293个这样的基因(图 4B)。在LUAD和LUSC中,与DARS2正负相关性排名前50的基因如图 4C~4F所示。在LUAD中,DARS2的表达与NDUFS1(r=0.638 1)、UCHL5(r=0.629 8)和WDR12(r=0.619 2)等基因呈显著正相关,与LTC4S(r=‒0.491 0)、FCGBP(r=‒0.488 9)和TLR2(r=-0.486 6)等基因呈显著负相关。在LUSC中,DARS2的表达与CENPL(r=0.620 5)、FAM72D(r=0.607 2)和NUF2(r=0.605 6)等基因呈显著正相关,与RRAS(r=-0.445 7)、LDLRAD2 (r=‒0.441 6)、IL34 (r=‒0.441 0)等基因呈现显著负相关。使用Metascape数据库,参数模块选择种属“人类”(H. sapiens),列表中输入与DARS2正负相关性排名前100的基因,进行通路功能富集分析。结果显示,在LUAD中主要富集在ncRNA代谢、细胞周期和线粒体基因表达等环节(图 5A),在LUSC中主要富集在有丝分裂细胞周期、细胞周期和纺锤体组装等环节(图 5B)。这些结果提示DARS2可能通过参与调控细胞周期信号通路促进NSCLC进展。
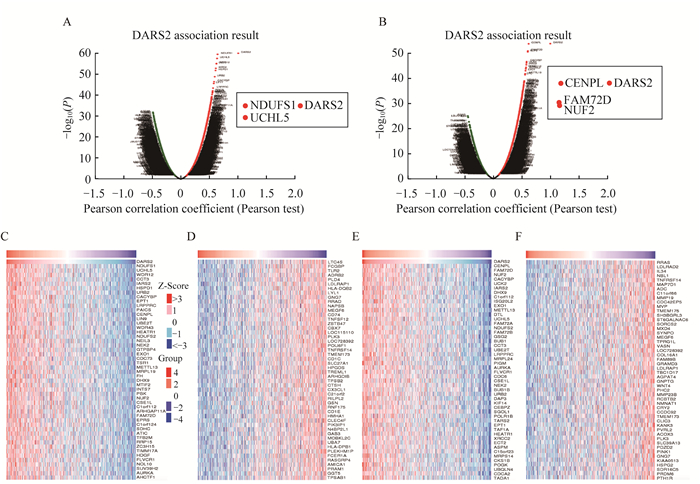
|
| A-B: Volcano map of the distribution of DARS2 co-expressed genes in LUAD and LUSC; C-F: Heat maps showed the top 50 significant genes positively or negatively correlated with DARS2 in LUAD (C-D) and LUSC (E-F), in which the blue part stands for positively correlated genes and the red part stands for negatively correlated genes (Linkedomics). 图 4 NSCLC中与DARS2共表达的基因 Fig 4 Genes in association with DARS2 in NSCLC |
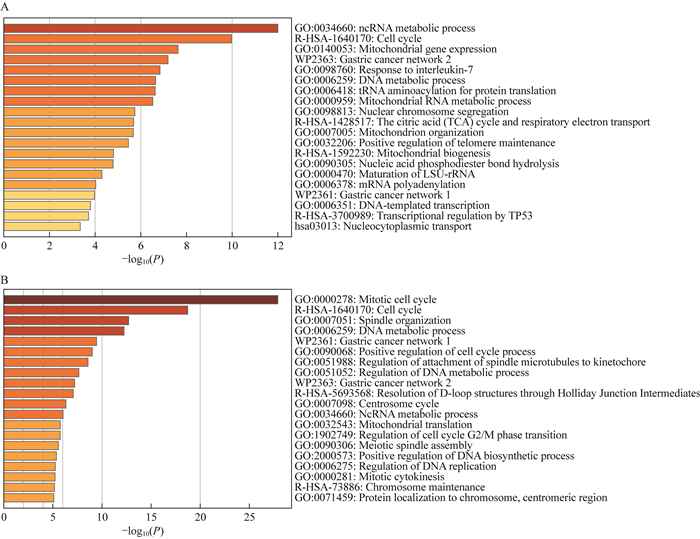
|
| Metascape analyses of the top 100 genes positively or negatively correlated with DARS2 in LUAD (A) and LUSC (B). 图 5 相关基因富集分析 Fig 5 Enrichment analysis of correlated genes |
我国统计结果显示,2020年全国新增肺癌诊断病例约82万例,相关死亡病例约71.5万例[10]。由于其复杂的生物过程且缺乏有效的生物标志物,肺癌患者的5年存活率较低(约20%)。AARS在生物体内广泛存在,其中37种AARS均由核基因组编码,主要参与细胞质和线粒体的蛋白质生物合成,此外还参与小分子代谢、细胞凋亡、肿瘤发生、组织发育、血管生成和免疫应答等[11]。研究结果表明,AARS的功能与癌症调控有关[12]。AARS通过调节肿瘤细胞生长、分化、细胞周期、细胞因子活性、RNA剪接、细胞黏附和血管生成等,在肿瘤发病机制中发挥重要作用。既往研究表明,异亮氨酰-tRNA合成酶2(isoleucyl-tRNA synthetase 2,IARS2)沉默可抑制A549和H1299细胞的活性,导致A549细胞的G0/G1停滞和线粒体凋亡,并通过AKT/MTOR途径调节肺癌细胞的增殖[13]。还有研究发现蛋氨酸-tRNA合成酶(methionyl-tRNA synthetase,MRS)在肺癌组织中的表达量增加且与细胞周期的进展密切相关[3]。
DARS2属于AARS家族,组织特异性DARS2缺失导致线粒体功能紊乱,进而导致心肌和骨骼肌的线粒体呼吸功能缺失[14]。研究发现,DARS2与神经系统疾病发生发展密切相关[15-16],如其突变可致涉及脑干和脊髓的脑白质病,并伴随过量乳酸堆积。目前DARS2在癌症方面相关研究较少。在乙肝病毒感染所致的肝癌中,转录因子NFAT5的表达被该病毒抑制进而会导致DARS2的表达水平上升,肝癌细胞凋亡率下降[17]。
