随着外科技术和设备的发展进步,腹腔镜手术已经成为重要的腹部微创手术[1]。腹腔空间不足常会造成腹腔镜手术实施困难[2],有时甚至无法完成手术。高气腹压可增加腹腔空间但会抬高膈肌,降低肺顺应性,功能残气量及无效腔减少,引起通气血流比值失调[3]。越来越多的研究表明:与传统的中度肌松相比,腹腔镜手术中给予深度肌松能够提供更好的手术条件[4]。2016年,Kim等[5]进行的一项61人的腹腔镜结直肠外科手术研究中表明,深度神经肌肉阻滞(neuromuscular blockade, NMB)可以降低维持手术视野需要的CO2气腹压[深度NMB组为9.3 mmHg(1 mmHg=0.133 kPa, 下同),中度NMB组为14 mmHg]。之前的关于深度NMB应用于腹腔镜手术的多项研究,其主要探讨方向是手术条件的改善而非围术期患者恢复[6-9]。相对于中度NMB,深度NMB应用于腹腔镜手术患者恢复质量是否改善有待进一步研究[10-12]。目前尚未见不同NMB深度对腹腔镜结直肠手术恢复质量尤其是围术期肺氧合功能和术后肺部并发症影响的报道。既往研究表明,腹腔镜手术常伴随的较长手术时间、特殊手术体位、气腹引起的高腹腔压力及高碳酸血症等因素均会加重对肺组织的损伤,增加术后肺部并发症的发生率[13]。我们假设与中度肌松相比,深肌松条件下术中维持的气腹压更低,对胸腔容积、肺顺应性等方面影响更小,继而改善围术期肺氧合功能,减少手术后肺部并发症发生率。本研究旨在观察和比较高剂量罗库溴铵与常规剂量罗库溴铵所引起的不同深度的NMB对腹腔镜结直肠手术一般手术条件、围术期肺氧合功能和术后肺损伤的影响,为进一步临床应用提供依据。
资料和方法一般资料 本研究经复旦大学附属肿瘤医院医学伦理委员会批准(伦理号:1503145-4)并取得所有患者的知情同意。以复旦大学附属肿瘤医院2017年6月—2018年3月收治的76例行腹腔镜结直肠手术患者为研究对象,排除标准(满足以下任意一项即予排除):患者年龄 < 18岁或 > 75岁;美国麻醉医师协会(American Society of Anesthesiologists,ASA)分级≥Ⅲ级;BMI < 18.5 kg/m2或≥35 kg/m2; 既往神经肌肉疾病或者恶性高热病史;重要脏器的衰竭;严重限制性肺病史; 严重慢性阻塞性肺病(GOLD Ⅲ级或Ⅳ级)史; 严重或未控制的支气管哮喘病、肺部感染或支气管扩张史;手术前使用正压通气(如阻塞性睡眠呼吸暂停综合征持续气道正压通气);胸廓畸形和胸内疾病;需要在手术后转入ICU;急性冠状动脉综合征或持续性室性快速性心律失常;拒绝或无法给予知情同意的个人;参与者在另一项介入性研究中既往腹部手术史;对舒更葡糖钠或罗库溴铵过敏[6-8, 10]。由计算机生成随机化表(在https://www.randomizer.org/form.htm获取),将76名受试者随机分为两组:深度NMB组(A组)38人和中度NMB组(B组)38人。所有手术均由同一组外科医师完成。
麻醉准备、诱导与维持 患者入手术室行生命体征监测,包括心电图,有创动脉压、脉搏血氧饱和度及麻醉深度监测。全身麻醉诱导采用丙泊酚靶控(使用Marsh药代动力学参数,气管插管时效应室靶浓度Ce为2.5 μg/mL)复合瑞芬太尼(0.2 μg·kg-1·min-1)输注及舒芬太尼0.3~0.4 μg/kg,罗库溴铵(美国默沙东公司,爱可松@注射液)0.6 mg/kg。术中全身麻醉维持采用七氟烷(0.6~0.8 MAC)和丙泊酚持续靶控输注(Ce=1.0 μg/mL),按需追加舒芬太尼,根据肌松监测情况间断给予罗库溴铵维持肌松,根据手术要求调整至合适的麻醉深度。术中维持麻醉深度指数(narcotrend index,NTI)为27~46,且血压波动在术前值的±20%以内。
机械通气、肌松监测与管理 根据预定义的公式[14]计算患者的理想体重(IBW):男性为50+0.91×[身高(cm)-152.4],女性为45.5+0.91×[身高(cm)-152.4]。机械通气设定为容量控制通气模式,潮气量设定为8 mL/kg[15],无呼气末正压(positive end-expiratory pressure,PEEP),FiO2设定为0.5,两组吸气与呼气比设定为1:2,使用Drager Fabius麻醉机(德国)进行通气。调节呼吸频率(以10次/min开始)以使呼末二氧化碳(end-tidal CO2, ET CO2)保持在35~45 mmHg之间。维持通气模式直至手术结束。
