2. 上海市血管病变调控与重塑重点实验室 上海 201399;
3. 复旦大学附属华山医院血管外科 上海 200040
2. Shanghai Key Laboratory of Vascular Lesions Regulation and Remodeling, Shanghai 201399, China;
3. Department of Vascular Surgery, Huashan Hospital, Fudan University, Shanghai 200040, China
血栓闭塞性脉管炎(thromboangiitis obliterans,TAO)也称为伯格病(Buerger disease),是一种节段性非动脉硬化性炎症和闭塞性疾病,主要影响四肢的中小血管和神经[1]。TAO与烟草接触密切相关,可能由于烟草接触提高了患者体内的氧化应激水平,导致血管收缩,TAO好发于年轻吸烟者[2]。TAO患者10年和15年截肢率分别为26%和34%,戒烟可明显降低截肢风险[3]。虽然TAO自发现以来已经过去了一个多世纪,但其发病机制尚无定论[4]。
外泌体是最小的膜囊泡,携带大量功能蛋白、mRNA和microRNA(miRNA)等,充当细胞间物质和信息交流的关键信使[5]。外泌体可以调节血管内皮细胞功能,抑制心脏损伤和促进心脏修复,在炎症中发挥调节作用,具有治疗炎症性疾病的潜力[6-9]。循环外泌体miRNA可能是多种疾病的生物标记物和治疗靶点,如终末期肾病[10]、心血管疾病[11]和几种类型的癌症[12-14]。外泌体可能通过调节炎症、自身免疫、促凝血和内皮损伤/功能障碍参与血管炎的发病机制[15]。讨论循环外泌体miRNA在TAO发病机制中生物学功能的研究较少。
我们研究了从TAO患者和配对正常对照受试者血浆中分离的外泌体miRNA表达谱,通过生物信息学分析鉴定出差异表达的miRNA(differentially expressed miRNA,DE-miRNA),探讨其和TAO患者血浆源性外泌体对人血管平滑肌细胞(human vascular smooth muscle cell,HVSMC)的影响,以期了解TAO患者血浆循环外泌体在TAO发病过程中的分子机制。
材料和方法细胞培养和处理 HVSMC和HEK-T293细胞来自武汉大学细胞库。HVSMCs在F12K培养基(21127022,美国Gibco公司)中培养,其中含有10%FBS(16000-044,美国Gibco公司)和1%青霉素/链霉素(15140-122,美国Gibco公司);HEK-T293细胞在Dulbecco改良的Eagle培养基(11995500BT,美国Gibco公司)中培养,添加10% FBS和1%青霉素/链霉素。
血浆样本采集 血液样本来源于2019年1—12月在复旦大学附属浦东医院诊断为TAO的3名患者和3名健康受试者,纳入标准:50岁前出现症状,吸烟/吸烟史,无其他动脉粥样硬化危险因素,上肢累及中等大小血管或浅表血栓性静脉炎。随机选取无血栓或动脉疾病史的年龄匹配的健康献血者作为对照组。排除其他原因导致的外周动脉疾病,如动脉粥样硬化、栓塞性血管闭塞、血脂异常、血管炎、创伤、放射性动脉炎、促凝状态。本研究获得浦东医院伦理委员会批准(编号:wz-003),参与者均签署知情同意书。
TAO患者和健康对照受试者各采集血样10 mL,4 ℃下1 900×g离心10 min。接着,将悬浮液转移到新的试管中,4 ℃下3 000×g离心15 min。‒80 ℃下储存悬浮液,用于分离外泌体。
外泌体分离和鉴定 根据标准方案[16],4 ℃下12 000×g超速离心30 min,分离提纯血浆样本中的外泌体。0.22 mm微孔膜过滤上清液,去除大膜泡。过滤后的上清液在4 ℃下120 000×g超速离心60 min,所得颗粒用10 mL PBS重悬,4 ℃下120 000×g再次超速离心60 min。
使用NanoSight LM10-HS系统(NS300,英国马尔文仪器有限公司)通过纳米粒子跟踪分析(nanoparticle tracking analysis,NTA)评估外泌体的大小分布。在120 kV透射电子显微镜下(transmission electron microscope,TEM)(JEM-1230,美国Joel公司)观察纯化外泌体的形态[17-18]。5 μL外泌体悬浮液放置于铜网聚醋酸甲基乙烯脂涂层碳稳定格栅,10 min后用滤纸将残留的悬浮液滤掉,向网格中添加2%磷钨酸(10 mL,pH=6.5),室温下染色2 min。
Western blot分析 观察前使网格干燥,Western blot检测CD63、CD9和CD81表达。使用RIPA蛋白裂解缓冲液(P0013B,上海碧云天生物技术有限公司)从获得的外泌体中分离全部蛋白,使用BCA蛋白分析试剂盒(PL212989,武汉博士德生物工程有限公司)测量蛋白浓度。通过10% SDS-PAGE分离蛋白质样品(20 μg),转移至聚偏二氟乙烯膜(polyvinylidene fluoride,PVDF)(IPVH00010,美国Millipore公司),37 ℃下用5%脱脂牛奶封闭2 h。分别用抗CD63抗体(A5271,美国ABclonal公司)、抗CD81抗体(ab109201,英国Abcam公司)、抗CD9抗体(ab92726,英国Abcam公司),抗钙结合蛋白抗体(10427-2-AP,美国Proteintech公司)和抗白蛋白抗体(16475-1-AP,美国Proteintech公司)与PVDF膜共同孵育一夜。用磷酸盐吐温缓冲液(phosphate buffered saline Tween,PBST)洗涤3次,将膜与辣根过氧化物酶结合的山羊抗兔IgG(111-035-003,美国Jackson Immuno Research公司)在37 ℃下孵育2 h。PBST洗涤3次,使用微孔电化学发光系统观察蛋白质条带。
