2. 上海市影像医学研究所 上海 200032;
3. 复旦大学附属中山医院介入治疗科 上海 200032
2. Shanghai Institute of Medical Imaging, Shanghai 200032, China;
3. Department of Interventional Radiology, Zhongshan Hospital, Fudan University, Shanghai 200032, China
高强度聚焦超声(high-intensity focused ultrasound,HIFU)是一种非侵袭性的热源性消融方式,是目前晚期恶性肿瘤的主要姑息疗法之一[1-3]。在超声的实时引导下,HIFU治疗可实现肿瘤精准定位及无创消融。腹部局部晚期肿瘤由于病灶侵犯邻近重要组织不能进行根治性切除术。HIFU作为一种缓解性治疗,能有效缓解患者的癌性疼痛及缩小肿瘤体积,包括肝脏、胰腺及子宫等各种实体肿瘤[4-5]。行HIFU的恶性肿瘤患者多处于疾病晚期,此类患者生存率有限,因此在HIFU治疗前后,采用影像学方法早期、精准、无创地监测肿瘤内部微循环血流灌注的改变,对临床评估疗效、调整治疗方案具有重要的意义[5]。目前临床常用的新版实体瘤疗效评价标准(modified response evaluation criteria in solid tumors,mRECIST)基于CT或MRI的成像结果,但是CT具有一定的辐射,MRI价格昂贵且检查时间长,限制了其在短期内重复评估肿瘤HIFU治疗疗效中的应用[6]。基于CT/MRI的mRECIST标准主要关注病灶大小变化。既往研究显示,在HIFU治疗中,病灶大小变化发生在HIFU治疗后至少3个月[7-8]。二维超声是目前临床上运用最广泛的影像学检查方法之一,能无创评估治疗前后病灶大小的改变及病灶内部是否有坏死区存在,超声造影(contrast enhanced ultrasound,CEUS)能实时反应肿瘤内微循环血流灌注情况[9]。动态超声造影技术(dynamic contrast enhanced ultrasound,DCEUS)结合CEUS动态实时显示肿瘤内微循环灌注及定量分析的优势,能客观评估病灶内部的血流灌注情况,已经广泛用于评估恶性肿瘤放化疗及靶向治疗等疗法的疗效评估[10-11]。
本研究前瞻性地观察局部晚期腹部恶性肿瘤病灶HIFU治疗前后的改变,探讨DCEUS在随访HIFU疗效中的临床价值。
资料和方法研究对象 本研究为前瞻性临床研究,得到复旦大学附属中山医院伦理委员会批准(伦理编号:B2021-144)。纳入标准:(1)经病理及临床诊断为晚期腹部恶性肿瘤的患者;(2)实体瘤;(3)病灶在二维超声上显示清晰;(4)病灶周围无扩张的肠道等重要脏器;(5)计划接受标准疗程的HIFU治疗。排除标准:(1)病灶不能在二维超声上清晰显示;(2)患者不能耐受HIFU治疗,如出现心肺功能欠佳、过度肥胖、严重的出血倾向、腹水或者黄疸等症状;(3)患者CEUS检查配合欠佳;(4)CEUS检查过程存储时间不足2 min,缺乏完整的DICOM文件。
临床观察指标 随访并比较所有患者HIFU治疗前后血清癌胚抗原(carcinoembryonic antigen,CEA)、糖类抗原19-9(serum carcinoma antigen 19-9,CA19-9)及糖类抗原125(serum carcinoma antigen 125,CA125)水平。
研究流程 对所有经病理证实且计划进行5个标准疗程HIFU治疗的腹部恶性肿瘤患者进行随访,记录患者临床信息(性别、年龄、血液肿瘤标志物水平)。在HIFU治疗开始前一天及开始进行HIFU治疗后4周内分别进行CEUS检查并存储图像信息。
首先采用二维超声确定病灶的位置,观察病灶大小、边界、形态及内部回声,有无坏死区。彩色血流成像检测病灶内部血流情况。探头固定在能完整显示肿瘤的切面,切换至造影模式,注射对比剂后,观察病灶增强时间及强化方式,并存储至少2 min的病灶增强-消退过程待后续分析。动态图像存储为DICOM格式并导出。