使用TOF-watch-SX监测仪的肌加速度图描记法监测神经肌肉功能。患者一侧上肢外展固定,表面皮肤清洁后粘贴电极于腕部尺神经,传感器探头置于拇示指间,并保持其间无任何阻力。温度探头贴于手掌面,维持术中掌面温度≥32 ℃。两组患者诱导前均进行检测仪的校准定标。随即启动肌松监测。诱导后通过间断滴定给药使患者达到各自的目标肌松监测深度。A组给予持续5 s的50 Hz超强刺激,间隔3 s后改为1 Hz的单刺激,即强直刺激后单刺激计数(post-tetanic count,PTC)监测。每隔6 min实施一次PTC,维持PTC为0~2;B组首先给予4个频率2 Hz、波宽0.2~0.3 ms的成串刺激,连续刺激的时串间距离为12 s,即TOF监测。维持TOF值为1~2。
两组患者均在麻醉诱导后(T1)、气腹后10 min(T2)、气腹后1 h(T3)、气腹结束后10 min(T4)共4个时间点记录呼吸参数,包括:潮气量(tidal volume,TV),气道峰压(airway peak pressure,Ppeak), PEEP等,计算动态肺顺应性(dynamic lung compliance,Cdyn)=TV/(Ppeak-PEEP)。在麻醉诱导前、T2、T3、T4共4个时间点记录患者平均动脉压及心率。在T1、T3、拔管后30 min(T5)和术后6 h(T6)记录动脉血气分析参数,包括:pH,PaCO2和PaO2。计算围术期氧合指数(oxygenation index, OI)=PaO2/FiO2。手术结束前均静脉给予盐酸格拉司琼注射液6 mg预防恶心呕吐,并予氟比洛芬酯50 mg镇痛。手术接近结束缝皮时,两组患者均静脉注射舒更葡糖钠,A组剂量4 mg/kg,B组剂量2 mg/kg。术毕待患者4次成串刺激比值(train-of-four-ratios, TOFR)恢复至90%以上且评估良好后拔管。术后使用患者静脉自控镇痛(patient-controlled analgesia,PCA),含凯纷1 mg/mL,舒芬太尼0.5 μg/mL,背景剂量4 mL/h,PCA剂量4 mL,锁定时间15 min。在苏醒阶段、苏醒后1~6 h、6~24 h、24~48 h共4个时间段记录患者的自觉疼痛评分。记录患者术后第1天(D2)及第6天(D7)的肺部并发症情况,出院后60天通过电话回访,跟踪调查患者后续恢复情况。
观察指标 由外科医师进行单盲整体手术条件评分,包括手术视野和肌肉松弛程度,分为极差(1分)、差(2分)、可接受(3分)、好(4分)和理想(5分),共5个等级;记录围手术期术中气腹压(intra-abdominal pressure,IAP)、呼吸和心血管参数等;计算并记录T1、T2、T3、T4共4个时间点Cdyn以及T1、T3、T5、T6 4个时间点OI数值;记录手术时和手术后患者疼痛情况,如腹部疼痛程度(使用疼痛数字化评价量表,numerical rating scale,NRS)、肩部疼痛的发生率等;记录肠功能恢复参数,如胃肠道排气时间。另外还观察了术后住院时间、术后D2和D7的肺部并发症(定义为符合以下6项发现中的3种或以上:咳嗽,分泌物增多,呼吸困难,胸痛或不适,体温高于38℃,HR高于100次/分[15])。记录D2和D7两组间术后肺部并发症(postoperative pulmonary complications,PPCs)以及30天内再住院率等。外科医师、评估手术条件评分和检查术后变量的研究者对患者分组不知情。
统计学方法 采用SPSS21.0统计学软件对数据进行分析。符合正态分布的定量数据资料以x±s表示,定性数据以频率和百分比形式表示。组间比较采用t检验;计数资料以百分数表示,采用χ2检验。P < 0.05为差异有统计学意义。
结果一般资料 深度NMB组(A组)和中度NMB组(B组)患者在性别、年龄、身高、体重、BMI、ASA分级等方面差异均无统计学意义(P > 0.05),表明均衡可比(表 1)。
| Items | Group A (n=38) |
Group B (n=38) |
P |
| Age (y) | 58.2±8.1 | 57.7±8.9 | 0.911 |
| Gender [n(%)] | |||
| Male | 20 (52.6) | 19 (50.0) | 0.180 |
| Female | 18 (47.4) | 19 (50.0) | 0.180 |
| Height (cm) | 163.8±7.4 | 163.4±6.9 | 0.842 |
| Body weight (kg) | 61.2±8.5 | 62.9±9.1 | 0.178 |
| BMI (kg/m2) | 22.8±1.3 | 23.4±1.3 | 0.507 |
| ASA [n(%)] | |||