本研究通过多种公共数据库进行比对分析,发现DARS2在NSCLC的两种亚型LUAD与LUSC中均高表达,且高表达DARS2的LUAD患者具有更短的生存期。为了探究DARS2在LUAD中生物学作用,构建了DARS2基因敲低的H1299细胞株,通过细胞增殖与划痕实验发现DARS2可以促进H1299细胞增殖迁移。通过cBioPortal数据库评估DARS2在肺癌中的变异模式,结果发现,DARS2在LUAD与LUSC中均发生了突变,错义突变是两者最普遍的突变模式。低甲基化作为一种刺激癌基因的机制,可促进肿瘤的发生,我们对DARS2进行了甲基化分析,与正常组织相比,在LUAD组织中DARS2表现出明显低甲基化。对与DARS2相关基因进行分析,Linkedomics显示LUAD中正相关性排名前3位的基因是NDUFS1、UCHL5和WDR12,LUSC中正相关性排名前3位的基因是CENPL、FAM72D和NUF2。NDUFS1是线粒体复合体Ⅰ的最大亚单位,先前研究显示NDUFS1是由2q33-miR-3130-5p轴靶向的一个潜在的肿瘤抑制因子,影响LUAD的侵袭性[18]。UCHL5在NSCLC组织中高表达,在LUAD细胞中敲除UCHL5能明显抑制细胞增殖并减少关键细胞周期蛋白的表达[19]。WDR12是一个癌基因,在多种人类恶性肿瘤中过表达,其过表达与不良的预后相关[20]。敲除CENPL可明显抑制CDK2和CCNE2的表达,并诱导LUAD的G0/G1停滞和凋亡[21]。FAM72D是控制细胞增殖和生存的FOXM1转录因子网络的一部分[22]。敲除NUF2能抑制LUAD细胞的增殖、迁移、侵袭、上皮-间质转化和集落形成[23]。富集分析结果显示,在LUAD与LUSC中,与DARS2相关的基因在细胞周期通路高度集中。关于DARS2调控细胞周期,有研究结果指出,ADP-DnaA转化为ATP-DnaA需要DARS2参与,并通过一个负反馈环以细胞周期依赖性方式进行调节[24]。
综上所述,本研究发现DARS2在NSCLC组织中高表达,DARS2过表达与LUAD患者不良预后相关,且与病理分期正相关。在LUAD细胞中敲低DARS2基因后,细胞的迁移和侵袭能力明显降低,DARS2可能以参与细胞周期通路的方式调控NSCLC进展。本研究局限性在于,相关分析结果多来自公共数据库,缺乏实验研究及临床样本分析,且未能补充DARS2过表达实验,后续将进一步明确DARS2在NSCLC中分子机制及作用。
作者贡献声明 艾尼瓦尔·肉孜 实验操作,数据分析,论文撰写。韩林晓,吴圆圆,朱文思 实验设计和指导。刘静,刘尽国 论文修订。周建 研究设计,论文修订。
利益冲突声明 所有作者均声明不存在利益冲突。
| [1] |
SIEGEL RL, MILLER KD, FUCHS HE, et al. Cancer statistics, 2022[J]. CA Cancer J Clin, 2022, 72(1): 7-33.
[DOI]
|
| [2] |
HERBST RS, MORGENSZTERN D, BOSHOFF C. The biology and management of non-small cell lung cancer[J]. Nature, 2018, 553(7689): 446-454.
[DOI]
|
| [3] |
BHOWAL P, KARMAKAR PB, DEY D, et al. Aminoacyl-tRNA synthetases, indispensable players in lung tumorigenesis[J]. Protein Pept Lett, 2022, 29(3): 208-217.
[DOI]
|
| [4] |
KONOVALOVA S, TYYNISMAA H. Mitochondrial. aminoacyl-tRNA synthetases in human disease[J]. Mol Genet Metab, 2013, 108(4): 206-211.