随后用脱机的Veubox软件(意大利Bracco公司)分析,生成定量分析时间-强度曲线(time-intensity curve,TIC)。观察对比TIC的形态。经过曲线拟合,得到相关CEUS定量参数。对比HIFU治疗前后腹部恶性肿瘤病灶的大小、CEUS增强-消退表现、TIC、定量参数及肿瘤标记物是否存在差异。
仪器及对比剂 超声仪器包括:ACUSON OXANA2(德国Siemens公司,腹部探头6 C-1)、ACUSON Sequoia(德国Siemens公司,腹部探头5 C-1),探头频率为1~5 MHz。超声对比剂为SonoVue(意大利Bracco公司),对比剂的使用剂量为1.5 mL/次,经肘静脉团注,5 mL生理盐水冲管。
时间-强度曲线及定量参数的生成 采用Veubox定量分析软件对感兴趣区(region of interest,ROI)进行动态定量分析。分别在肿瘤区域及肿瘤周围正常组织设置2个ROI,保持2个ROI形状、大小及距体表深度相同,并注意避开周围主要大血管及瘤内坏死组织(图 1)。利用Veubox软件自动生成TIC,观察曲线形态、峰值强度及曲线下面积(area under curve,AUC)。
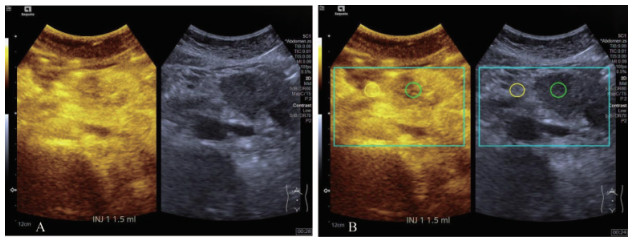
|
| A: Contrast enhanced ultrasound (CEUS) image of a pancreatic ductal adenocarcinoma (PDAC). B: Blue and green boxes represent respiratory compensation areas. Two regions of interest (ROIs) were placed within respiratory compensation areas, at the same depth and with the same shape. Green ROI and yellow ROI were placed within tumor area and surrounding normal tissue respectively. Then, the time intensity curve (TIC) was generated automatically. 图 1 超声造影定量分析感兴趣区放置位置 Fig 1 The placement of regions of interest during quantitative analysis of contrast enhanced ultrasound |
随后经过曲线拟合,当TIC拟合优度(quality of fit,QOF)超过75%时,所得的定量参数才有效并被纳入研究。得到相关的超声造影定量参数,包括峰值强度(peak enhancement,PE)、上升时间(rise time,RT)、达峰时间(time to peak,TTP)、平均渡越时间(mean transit time,mTT)及AUC。
高强度聚焦超声治疗 HIFU治疗仪器为HIFUNIT9000机型的聚焦超声肿瘤消融机(上海爱申科技发展股份有限公司),声强最大为12 000 W/cm2。治疗过程中患者取平卧位,超声定位于肿瘤部位,转换成内置探头,超声换能器个数选择至多6个,超声发射时间为200 ms,两次超声发射之间的间隔时间为400 ms,每个治疗点的超声发射次数为8。在工作界面勾画超声治疗靶区,根据患者对热的耐受程度,电功率调整在60%~100%,治疗完一个靶区再根据纵切或横切对肿瘤进行由左向右或自上而下的移动(每个治疗面的间隔为3 mm,每个治疗点/焦域范围为3 mm×3 mm×8 mm),再次勾画靶区后再进行超声治疗,依此类推。一次治疗时间共30 min,每个病灶治疗5次为一个疗程。
统计学分析 采用SPSS 20.0及GraphPad Prism 5进行统计分析。患者年龄、性别、肿瘤位置及诊断方法均采用描述性统计。计数资料采用Fisher确切概率法进行统计学分析。所有连续性变量采用x±s表达。利用Kolmogorov-Smirnov检验验证连续性变量的正态性,符合正态分布的变量用配对t检验,不符合正态分布的数据用Wilconox检验,验证其是否有显著差异。P < 0.05为差异有统计学意义。