| Ⅰ | 24 (63.2) | 21 (55.3) | 0.151 |
| Ⅱ | 14 (36.8) | 17 (44.7) | 0.171 |
| Surgical technic [n(%)] | |||
| Laparoscopic right hemicolectomy | 14 (36.8) | 16 (42.2) | 0.194 |
| Laparoscopic Dixon | 15 (39.5) | 14 (36.8) | 0.180 |
| Laparoscopic Miles | 8 (21.1) | 6 (15.8) | 0.200 |
| Laparoscopic left hemicolectomy | 1 (2.6) | 2 (5.2) | 0.390 |
| Group A:Patients treated with deep neuromuscular blockade; Group B:Patients treated with moderate neuromuscular blockade. Data are reported as means±SD. | |||
两组患者围手术期参数比较 与B组相比,A组整体手术评分显著增加(P < 0.01),满足手术所需的平均IAP显著降低(P < 0.01)。两组患者平均心率和血压差异无统计学意义(P > 0.05,表 2)。
| Items | Group A(n=38) | Group B(n=38) | P | |
| Mean IAP (mmHg) | 8.7±1.2 | 12.0±0.6 | < 0.01 | |
| Overall surgical condition score | 4.61±0.12 | 3.92±0.09 | < 0.01 | |
| Operative time (min) | 205.3±64.0 | 258.3±75.0 | 0.312 | |
| Mean heart rate (once/min) | Pre-anesthesia | 72.1±10.1 | 72.2±11.4 | 0.960 |
| 10 mins after pneumoperitoneum | 69.6±13.3 | 69.5±15.2 | 0.992 | |
| 1 h after pneumoperitoneum | 63.3±9.5 | 68.7±8.7 | 0.139 | |
| 10 mins after pneumoperitoneum end | 63.0±9.4 | 66.3±8.9 | 0.141 | |
| Average blood pressure (mmHg) | Pre-anesthesia | 96.7±10.6 | 93.9±11.1 | 0.356 |
| 10 mins after pneumoperitoneum | 85.3±9.4 | 81.9±10.5 | 0.309 | |
| 1 h after pneumoperitoneum | 86.6±7.7 | 87.1±14.4 | 0.691 | |
| 10 mins after pneumoperitoneum end | 81.7±8.4 | 82.8±10.3 | 0.787 | |
| Group A:Patients treated with deep neuromuscular blockade; Group B:Patients treated with moderate neuromuscular blockade. Data are reported as means±SD. | ||||
两组患者手术后恢复质量参数比较 与B组相比,A组腹部疼痛评分(NRS法)显著降低(P < 0.05)。A组胃肠道通气时间缩短(P < 0.05)。两组患者在术后肩部疼痛、苏醒室时间、术后住院时间、30天内再住院率方面差异无统计学意义(P > 0.05,表 3)。
| Items | Group A(n=38) | Group B(n=38) | P | |
| Abdominal pain score(NRS method) | Recovery stage | 2.6±0.5 | 3.3±0.4 | < 0.05 |
| 1-6 h after recovery | 3.1±0.9 | 4.2±0.8 | < 0.05 | |
| 6-24 h after recovery | 2.5±0.6 | 4.1±1.1 | < 0.05 | |