miRNA文库的构建和测序 根据标准方法,使用RNAiso Plus[9109,宝生物工程(大连)有限公司]从外泌体中提取全部RNA。miRNA文库的构建和测序由研载生物科技(中国上海)有限公司完成。使用18~30个核苷酸的小RNA用于文库构建,PCR扩增并测序小RNA。使用blast软件将获得的有效读数与Rfam[19]数据库(10.0版)比对,E阈值截止值为≤0.01。使用miRDeep 2软件,用未被注释的miRNA预测新的miRNA[20]。
DE-miRNAs的筛选及功能分析 根据{log2(fold change)}≥1.5的标准,使用R语言的DEseq(1.18.0)包,在TAO和正常对照组的的外泌体之间进行筛选,控制FDR < 0.05[21]。利用miRanda数据库对所鉴定的DE-miRNAs的靶基因进行预测。利用超几何分布检验统计量,进行GO(Gene Ontology)[22]功能和KEGG(Kyoto Encyclopedia of Genes and Genomes)[23]路径富集分析。
RT-qPCR定量检测外泌体miRNA 将全部RNA(0.5 μg)反转录为cDNA,使用PrimeScriptTM RT Master Mix试剂盒[RR036A,宝生物工程(大连)有限公司]检测miRNA水平或使用PrimeScriptTMII第一链cDNA合成试剂盒[6210,宝生物工程(大连)有限公司]检测mRNA表达。使用Power SYBR Green PCR主混合试剂盒(4367659,美国Thermo Fisher Scientific公司)扩增cDNA产物。RT-qPCR反应条件为:95 ℃预变性2 min;95 ℃ 15 s,循环40次,60 ℃ 60 s,循环40次;95 ℃下15 s、60 ℃下60 s和95℃下15 s。引物序列如表 1所示,U6用作内参,cel-miRR-39-3p标准RNA(miRB0000010-3-1,广州锐博生物公司)用作外部参考。甘油醛-3-磷酸脱氢酶(glyceraldehyde-3-phosphate dehydrogenase,GAPDH)用作mRNA的管家基因。使用2‒△△Ct方法计算miRNA的相对水平和相对mRNA表达。
| Primer | Sequence(5ʹ-3ʹ) |
| VCAM1-hF | GGGAAGATGGTCGTGATCCTT |
| VCAM1-hR | TCTGGGGTGGTCTCGATTTTA |
| IGF1R-hF | TCGACATCCGCAACGACTATC |
| IGF1R-hR | CCAGGGCGTAGTTGTAGAAGAG |
| hsa-let-7b-3p-RT | GTCGTATCCAGTGCAGGGTCCGAGGTATTCGCACTGGATACGACGGGAAG |
| hsa-let-7b-3p-F | GCCTATACAACCTACTGC |
| hsa-miR-127-3p-RT | GTCGTATCCAGTGCAGGGTCCGAGGTATTCGCACTGGATACGACAGCCAA |
| hsa-miR-127-3p-F | GCTCGGATCCGTCTGAG |
| hsa-miR-223-5p-RT | GTCGTATCCAGTGCAGGGTCCGAGGTATTCGCACTGGATACGACAACTCA |
| hsa-miR-223-5p-F | GCCGTGTATTTGACAAGC |
| novel19_mature-RT | GTCGTATCCAGTGCAGGGTCCGAGGTATTCGCACTGGATACGACCCCACA |
| novel19_mature-F | GCGAATCAGTGAGAC |
| novel227_mature-RT | GTCGTATCCAGTGCAGGGTCCGAGGTATTCGCACTGGATACGACCCTTCC |
| novel227_mature-F | GCGGGAAACTCTGGT |
| hsa-miR-6529-5p-RT | GTCGTATCCAGTGCAGGGTCCGAGGTATTCGCACTGGATACGACCACTCT |
| hsa-miR-6529-5p-F | GCGAGAGATCAGAGGCGC |
| cel-miR-39-RT | GTCGTATCCAGTGCAGGGTCCGAGGTATTCGCACTGGATACGACCAAGCT |
| cel-miR-39-F | GGCCTCACCGGGTGTAAATCAG |
| Universal downstream | GTGCAGGGTCCGAGGT |
| hsa-U6-RT | GTCGTATCCAGTGCAGGGTCCGAGGTATTCGCACTGGATACGACAAAATATG |
| hsa-U6-F | CTCGCTTCGGCAGCACA |
| hsa-U6-R | AACGCTTCACGAATTTGCGT |
| GAPDH-hF | TGACAACTTTGGTATCGTGGAAGG |
| GAPDH-hR | AGGCAGGGATGATGTTCTGGAGAG |
外泌体内化的共聚焦扫描显微镜分析 使用PKH67染色试剂盒(PKH67GL-1KT,美国Sigma公司)标记PKH67(绿色荧光),观察HVSMC中的外泌体内化[24]。将从TAO患者血浆中分离的700 μL外泌体添加到1 300 μL稀释剂C中,再添加16 μL PKH67染料和2 mL稀释剂C。混合物在室温下培养5 min,并添加1%牛血清白蛋白(V900933-100G,美国Sigma公司)4 mL以结合过量的染料。混合物在4 ℃下120 000×g离心90 min,沉淀物(PKH67标记的外泌体)用300 μL PBS重悬。