[DOI]
|
| [5] |
YANG L, ZHANG R, GUO G, et al. Development and validation of a prediction model for lung adenocarcinoma based on RNA-binding protein[J]. Ann Transl Med, 2021, 9(6): 474.
[DOI]
|
| [6] |
SUN N, CHU J, HU W, et al. A novel 14-gene signature for overall survival in lung adenocarcinoma based on the Bayesian hierarchical Cox proportional hazards model[J]. Sci Rep, 2022, 12(1): 27.
[DOI]
|
| [7] |
WU Y, LIU Z, WEI X, et al. Identification of the functions and prognostic values of RNA binding proteins in bladder cancer[J]. Front Genet, 2021, 12: 574196.
[DOI]
|
| [8] |
陈钦钦, 杨凌志, 彭心宇. 胰腺癌中关键RNA结合蛋白的功能及预后评估价值[J]. 中国现代医药杂志, 2021, 23(1): 5-11. [CNKI]
|
| [9] |
NAG S, GOSWAMI B, MANDAL SDAS, et al. Cooperation and competition by RNA-binding proteins in cancer[J]. Semin Cancer Biol, 2022, 86(Pt 3): 286-297.
|
| [10] |
CHEN P, LIU Y, WEN Y, et al. Non-small cell lung cancer in China[J]. Cancer Commun (Lond), 2022, 42(10): 937-970.
[DOI]
|
| [11] |
李光, 周小龙, 王恩多. 氨基酰-tRNA合成酶与神经系统疾病[J]. 生命科学, 2020, 32(8): 763-772. [DOI]
|
| [12] |
WANG J, YANG XL. Novel functions of cytoplasmic aminoacyl-tRNA synthetases shaping the hallmarks of cancer[J]. Enzymes, 2020, 48: 397-423.
|
| [13] |
DI X, JIN X, MA H, et al. The oncogene IARS2 promotes non-small cell lung cancer tumorigenesis by activating the AKT/MTOR pathway[J]. Front Oncol, 2019, 9: 393.
[DOI]
|
| [14] |
SEIFERLING D, SZCZEPANOWSKA K, BECKER C, et al. Loss of CLPP alleviates mitochondrial cardiomyopathy without affecting the mammalian UPRmt[J]. EMBO Rep, 2016, 17(7): 953-964.
[DOI]
|
| [15] |
吴腾辉, 彭镜, 张慈柳, 等. 氨酰基-tRNA合成酶基因变异10例分析[J]. 中国当代儿科杂志, 2020, 22(6): 595-601. [CNKI]
|
| [16] |
SMITH FINE A, KAUFMAN M, GOODMAN J, et al. Wearable sensors detect impaired gait and coordination in LBSL during remote assessments[J]. Ann Clin Transl Neurol, 2022, 9(4): 468-477.
[DOI]
|
| [17] |
QIN X, LI C, GUO T, et al. Upregulation of DARS2 by HBV promotes hepatocarcinogenesis through the miR-30e-5p/MAPK/NFAT5 pathway[J]. J Exp Clin Cancer Res, 2017, 36(1): 148.
[DOI]
|
| [18] |
ZHAN J, SUN S, CHEN Y, et al. MiR-3130-5p is an intermediate modulator of 2q33 and influences the invasiveness of lung adenocarcinoma by targeting NDUFS1[J]. Cancer Med, 2021, 10(11): 3700-3714.
[DOI]
|
| [19] |
ZHANG J, XU H, YANG X, et al. Deubiquitinase UCHL5 is elevated and associated with a poor clinical outcome in lung adenocarcinoma (LUAD)[J]. J Cancer, 2020, 11(22): 6675-6685.
[DOI]
|
| [20] |
EID RA, ELDEEN MA, SOLTAN MA, et al. Integrative analysis of WDR12 as a potential prognostic and immunological biomarker in multiple human tumors[J]. Front Genet, 2022, 13: 1008502.
|
| [21] |
FENG Z, CHEN Y, CAI C, et al. Pan-cancer and single-cell analysis reveals CENPL as a cancer prognosis and immune infiltration-related biomarker[J]. Front Immunol, 2022, 13: 916594.
[DOI]
|
| [22] |
CHATONNET F, PIGNARRE A, SERANDOUR AA, et al. The hydroxymethylome of multiple myeloma identifies FAM72D as a 1q21 marker linked to proliferation[J]. Haematologica, 2020, 105(3): 774-783.
[DOI]
|
| [23] |
JIANG F, HUANG X, YANG X, et al. NUF2 expression promotes lung adenocarcinoma progression and is associated with poor prognosis[J]. Front Oncol, 2022, 12: 795971.
[DOI]
|
| [24] |
MIYOSHI K, TATSUMOTO Y, OZAKI S, et al. Negative feedback for DARS2-Fis complex by ATP-DnaA supports the cell cycle-coordinated regulation for chromosome replication[J]. Nucleic Acids Res, 2021, 49(22): 12820-12835.
[DOI]
|
 2023, Vol. 50
2023, Vol. 50