结果患者临床信息 2018年9月至2021年5月共有19名腹部恶性肿瘤患者被纳入研究,其中女性10例,男性9例,年龄43~84岁,平均(58.84±10.08)岁。19个(穿刺活检/剖腹探查:15/4)病灶均为经病理证实的腹部恶性肿瘤且都经过5个标准疗程的HIFU治疗。病灶分别位于胰腺(11个)、肝脏(3个)、盆腔(4个)及腹膜后(1个)。
HIFU治疗前后随访患者血清肿瘤标志物水平,CEA [(31.01±82.51) ng/mL vs. (26.74±75.67) ng/mL]、CA19-9 [(1343.58±3073.21 U/mL vs. (1090.28±2802.51) U/mL]及CA125 [(30.18±24.48) U/mL vs. (36.32±61.83) U/mL]水平在治疗前后均无明显变化。
二维超声表现 HIFU治疗前后,19例腹部恶性肿瘤病灶在超声表现为低回声(n=18)或等回声(n=1)实质团块,边界欠清(n=13),形态不规则(n=10)。治疗前仅有15.79%(3/19)的病灶内可测及点状彩色血流信号。与治疗前相比,HIFU治疗后病灶大小未出现明显缩小[(55.42±22.24) mm vs. (53.16±25.81) mm,P>0.05,图 2]。

|
| A: Before high-intensity focused ultrasound (HIFU), a LAPC lesion showed heterogeneous hypoechoic solid lesion with irregular shape and unclear margin on B mode ultrasound. B-D: After injection of SonoVeu, the lesion showed heterogeneously hypoenhancement during arterial phase, venous and late phase.E-H: After HIFU treatment, the LAPC lesion showed similar features on B mode ultrasound and CEUS examination images. 图 2 高强度聚焦超声治疗前后局晚期胰腺癌二维超声及超声造影表现 Fig 2 Contrast enhanced ultrasound features of locally advanced pancreatic cancer before and after HIFU treatment |
超声造影表现 注射超声对比剂后,在HIFU治疗前,19例腹部恶性肿瘤病灶在动脉期主要表现为不均匀(n=19)的高回声增强(n=7)或低回声增强(n=9),静脉期/门脉期及延迟期均呈低回声增强(n=17)改变。4个病灶内部可见始终不增强的坏死区。与HIFU治疗前相比,治疗后腹部恶性肿瘤病灶的超声造影表现与治疗前没有明显差异(图 2)。
动态超声造影时间-强度曲线 所有病灶的CEUS过程均通过Vuebox定量分析软件自动生成TIC。所有TIC均包括上升支及下降支,上升支陡直,迅速达峰,下降支平缓,逐渐恢复至基线水平。同HIFU治疗前相比,治疗后腹部恶性肿瘤病灶TIC峰值强度降低,AUC减小(图 3)。
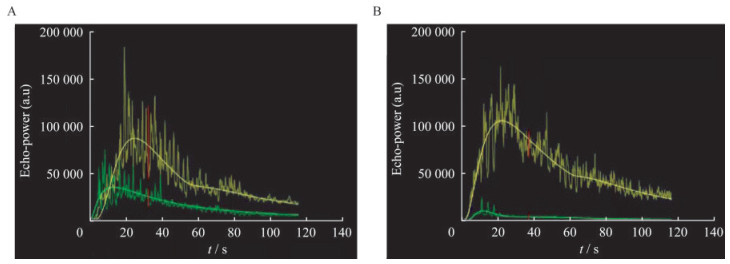
|
| Green curve and yellow curve represent microperfusion within tumor area and surrounding normal tissue respectively. A and B: While compared with TIC before HIFU treatment and after treatment, the peak enhancement and area under curve (AUC) of TIC within tumor area were smaller than before. 图 3 强度聚焦超声治疗前后时间-强度曲线比较 Fig 3 Comparison of time intensity curve between before and after HIFU treatment |