将密度为1×105/孔的HVSMC接种到24孔培养板中,培养过夜。向细胞中加入10 μg/mL PKH67标记的外泌体,共孵育24 h和48 h后,用4%多聚甲醛固定细胞20 min,加入0.1% Triton X-100,室温下培养20 min。PBS洗涤后,用DAPI对细胞进行染色,在激光共聚焦扫描显微镜下观察(TCS SP8,德国Leica公司)。
细胞分组 MiR-223-5p模拟物、阴性对照(NC)模拟物和MiR-223-5p抑制剂购自广州锐博生物技术有限公司。将密度为1×105/孔的HVSMC接种到24孔培养皿中,培养过夜。将细胞分为5组:空白组、NC组、外泌体组、miRNA模拟物组和miRNA抑制剂+外泌体组。使用Lipofectamine 2000,用NC模拟物和miR-223-5p模拟物分别转染NC组和miRNA模拟物组中的细胞。外泌体组中的细胞用100 μg TAO患者血浆源性外泌体进行处理。对于miRNA抑制剂+外泌体组的细胞,先用Lipofectamine 2000转染miR-223-5p抑制剂,再用100 μg TAO患者血浆来源外泌体进行处理。空白组细胞不作处理。
细胞活力与凋亡 使用CCK-8试剂盒(C0038,上海碧云天生物技术有限公司)检测HVSMC的细胞活力。细胞培养24、48和72 h,每孔中添加10 μL CCK-8试剂。孵育2 h后,使用微孔板读取器(MK3,美国Thermo Fisher Scientific公司)在450 nm处测量吸光度。
使用Annexin V-FITC凋亡检测试剂盒(556420,美国Pharmingen公司)检测HVSMC的细胞凋亡。收集细胞,200×g离心5 min。PBS洗涤,100 μL 1×结合缓冲液重悬。加入5 μL FITC-Annexin V和5 μL PI(50 mg/mL),25 ℃黑暗环境中培养15 min。加入400 μL 1×结合缓冲液,通过流式细胞仪(FACScalibur,美国BD公司)获取图像,分析细胞凋亡率。所有实验均重复3次。
双荧光素酶报告试验 合成hsa-miR-223-5p和VCAM-1 3’非翻译区(3’-UTR)序列。使用pGL3基本载体[通用生物系统(安徽)有限公司]构建3’-UTR血管内皮细胞黏附分子1(vascular endothelial cell adhesion molecule-1,VCAM-1)报告质粒(pGL3-VCAM1)。使用Lipofectamine 2000将pGL3-VCAM1(500 ng)或pGL3基本载体(500 ng)与miR-223-5p模拟物(100 nmol/L)或NC模拟物(100 nmol/L)共转染至HEK-T293细胞。转染后48 h,使用双荧光素酶报告分析系统(E1910,美国Promega公司)测量荧光素酶活性。
统计学分析 使用GraphPad Prism 5软件进行统计分析。两组数据的比较采用t检验。两组以上的比较采用方差分析(ANOVA)。P < 0.05为差异有统计学意义。
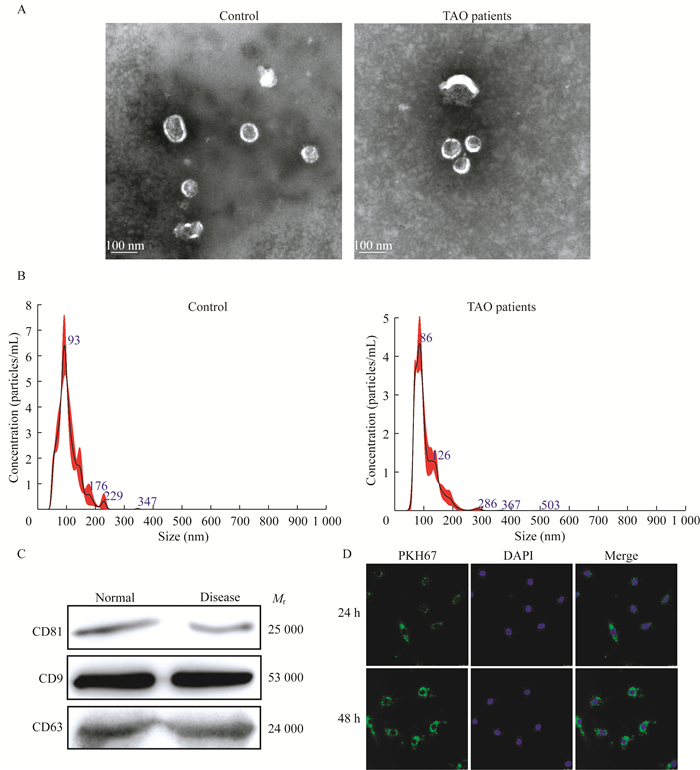
结果TAO患者和对照组血浆外泌体的鉴定 患者和健康对照者的人口学数据无显著差异(表 2)。分别从3名TAO患者和3名健康对照者的血浆样本中分离出外泌体。透射电子显微镜(transmission electron microscope,TEM)结果显示纯化的外泌体颗粒直径为40~100 nm(图 1A),纳米颗粒跟踪分析(nano-paticle tracking analysis,NTA)显示从TAO患者和健康对照提取的血浆来源的外泌体具有相似的粒径分布(图 1B),浓度分别为9.44×1010/mL和1.456×1010 /mL。以CD81、CD9和CD63为外泌体标记物,以钙结合蛋白和白蛋白为NC标记物[25]。Western blot检测发现,CD81、CD9和CD63在外泌体中均有表达(图 1C)。结果表明,从TAO患者和健康对照者的血浆中成功分离出外泌体。
| Case | Gender | Age(y) | Drug usage | SBP(mmHg) | DBP(mmHg) | Blood glucose(mmol/L) | LDL(mmol/L) | TG(mmol/L) | Complication | Therapeutic schedule |
| TAO 1 | Male | 26 | Alprostadil | 116 | 76 | 4.67 | 3.11 | 1.07 | No | Lower extremity arterial balloon dilatation |
| TAO 2 | Male | 36 | Berprost sodium | 138 | 90 | 4.68 | 2.62 | 0.84 | No | Lower extremity arterial balloon dilatation |
| TAO 3 | Male | 36 | Alprostadil | 131 | 91 | 4.42 | 2.22 | 1.61 | No | Lower extremity arterial balloon dilatation |
| SBP:Systolic blood pressure;DBP:Diastolic blood pressure;LDL:Low density lipoprotein;TG:Triglyceride; | ||||||||||