| 24-48 h after recovery | 1.9±0.9 | 3.5±0.8 | < 0.05 | |
| Shoulder pain cases [n(%)] | Recovery stage | 3 (7.9) | 8 (21.1) | 0.07 |
| 1-6 h after recovery | 5 (13.2) | 11 (29.0) | 0.06 | |
| 6-24 h after recovery | 5 (13.2) | 10 (26.3) | 0.08 | |
| 24-48 h after recovery | 1 (2.6) | 3 (7.9) | 0.250 | |
| PACU time (min) | 45.1±11.2 | 47.9±15.9 | 0.397 | |
| Gas passing time (hours) | 22.3±12.4 | 36.7±14.4 | < 0.05 | |
| Postoperative hospital stay (days) | 5.2±2.2 | 5.3±2.3 | 0.662 | |
| 30-day readmission [n(%)] | 1 (2.6) | 3 (7.9) | 0.250 | |
| Group A:Patients treated with deep neuromuscular blockade; Group B: Patients treated with moderate neuromuscular blockade.Data are reported as means±SD. | ||||
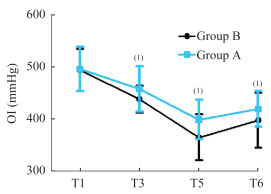
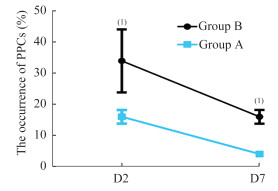
两组患者围术期肺氧合功能及PPCs比较 与T1相比,Cdyn在T2均降低(P < 0.05); 与B组相比,A组在T3-T4时Cdyn明显增加(P < 0.05,图 1)。与T1相比,两组T3、T5和T6的OI值较低(P < 0.05),而A组在T3、T5和T6的OI值高于B组(P < 0.05,图 2)。与B组相比,A组PPCs发生率降低(P < 0.05,图 3)。

|
| Cdyn:Intraoperative pulmonary dynamic compliance; T1:After induction of anesthesia.T2:10 min after pneumoperitoneum; T3:60 min after pneumoperitoneum; T4:10 min after pneumoperitoneum stopped. Group A:Deep neuromuscular blockade; group B:moderate neuromuscular blockade.Data are reported as means±SD.(1)P < 0.05, vs.group B (t-test). 图 1 两组患者术中动态肺顺应性比较 Fig 1 Comparison of intraoperative pulmonary dynamic compliance in the two groups |
|
| Oxygenation index (OI) during the operative (T1, after induction of anesthesia; T3, 60 min after pneumoperitoneum) and postoperative period (T5, 30 min after extubation; T6, 6-hour after surgery) in patients treated with deep neuromuscular blockade (group A) and moderate neuromuscular blockade(group B).Data are reported as means±SD.(1)P < 0.05 vs. with group B (t-test). 图 2 两组患者围术期氧合指数比较 Fig 2 Differences in perioperative oxygenation index in the two groups |
|