动态超声造影定量参数 所有TIC拟合度均大于75%。HIFU治疗前后CEUS定量参数比较结果显示,治疗后病灶的PE [(32920.63±24822.71) a.u vs. (17 296.11±16 570.41) a.u,P=0.001]及AUC[(624 665.28±593 711.71) a.u vs. (309 148.00±366 938.07) a.u,P=0.002]均显著低于治疗前,但RT、mTT及TTP较治疗前没有明显差异(表 1)。
| Parameters | Before treatment | After treatment | t/Z | P |
| PE (a.u) | 32 920.63±24 822.71 | 17 296.11±16 570.41 | t = 3.783 | 0.001 |
| RT (s) | 9.63±4.44 | 11.05±5.74 | Z = -0.783 | 0.434 |
| TTP (s) | 15.74±6.19 | 18.05±7.84 | Z = -1.008 | 0.314 |
| mTT (s) | 75.74±64.59 | 83.79±100.29 | Z = -0.362 | 0.717 |
| AUC (a.u) | 624 665.28±593 711.71 | 309 148.00±366 938.07 | Z = -3.139 | 0.002 |
| HIFU:High-intensity focused ultrasound;PE:Peak enhancement;RT:Rise time;TTP:Time to peak;mTT:Mean transit time;AUC:Area under curve. | ||||
HIFU作为一种新型的非侵袭性热消融方式,在缓解晚期腹部恶性肿瘤患者疼痛的同时,能有效减少肿瘤血供,达到抑制肿瘤生长的目的[7, 12]。在HIFU治疗过程中,高强度超声波在体内聚焦,局部温度能迅速升高至60 ℃以上,引起肿瘤细胞的凝固性坏死及局部微循环改变,在显微镜下能观察到病灶内多发散在的异常血管腔及血管炎,从而导致病灶灌注降低[5, 13]。
采用影像学方法,早期、精准、无创地评估HIFU治疗的疗效,对于指导晚期腹部恶性肿瘤患者的后续治疗方案具有重要价值。以往研究显示腹部恶性肿瘤病灶HIFU治疗后,CT及MRI短期随访肿瘤体积未见任何显著变化,术后6周肿瘤体积缩小约30%,术后3个月肿瘤体积缩小超过60%[7-8]。但是CT及MRI检查不能定量显示病灶内部微循环血流灌注的改变,且不建议在短期内多次重复检查[14]。
腹部超声凭借其无创、敏感、高空间分辨率等优势,能在肿瘤微创治疗前后短期内多次随访。研究证实,肿瘤热消融后短期内,往往在病灶大小还未发生显著变化时,就出现了内部微循环血流灌注的改变[15]。因此在消融治疗后早期、敏感、精准地评估肿瘤内部微循环血流灌注的变化,有助于及时监测疗效。在本组病例中,HIFU治疗前后对比,二维超声显示病灶大小及内部回声未见明显变化。而HIFU治疗后腹部恶性肿瘤病灶内部的超声造影定量参数PE及AUC均较治疗前明显降低。表明动态超声造影及定量分析技术能较常规超声更敏感地显示HIFU后肿瘤内部微循环血流灌注的改变。
低机械指数实时超声造影检查能观察到病灶内超声对比剂从进入到消退的全过程,为评估肿瘤病灶内的微循环血流灌注提供了有效手段[16-18]。然而常规的CEUS检查主要通过视觉评估病灶的增强-消退改变,对于HIFU治疗后短期内腹部恶性肿瘤病灶内血流评估缺乏敏感性。本研究中,HIFU治疗前后病灶增强消退方式未见明显改变,主要原因可能在于HIFU治疗前,病灶在CEUS动脉期表现为低增强;其次HIFU虽然能引起病灶内部微循环改变,除非病灶内部出现严重坏死,在CEUS上表现为未增强,否则HIFU引起的灌注改变不足以用肉眼观察。