|
| A: Representative transmission electron microscopic images of exosomes; B: NTA analysis of plasma-derived exosomes from TAO patients and healthy controls; C: Expression of CD81, CD9, CD63, calnexin and albumin determined by Western blot; D: PKH67-labelled exosomes could be taken up by HVSMCs after co-cultured for 24 h and 48 h (400×). 图 1 TAO患者和健康对照血浆源性外泌体的特征 Fig 1 Characterization of plasma-derived exosomes from TAO patients and healthy controls |
为检测TAO患者血浆来源的外泌体是否能被HVSMC吸收和内化,用PKH67标记外泌体(绿色荧光)。外泌体与HVSMC共培养24 h和48 h后,多数HVSMC显示绿色荧光,共培养48 h后更多外泌体被HVSMC内化(图 1D)。结果表明,TAO患者血浆来源的外泌体可被HVSMC吸收和内化。
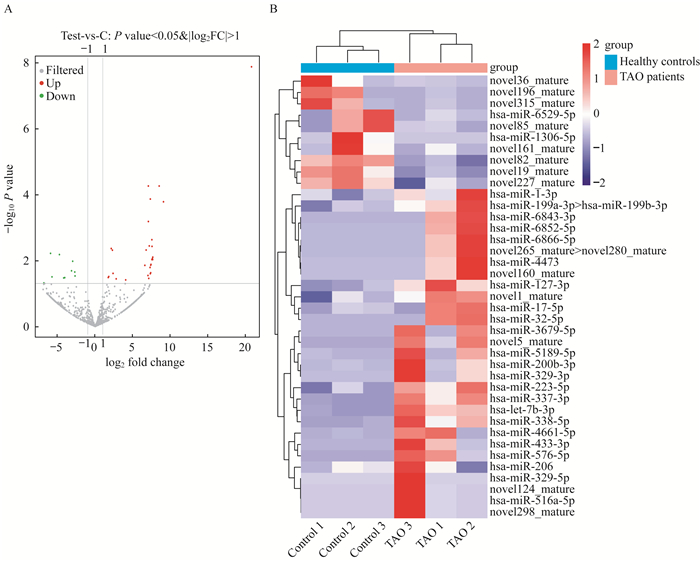
外泌体的MiRNA表达谱和DE-miRNA筛选 为确定可能参与TAO发病机制的外泌体miRNA,我们使用miRNA-seq分析来自TAO患者和健康对照的血浆外泌体miRNA谱。结果发现,所有样本产生的有效读数在12.08~16.03 moL,注释的miRNA占总RNA的8.88%~24.69%。共发现523个已知miRNA和309个新预测miRNA。基于{log2(fold change)}≥1.0原则,控制FDR < 0.05,比较TAO患者与健康对照者的外泌体miRNA,发现39个DE-miRNA,包括10个下调和29个上调的miRNA(图 2A)。DE-miRNA表达的热图分布如图 2所示。
|
| A: Volcano plot of DE-miRNAs. The dots above 1.3013 (-log0.05) and to the right of x=1 represented up-regulation; The dots above y=1.3013 (-log0.05) and to the left of x=-1 represented down-regulation. B: Heatmap of DE-miRNAs. 图 2 筛选TAO患者和健康对照之间的DE-miRNA Fig 2 Screening of DE-miRNAs between TAO patients and healthy controls |
DE-miRNAs的GO功能和KEGG途径富集分析 为解释TAO中已鉴定的DE-miRNA功能,使用miRanda数据库预测目标基因,共获得19 709个预测目标基因,进行GO功能和KEGG途径富集分析。GO-terms结果表明,已鉴定的DE-miRNA的功能与生物过程中的转录调控和信号转导密切相关,细胞成分包括细胞核、细胞质和细胞液,分子功能有金属离子结合、ATP结合和DNA结合(图 3A)。我们分析了前20个KEGG通路,发现TAO组和健康对照组的差异有统计学意义(P < 0.05,图 3B)。结果表明,这些DE-miRNA在MAPK信号通路、Rap1信号通路、cAMP信号通路、催产素信号通路和GnRH信号通路中显著富集。

|
| A: The top 30 significant GO terms (biological process, cellular component and molecular function); B: The top 20 significant KEGG pathways. Number: The count of genes significantly enriched in a pathway. 图 3 GO功能和KEGG富集分析用于已鉴定的DE-miRNA Fig 3 GO function and KEGG pathway enrichment analyses for the identified DE-miRNAs |
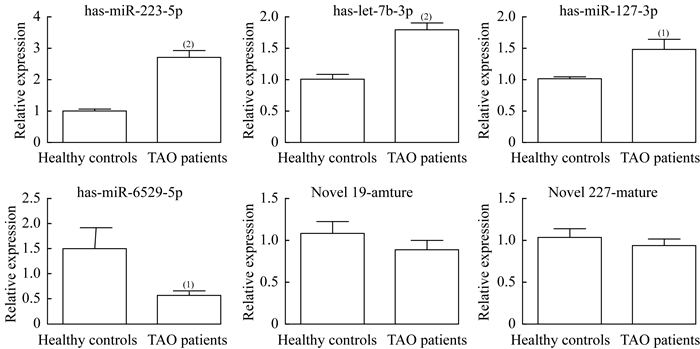
用RT-qPCR验证外泌体中DE-miRNA,选取前6个DE-miRNAs,包括3个上调miRNA(miR-223-5p、miR-let-7b-3p和miR-127-3p)和3个下调miRNA(miR-6529-5p、novel 19_mature和novel 227_mature),通过RT-qPCR进行验证。与对照组相比,TAO患者血浆源性外泌体中miR-223-5p、miR-let-7b-3p和miR-127-3p水平显著升高(P < 0.001,P < 0.001,P=0.035);而TAO患者血浆源性外泌体中miR-6529-5p水平显著降低(P=0.037,图 4)。在TAO患者和健康对照者的血浆源性外泌体中,未发现novel 19_mature和novel 227_mature(P=0.306,P=0.408,图 4)。结果显示,测序分析结果与RT-qPCR结果的一致性为66.67%,表明测序结果具有较高的可靠性。由于miR-223-5p较miR-let-7b-3p和miR-127-3p升高更明显,在接下来的实验中将予以重点关注。
|
| vs. Healthy controls, (1)P < 0.01, (2)P < 0.05. 图 4 RT-qPCR检测TAO患者和健康对照者血浆分离的外泌体中DE-miRNA表达水平 Fig 4 Expression level of DE-miRNAs in the exosomes isolated from the plasma of TAO patients and healthy controls by RT-PCR |
TAO患者血浆源性外泌体通过上调miR-223-5p抑制HVSMC的细胞活力和促进细胞凋亡。为证明TAO血浆来源的外泌体和miR-223-5p是否影响HVSMC的细胞活力和凋亡,使用miR-223-5p模拟物过表达miR-223-5p,使用miR-223-5p抑制剂阻断miR-223-5p表达。空白组和NC组之间的miR-223-5p水平差异无统计学意义(P=0.240,图 5A)。细胞与TAO患者血浆源性外泌体和miR-223-5p模拟物共同孵育后,miR-223-5p水平显著升高(P < 0.001),与NC组相比,miRNA模拟物组的miR-223-5p水平升高100倍以上(图 5A)。miR-223-5p抑制剂与TAO患者血浆源性外泌体结合后,外泌体诱导的miR-223-5p恢复至空白组水平(图 5)。检测VCAM-1和胰岛素样生长因子1受体(insulin-like growth factor-1 receptor,IGF-1R)的表达。发现空白组和NC组之间的VCAM-1和IGF-1R表达差异无统计学意义(P=0.248,P=0.418,图 5B、5C)。与空白组相比,外泌体组和miR-223-5p模拟组的VCAM-1(P=0.005,P=0.005)和IGF-1R(P=0.004,P=0.003)表达明显下调(图 5B、5C);miRNA抑制剂+外泌体组的VCAM-1表达明显高于外泌体组(P=0.011)和miR-223-5p模拟物组(P=0.004,图 5B);miRNA抑制剂+外泌体组的IGF-1R表达明显高于外泌体组(P=0.010)和miR-223-5p模拟物组(P=0.006,图 5C)。

|
| (1) vs. Blank group, P < 0.05; (2) vs. Exosome group, P < 0.05; (3) vs. miRNA mimic group, P < 0.05. 图 5 miR-223-5p和外泌体对不同处理的HVSMC的影响 Fig 5 Effects of miR-223-5p and exosomes on HVSMC with different treatments |
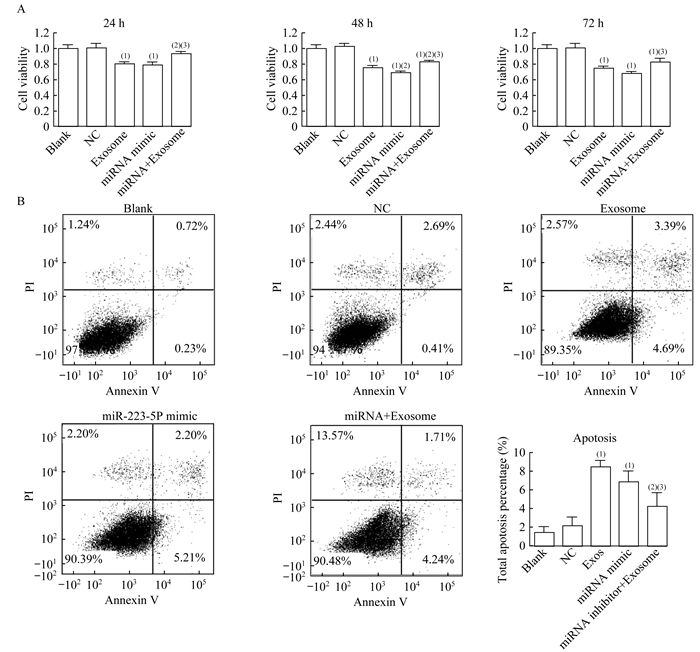
我们评估了不同处理对HVSMC细胞活力和凋亡的影响。与NC组相比,TAO血浆分离外泌体和miR-223-5p模拟物转染后24、48和72 h,HVSMC的细胞活力均被显著抑制(P均 < 0.001,图 6A)。在3个时间点,与单独使用外泌体处理相比,联合使用外泌体和miR-223-5p抑制剂显著恢复了细胞活力(P < 0.001,P < 0.001,P=0.011,图 6A),表明TAO患者血浆外泌体对HVSMC细胞活力的抑制作用是通过miR-223-5p的过表达介导的。流式细胞检测结果显示,与空白组相比,外泌体组和miR-223-5p模拟物组细胞凋亡显著增加(P=0.001,P=0.001,图 6B)。miR-223-5p抑制剂下调miR-223-5p,可显著改善TAO患者血浆外泌体诱导的细胞凋亡(P < 0.001,图 6B)。结果表明,TAO患者血浆外泌体诱导的细胞凋亡通过上调HVSMC的miR-223-5p介导。
|
| (1) vs. Blank group, P < 0.05; (2) vs. Exosome group, P < 0.05; (3) vs. miRNA mimics group, P < 0.05. 图 6 miR-223-5p和外泌体对不同处理的HVSMC细胞活力和凋亡的影响 Fig 6 Effects of miR-223-5p and exosomes on cell viability and apoptosis of HVSMCs with different treatments |
VCAM-1直接与miR-223-5p结合 TargetScan Human 7.1预测VCAM-1是miR-223-5p的下游基因(图 7A),因为VCAM-1的3’-UTR包含miR-223-5p的结合位点。采用双荧光素酶报告试验发现,转染NC模拟物的pGL3基本载体、转染miR-223-5p模拟物的pGL基本载体和转染NC模拟物的pGL-VCAM-1之间的相对荧光素酶活性差异无统计学意义(P=0.659,图 7)。在pGL-VCAM-1中,与转染NC模拟物相比,转染miR-223-5p后的相对荧光素酶活性显著降低(P=0.006,图 7)。结果表明,VCAM-1是miR-223-5p的下游靶点。
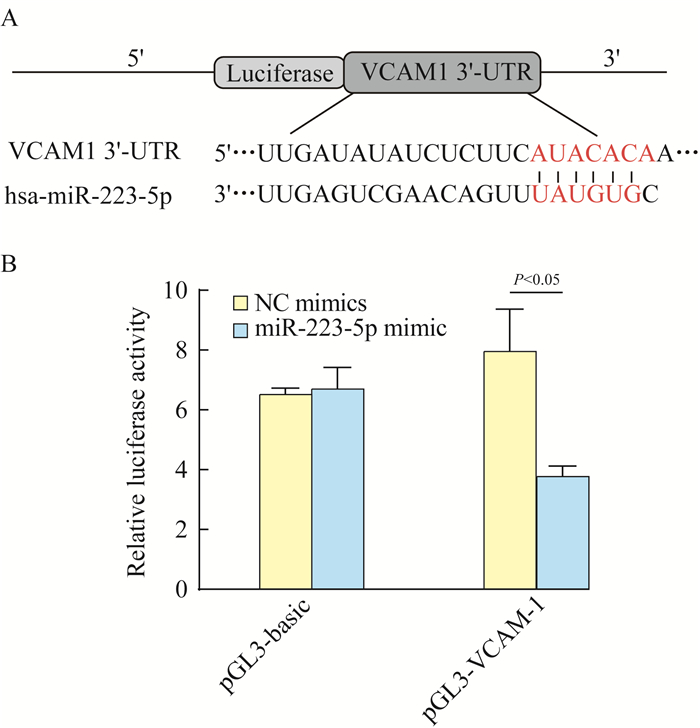
|
| 图 7 荧光素酶报告分析法测定miR-223-5p和VCAM-1之间的相互作用 Fig 7 Interaction between miR-223-5p and VCAM-1 determined by luciferase reporter assay |