基于超声造影的TIC,以曲线的形态直观反映ROI内的血流灌注情况[9, 19]。HIFU治疗过程中因局部高温及空化效应引起的肿瘤内部微循环灌注的改变,在本研究中表现为开始HIFU治疗后4周内,TIC峰值强度降低、AUC缩小。TIC的变化恰好可以通过病理表现来解释。TIC较常规CEUS更加直观、敏感地反映病灶内微循环血流灌注的变化。
通过进一步的曲线拟合,得到的超声造影定量参数中,HIFU治疗后腹部恶性肿瘤病灶PE及AUC均较治疗前明显降低。以往研究显示,超声造影定量参数PE及AUC不但与病灶内部的灌注强度呈正相关,而且与肿瘤患者预后相关[11]。而时间相关参数如RT、TTP及mTT则与对比剂微泡在ROI内的停留时间相关,同时还受对比剂的注射方式影响[20]。DCEUS及超声造影定量参数能定量、敏感地反映HIFU治疗后腹部恶性肿瘤病灶内灌注降低,为超声监测HIFU治疗后疗效提供了有效的手段。
动态超声造影作为一种无创、简便、可重复的影像学检查方式,可以有效、定量监测腹部恶性肿瘤病灶HIFU治疗后早期病灶内微循环血流灌注的改变,能在肿瘤治疗过程中提供一定的指导作用,PE及AUC是具有潜在意义的评估参数。
作者贡献声明 左丹 论文撰写和修订。刘凌晓 数据统计和分析,论文撰写。张琪,田晓梵 数据统计与分析。邱艺杰 数据采集及整理。王文平 论文构思与修订。董怡 研究设计,论文修订。
利益冲突声明 所有作者均声明不存在利益冲突。
| [1] |
KAMISAWA T, WOOD LD, ITOI T, et al. Pancreatic cancer[J]. Lancet, 2016, 388(10039): 73-85.
[DOI]
|
| [2] |
SIEGEL RL, MILLER KD, JEMAL A. Cancer statistics, 2020[J]. CA Cancer J Clin, 2020, 70(1): 7-30.
[DOI]
|
| [3] |
陆云飞, 徐伟, 丁小波, 等. 高强度聚焦超声治疗腹盆腔恶性实体肿瘤的疗效观察[J]. 中国社区医师, 2019, 35(35): 45-47. [CNKI]
|
| [4] |
CHEUNG VYT. High-intensity focused ultrasound therapy[J]. Best Pract Res Clin Obstet Gynaecol, 2018, 46: 74-83.
[DOI]
|
| [5] |
IZADIFAR Z, IZADIFAR Z, CHAPMAN D, et al. An introduction to high intensity focused ultrasound systematic review on principles, devices, and clinical applications[J]. J Clin Med, 2020, 9(2): 460.
[DOI]
|
| [6] |
EISENHAUER EA, THERASSE P, BOGAERTS J, et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1)[J]. Eur J Cancer, 2009, 45(2): 228-247.
[DOI]
|
| [7] |
STRUNK HM, HENSELER J, RAUCH M, et al. Clinical use of high-intensity focused ultrasound (HIFU) for tumor and pain reduction in advanced pancreatic cancer[J]. Rofo, 2016, 188(7): 662-670.
[DOI]
|
| [8] |
MARINOVA M, RAUCH M, MUCKE M, et al. High-intensity focused ultrasound (HIFU) for pancreatic carcinoma: evaluation of feasibility, reduction of tumour volume and pain intensity[J]. Eur Radiol, 2016, 26(11): 4047-4056.