外泌体miRNA在许多疾病的发生发展中起关键作用,被认为是可用于临床的潜在非侵入性生物标记物[26-27],现已证明循环外泌体miRNA对几种人类癌症的临床意义[28-29]。本研究关注TAO患者循环外泌体miRNA谱。我们从TAO患者和健康对照血浆中提取纯化外泌体,比较外泌体miRNA表达谱,发现39个DE-miRNA,在一定范围内富集并调控转录相关的一系列生物学过程和调控各种信号通路,如cAMP信号通路和MAPK通路,这与已知研究一致,表明这两种途径在炎症性疾病中发挥作用,并可能作为治疗靶点[30-31]。RT-qPCR进一步验证测序分析,表明测序结果具有较高的可靠性。
外泌体作为蛋白质和核酸的载体,可以将生物活性物质从供体细胞传递到受体细胞,从而影响受体细胞的生物功能[32]。我们观察到TAO患者血浆外泌体可被HVSMC吸收和内化,用其处理HVSMC可抑制其细胞活力并诱导凋亡。与空白组相比,TAO患者血浆外泌体可显著增加miR-223-5P水平和下调VCAM-1和IGF-1R。Ham等[33]指出,乳腺癌来源的外泌体可部分通过gp130/STAT3信号传导诱导巨噬细胞分泌IL-6和促生存表型。另一项研究发现,缺氧可促进胃癌细胞增殖、侵袭、迁移和上皮-间充质转化,从而促进胃癌进展和转移。结合本研究结果,推断TAO患者血浆来源的外泌体通过调节HVSMC的存活和凋亡,促进TAO发生和发展。
由于miR-223-5p上调作用明显,我们进一步探索外源性miR-223-5p对HVSMC的影响,发现miR-223-5p过表达显著抑制细胞活力并诱导细胞凋亡,而miR-223-5p下调可抑制TAO患者血浆外泌体诱导HVSMC细胞凋亡和细胞活力降低的作用。这表明miR-223-5p介导了TAO患者血浆外泌体对HVSMC的促凋亡作用。miR-223是一种抗炎性miRNA,主要存在于髓样细胞中,并参与转录后调节一组对炎症、细胞增殖和侵袭至关重要的基因[34]。血管平滑肌细胞中富含miR-223,其在血管平滑肌细胞增殖、迁移和血管重塑中的作用已明确[35-36]。miR-223也是外泌体中一个重要的miRNA,通过外泌体的细胞间转移参与受体细胞的生物功能调节[37-38]。在小鼠模型中miR-223-5p与脓毒症诱导的炎症和心肌功能障碍有关[39]。循环外泌体中的miR-223-5p可能是高血压的诊断生物标志物[40]。越来越多的证据表明,循环外泌体中的miR-223-5p或miR-223水平与香烟烟雾有关,香烟烟雾是导致TAO的关键因素[41]。本研究表明,TAO循环外泌体的miR-223-5p抑制HVSMC细胞活力并诱导细胞凋亡。因此,循环外泌体miR-223-5p可能在TAO的发生发展中起调节作用。
VCAM-1是参与炎症相关血管黏附的重要血管内皮炎症标记物,可能是一种血管功能障碍的生物标记物[42]。TAO患者增厚的血管内皮细胞和一些炎症细胞中VCAM-1表达增加[43]。人脐静脉内皮细胞中血小板衍生微粒刺激引起VCAM-1上调[44]。IGF-1R是胰岛素受体家族的一员,参与心脏和神经系统的多种生理功能[45]。斑块组织中IGF-1R表达降低与临床心血管事件有关[46]。血小板分泌的miR-223通过靶向IGF-1R促进内皮细胞凋亡[47]。Wang等[48]研究发现,血小板来源miR-223对动脉血栓形成的调节功能通过血管壁IGF-1R介导。暴露于TAO患者血浆外泌体后,HVSMC中VCAM-1和IGF-1R的表达显著下调,通过miR-223-5p抑制剂可使下调恢复。这些结果均表明,VCAM-1是miR-223的下游靶点。双荧光素酶报告分析证实VCAM-1直接与miR-223-5p结合。我们发现,TAO患者血浆外泌体通过miR223-5p/VCAM-1途径对HVSMC产生促凋亡活性,并介导IGF-1R表达。通过IL-6/信号转导和转录激活因子(signal transducer and activator of transcription 3,STAT3)途径,调节VCAM-1和细胞间黏附分子(intercellular adhesion molecule-1,ICAM-1)的表达[49]。miR-223通过靶向STAT3促进巨噬细胞产生IL-6[50]。我们推测,血浆外泌体miR-223-5p可能通过IL-6/STAT3途径,促进TAO发生发展。
本研究有一定的局限性:首先,样本量小,大样本量研究对于验证我们的发现至关重要。第二,通过超速离心分离外泌体,由于小而易碎的颗粒或沉淀效率,收集的外泌体数量并不一致,今后可应用分离纯度更高的新方法[32]。第三,从TAO患者和健康对照中分离出外泌体,由于外泌体的体内来源复杂,虽然HVSMC的细胞活力和凋亡受外泌体或miR-223-5p的影响,但尚未发现其分子机制和其他因素。
综上所述,TAO患者血浆来源的外泌体可抑制HVSMC的细胞活力并诱导细胞凋亡,其机制涉及靶向VCAM-1的外体miR-223-5p的上调。外泌体miR-223-5p可能在TAO的发病机制中发挥作用,为miR-223-5p/VCAM-1作为TAO治疗的新靶点和途径提供了参考。
作者贡献声明 陈波 数据采集,统计分析,论文撰写和修订。林学广 数据采集,统计分析。邓颖 数据分析和解释。王博 数据采集。童进东,余波 论文构思。史卫军 论文修订。汤敬东 论文构思和修订。
利益冲突声明 所有作者均声明不存在利益冲突。
| [1] |
YANG L, TANG J, TIAN Y, et al. The preliminary outcome of laser-assisted angioplasty combined with endovascular radiofrequency ablation for thromboangiitis obliterans[J]. Adv Ther, 2021, 38(12): 5700-5709.
[DOI]
|
| [2] |
SHAREBIANI H, FAZELI B, MANISCALCO R, et al. The imbalance among oxidative biomarkers and antioxidant defense systems in thromboangiitis obliterans (Winiwarter-Buerger disease)[J]. J Clin Med, 2020, 9(4): 1-15.
|
| [3] |
LE JONCOUR A, SOUDET S, DUPONT A, et al. Long-term outcome and prognostic factors of complications in thromboangiitis obliterans (Buerger's disease): a multicenter study of 224 patients[J]. J Am Heart Assoc, 2018, 7(23): 1-9.
|
| [4] |
LI MD, WANG YF, YANG MW, et al. Risk factors, mechanisms and treatments of thromboangiitis obliterans: an overview of recent research[J]. Curr Med Chem, 2020, 27(35): 6057-6072.
[DOI]
|
| [5] |
WANG Y, LIU Q, WANG F. Potential roles of exosome noncoding RNAs in cancer chemoresistance (Review)[J]. Oncol Rep, 2021, 45(2): 439-447.
|
| [6] |
HEIDARI N, ABBASI-KENARSARI H, NAMAKI S, et al. Adipose-derived mesenchymal stem cell-secreted exosome alleviates dextran sulfate sodium-induced acute colitis by Treg cell induction and inflammatory cytokine reduction[J]. J Cell Physiol, 2021, 236(8): 5906-5920.
[DOI]
|
| [7] |
SKURATOVSKAIA D, VULF M, KHAZIAKHMATOVA O, et al. Exosome limitations in the treatment of inflammatory diseases[J]. Curr Pharm Des, 2021, 27(28): 3105-3121.
[DOI]
|
| [8] |
MIGNEAULT F, DIEUDE M, TURGEON J, et al. Apoptotic exosome-like vesicles regulate endothelial gene expression, inflammatory signaling, and function through the NF-kappaB signaling pathway[J]. Sci Rep, 2020, 10(1): 1-15.
[DOI]
|
| [9] |
WANG L, JIA Q, CHEN X, et al. Role of cardiac progenitor cell-derived exosome-mediated microRNA-210 in cardiovascular disease[J]. J Cell Mol Med, 2019, 23(11): 7124-7131.
[DOI]
|
| [10] |
LIN X, ZHU T, XU F, et al. Plasma exosomes derived from patients with end-stage renal disease and renal transplant recipients have different effects on vascular calcification[J]. Front Cell Dev Biol, 2020, 8: 1-10.
[DOI]
|
| [11] |
LIN Y, ANDERSON JD, RAHNAMA L, et al. Exosomes in disease and regeneration: biological functions, diagnostics, and beneficial effects[J]. Am J Physiol Heart Circ Physiol, 2020, 319(6): H1162-H1180.
[DOI]
|
| [12] |
XU Z, ZENG S, GONG Z, et al. Exosome-based immunotherapy: a promising approach for cancer treatment[J]. Mol Cancer, 2020, 19(1): 1-16.
[DOI]
|
| [13] |
ZANG X, GU J, ZHANG J, et al. Exosome-transmitted lncRNA UFC1 promotes non-small-cell lung cancer progression by EZH2-mediated epigenetic silencing of PTEN expression[J]. Cell Death Dis, 2020, 11(4): 1-13.
|
| [14] |
MUGHEES M, KUMAR K, WAJID S. Exosome vesicle as a nano-therapeutic carrier for breast cancer[J]. J Drug Target, 2021, 29(2): 121-130.
[DOI]
|
| [15] |
HOSSEINI M, ROSHANGAR L, RAEISI S, et al. The therapeutic applications of exosomes in different types of diseases: a review[J]. Curr Mol Med, 2021, 21(2): 87-95.
[DOI]
|
| [16] |
LASSER C, ELDH M, LOTVALL J. Isolation and characterization of RNA-containing exosomes[J]. J Vis Exp, 2012(59): e3037: 1-6.
|
| [17] |
GURUNATHAN S, KANG MH, JEYARAJ M, et al. Review of the isolation, characterization, biological function, and multifarious therapeutic approaches of exosomes[J]. Cells, 2019, 8(4): 307.
[DOI]
|
| [18] |
ZHU L, SUN HT, WANG S, et al. Isolation and characterization of exosomes for cancer research[J]. J Hematol Oncol, 2020, 13(1): 1-37.
[DOI]
|
| [19] |
NAWROCKI EP, BURGE SW, BATEMAN A, et al. Rfam 12.0: updates to the RNA families database[J]. Nucleic Acids Res, 2015, 43(Database issue): D130-D137.
|
| [20] |
FRIEDLANDER MR, MACKOWIAK SD, LI N, et al. miRDeep2 accurately identifies known and hundreds of novel microRNA genes in seven animal clades[J]. Nucleic Acids Res, 2012, 40(1): 37-52.
[DOI]
|
| [21] |
GAO D, KIM J, KIM H, et al. A survey of statistical software for analysing RNA-seq data[J]. Hum Genomics, 2010, 5(1): 56-60.
[DOI]
|
| [22] |
ZHANG J, ZOU S, DENG L. Gene Ontology-based function prediction of long non-coding RNAs using bi-random walk[J]. BMC Med Genomics, 2018, 11(Suppl 5): 1-10. [CNKI]
|
| [23] |
KANEHISA M, SATO Y. KEGG Mapper for inferring cellular functions from protein sequences[J]. Protein Sci, 2020, 29(1): 28-35.
[DOI]
|
| [24] |
ROUSSELLE C, BARBIER M, COMTE VV, et al. Innocuousness and intracellular distribution of PKH67: a fluorescent probe for cell proliferation assessment[J]. In Vitro Cell Dev Biol Anim, 2001, 37(10): 646-655.
[DOI]
|
| [25] |
MAHGOUB EO, RAZMARA E, BITARAF A, et al. Advances of exosome isolation techniques in lung cancer[J]. Mol Biol Rep, 2020, 47(9): 7229-7251.
[DOI]
|
| [26] |
LI Z, YE L, WANG L, et al. Identification of miRNA signatures in serum exosomes as a potential biomarker after radiotherapy treatment in glioma patients[J]. Ann Diagn Pathol, 2020, 44: 151436.