[DOI]
|
| [9] |
DIETRICH CF, NOLSOE CP, BARR RG, et al. Guidelines and good clinical practice recommendations for contrast-enhanced ultrasound (CEUS) in the Liver-Update 2020 WFUMB in Cooperation with EFSUMB, AFSUMB, AIUM, and FLAUS[J]. Ultrasound Med Biol, 2020, 46(10): 2579-2604.
[DOI]
|
| [10] |
ZHANG Q, WU L, YANG D, et al. Clinical application of dynamic contrast enhanced ultrasound in monitoring the treatment response of chemoradiotherapy of pancreatic ductal adenocarcinoma[J]. Clin Hemorheol Microcirc, 2020, 75(3): 325-334.
[DOI]
|
| [11] |
DALAH E, TAI A, OSHIMA K, et al. PET-based treatment response assessment for neoadjuvant chemoradiation in pancreatic adenocarcinoma: an exploratory study[J]. Transl Oncol, 2018, 11(5): 1104-1109.
[DOI]
|
| [12] |
MARINOVA M, HUXOLD HC, HENSELER J, et al. Clinical effectiveness and potential survival benefit of US-guided high-intensity focused ultrasound therapy in patients with advanced-stage pancreatic cancer[J]. Ultraschall Med, 2019, 40(5): 625-637.
[DOI]
|
| [13] |
WU F, CHEN W Z, BAI J, et al. Pathological changes in human malignant carcinoma treated with high-intensity focused ultrasound[J]. Ultrasound Med Biol, 2001, 27(8): 1099-1106.
[DOI]
|
| [14] |
GUO X, ZHU H, ZHOU K, et al. Effects of high-intensity focused ultrasound treatment on peripancreatic arterial and venous blood vessels in pancreatic cancer[J]. Oncol Lett, 2020, 19(6): 3839-3850.
|
| [15] |
DAN Z, YI F, QI Z, et al. The value of dynamic contrast enhanced ultrasound (DCE-US) in monitoring treatment effect of high-intensity focused ultrasound (HIFU) in locally advanced pancreatic cancer (LAPC)[J]. Clin Hemorheol Microcirc, 2021, 77(3): 323-333.
[DOI]
|
| [16] |
HUDSON JM, WILLIAMS R, TREMBLAY-DARVEAU C, et al. Dynamic contrast enhanced ultrasound for therapy monitoring[J]. Eur J Radiol, 2015, 84(9): 1650-1657.
[DOI]
|
| [17] |
SIDHU PS, CANTISANI V, DIETRICH CF, et al. The EFSUMB guidelines and recommendations for the clinical practice of contrast-enhanced ultrasound (CEUS) in non-hepatic applications: update 2017 (long version)[J]. Ultraschall Med, 2018, 39(2): e2-e44.
[DOI]
|
| [18] |
曹佳颖, 范培丽, 董怡, 等. 二维与三维超声造影定量分析技术评估肝肿瘤的对比研究[J]. 中华超声影像学杂志, 2018, 27(5): 397-400. [DOI]
|
| [19] |
DIETRICH CF, AVERKIOU MA, CORREAS JM, et al. An EFSUMB introduction into Dynamic Contrast-Enhanced Ultrasound (DCE-US) for quantification of tumour perfusion[J]. Ultraschall Med, 2012, 33(4): 344-351.
[DOI]
|
| [20] |
WIESINGER I, BEYER LP, ZAUSIG N, et al. Evaluation of integrated color-coded perfusion analysis for contrast-enhanced ultrasound (CEUS) after percutaneous interventions for malignant liver lesions: first results[J]. Clin Hemorheol Microcirc, 2018, 69(1-2): 59-67.
[DOI]
|
 2022, Vol. 49
2022, Vol. 49