[DOI]
|
| [27] |
ZHAO M, LI YP, GENG XR, et al. Expression level of miRNA-126 in serum exosomes of allergic asthma patients and lung tissues of asthmatic mice[J]. Curr Drug Metab, 2019, 20(10): 799-803.
[DOI]
|
| [28] |
SMOLARZ M, WIDLAK P. Serum exosomes and their miRNA load-A potential biomarker of lung cancer[J]. Cancers (Basel), 2021, 13(6): 1-20.
|
| [29] |
NIE H, XIE X, ZHANG D, et al. Use of lung-specific exosomes for miRNA-126 delivery in non-small cell lung cancer[J]. Nanoscale, 2020, 12(2): 877-887.
[DOI]
|
| [30] |
ESSAM RM, AHMED LA, ABDELSALAM RM, et al. Phosphodiestrase-1 and 4 inhibitors ameliorate liver fibrosis in rats: Modulation of cAMP/CREB/TLR4 inflammatory and fibrogenic pathways[J]. Life Sci, 2019, 222: 245-254.
[DOI]
|
| [31] |
MURAKI Y, NAITO T, TOHYAMA K, et al. Improvement of pulmonary arterial hypertension, inflammatory response, and epithelium injury by dual activation of cAMP/cGMP pathway in a rat model of monocrotaline-induced pulmonary hypertension[J]. Biosci Biotechnol Biochem, 2019, 83(6): 1000-1010.
[DOI]
|
| [32] |
DING L, YANG X, GAO Z, et al. A holistic review of the state-of-the-art Microfluidics for exosome separation: An overview of the current status, existing obstacles, and future outlook[J]. Small, 2021, 17(29): 1-19.
|
| [33] |
HAM S, LIMA LG, CHAI E, et al. Breast cancer-derived exosomes alter macrophage polarization via gp130/STAT3 signaling[J]. Front Immunol, 2018, 9: 1-10.
[DOI]
|
| [34] |
WANG Y, JIAO J, REN P, et al. Upregulation of miRNA-223-3p ameliorates RIP3-mediated necroptosis and inflammatory responses via targeting RIP3 after spinal cord injury[J]. J Cell Biochem, 2019.
[DOI]
|
| [35] |
ORENES-PINERO E, MARIN F, LIP GY. miRNA-197 and miRNA-223 and cardiovascular death in coronary artery disease patients[J]. Ann Transl Med, 2016, 4(10): 1-3.
|
| [36] |
SCHULTE C, MOLZ S, APPELBAUM S, et al. miRNA-197 and miRNA-223 predict cardiovascular death in a cohort of patients with symptomatic coronary artery disease[J]. PLoS One, 2015, 10(12): 1-12.
|
| [37] |
LI S, CHEN L, ZHOU X, et al. miRNA-223-3p and let-7b-3p as potential blood biomarkers associated with the ischemic penumbra in rats[J]. Acta Neurobiol Exp (Wars), 2019, 79(2): 205-216.
[DOI]
|
| [38] |
WAN L, YUAN X, LIU M, et al. miRNA-223-3p regulates NLRP3 to promote apoptosis and inhibit proliferation of hep3B cells[J]. Exp Ther Med, 2018, 15(3): 2429-2435.
|
| [39] |
WANG X, HUANG W, YANG Y, et al. Loss of duplexmiR-223 (5p and 3p) aggravates myocardial depression and mortality in polymicrobial sepsis[J]. Biochim Biophys Acta, 2014, 1842(5): 701-711.
[DOI]
|
| [40] |
WU X, YANG J, YU L, et al. Plasma miRNA-223 correlates with risk, inflammatory markers as well as prognosis in sepsis patients[J]. Medicine (Baltimore), 2018, 97(27): 1-6.
|
| [41] |
SHI X, XIE X, SUN Y, et al. Paeonol inhibits NLRP3 mediated inflammation in rat endothelial cells by elevating hyperlipidemic rats plasma exosomal miRNA-223[J]. Eur J Pharmacol, 2020, 885: 173473: 1-8.
|
| [42] |
TRONCOSO MF, ORTIZ-QUINTERO J, GARRIDO-MORENO V, et al. VCAM-1 as a predictor biomarker in cardiovascular disease[J]. Biochim Biophys Acta Mol Basis Dis, 2021, 1867(9): 166170: 1-13.
|
| [43] |
CARR RM. VCAM-1: closing the gap between lipotoxicity and endothelial dysfunction in nonalcoholic steatohepatitis[J]. J Clin Invest, 2021, 131(6): 1-3.
|
| [44] |
KONG LJ, WANG YN, WANG Z, et al. NOD2 induces VCAM-1 and ET-1 gene expression via NF-kappaB in human umbilical vein endothelial cells with muramyl dipeptide stimulation[J]. Herz, 2021, 46(Suppl 2): 265-271.
|
| [45] |
ESSANDOH K, DENG S, WANG X, et al. Tsg101 positively regulates physiologic-like cardiac hypertrophy through FIP3-mediated endosomal recycling of IGF-1R[J]. FASEB J, 2019, 33(6): 7451-7466.
[DOI]
|
| [46] |
SOHRABI M, FLODEN AM, MANOCHA GD, et al. IGF-1R inhibitor ameliorates neuroinflammation in an Alzheimer's disease transgenic mouse model[J]. Front Cell Neurosci, 2020, 14: 1-14.
|
| [47] |
HUANG K, DONG X, SUI C, et al. MiR-223 suppresses endometrial carcinoma cells proliferation by targeting IGF-1R[J]. Am J Transl Res, 2014, 6(6): 841-849.
|
| [48] |
WANG H, WANG Q, KLEIMAN K, et al. Hematopoietic deficiency of miR-223 attenuates thrombosis in response to photochemical injury in mice[J]. Sci Rep, 2017, 7(1): 1-7.
[DOI]
|
| [49] |
SONG F, JI B, CHEN T. Cilostazol on the expression of ICAM-1, VCAM-1 and inflammatory factors in plasma in patients with thromboangiitis obliterans[J]. Exp Ther Med, 2018, 16(3): 2349-2354.
|
| [50] |
CHEN Q, WANG H, LIU Y, et al. Inducible microRNA-223 down-regulation promotes TLR-triggered IL-6 and IL-1beta production in macrophages by targeting STAT3[J]. PLoS One, 2012, 7(8): 1-12.
|
 2022, Vol. 49
2022, Vol. 49