| PPCs:Occurrence of postoperative pulmonary complications.Group A:Deep neuromuscular blockade; Group B:Moderate neuromuscular blockade.Data are reported as means±SD.(1)P < 0.05 vs.group B (chi-squared test). 图 3 两组患者术后肺部并发症比较 Fig 3 Comparison of postoperative pulmonary complications in the two groups |
腹腔镜手术一般可以通过增加气腹压、调整患者体位和深度肌松3种常用方式改善手术的操作视野。增加气腹压会引起病理生理学改变,例如心血管系统、肺通气和内脏灌注方面的副作用[2],增加术后肩部疼痛等。因此,腹腔镜手术中气腹压应限制在一定范围内,临床一般为8~12 mmHg,推荐使用适合手术视野的最小气压,而非常规气压[16]。有研究认为深度NMB可减少气腹压力,减少手术应激反应和手术后疼痛,同时扩大手术视野,改善手术条件[17-18]。随着舒更葡糖钠(一种修饰的γ-环糊精,为罗库溴铵逆转剂)在临床上的应用,高剂量罗库溴铵引起的深度神经肌肉阻滞在腹腔镜手术中的应用范围也在逐渐扩大[19]。
膈肌的肌松水平对腹腔镜手术条件有重要影响。其收缩和意外活动会引起腹腔压力和脏器位置的改变,影响外科医师的手术操作,甚至可能导致器械刺破脏器或血管而造成严重的手术并发症[20]。临床常使用拇收肌作为肌松监测中评估的参考肌肉,膈肌肌松效应的持续时间比拇收肌短约30%,会引起麻醉医师对患者肌松程度的误判[21]。当加深NMB程度时肌松剂的剂量增加可能无法使TOF计数继续变化,因此,临床可使用PTC监测。Martini等[22]在系统化评估深度及中度NMB对腹腔镜手术条件的影响时提出,PTC < 3可认为深度肌松,TOF 1~2认为中度肌松。
Blobner等[23]报道认为,深度肌松可提高腹腔镜胆囊切除术视野评分,并使围手术期不良事件的绝对风险降低44%。Bruintjes等[24]的Meta分析纳入了12个随机对照研究,结果表明,高剂量罗库溴铵引起的深度NMB能改善多种手术方案的手术条件。其中多数研究的评分系统均结合手术空间、肌肉收缩、处理策略等,做成数字化的评分系统; 所有研究中,高剂量罗库溴铵组的评分平均改善约20%。在本研究中,深度NMB组整体手术评分显著增加(P < 0.01),提示高剂量罗库溴铵的使用达到了改善手术视野和获得良好手术条件的目的。满足手术所需的平均IAP显著降低(P < 0.01),将气腹压限制在较低范围内,降低了气腹压引起的内脏损伤和心血管系统方面的副作用。
Boon等[25]在分析260例深度和中度NMB对腹腔镜手术的围术期影响时发现:多种手术方案中,深度NMB组在血流动力学、手术时间、麻醉时间、手术后恢复和手术后疼痛均优于中度NMB组。腹腔镜手术后疼痛的原因是复杂的,可能涉及高气腹压力、组织损伤、二氧化碳激活腹膜伤害感受器、组织促炎细胞因子的表达以及腹肌顺应性减弱等。本研究结果表明深度NMB组腹部疼痛评分降低(P < 0.05),提示深度NMB能够减少手术后腹部疼痛,但对肩部疼痛的影响不大(P > 0.05),其原因可能是气腹压力的降低以及深肌松条件下腹腔压力对腹壁损伤减小,其具体机制仍待深入研究。NMB组肠道通气时间缩短(P < 0.05),可能是由于深度NMB减少了机体的应激反应,促进了胃肠道功能的恢复。
多种因素影响人正常肺功能,如气道通畅、胸廓完整、肺组织顺应性及血流灌注良好等[26]。外科手术操作不当可能引起膈神经受损,术后引流管放置位置不佳一定程度上会影响膈肌活动,术后疼痛以及肠腔内积气、积液可导致呼吸运动受限、肺部排痰能力减弱从而增加肺部并发症发生率,对肺功能造成损害[10, 14, 18]。
腹腔内高气腹压会对机体呼吸功能造成较显著影响,其可使膈肌上移,降低膈肌活动度,进而增加胸腔压力,限制肺扩张,直接或间接增加肺不张发生率[27]。而肺不张是引起术后低氧血症的主要原因之一,其可引发一系列术后不良反应如肺炎及呼吸衰竭等[28]。当气腹压力为10 mmHg时,有研究显示压力增加5 mmHg,其气道阻力、平台压及气道峰压增加12%、12.3%及5.5%[29-30];当气腹压大于14 mmHg,患者肺顺应性下降45%,气道峰压上升50%[31]。本研究结果显示深度NMB能够增加OI(P < 0.05)及Cdyn(P < 0.05),同时也降低了PPCs(P < 0.05)。可能原因考虑与中度NMB相比,深度NMB条件下术中维持的气腹压更低,对胸腔容积、肺顺应性以及通气血流比值等方面影响减小,肺不张及急性肺损伤发生率减少,围术期相关肺损伤更小。此外,前述研究结果表明深度NMB改善手术条件,提高手术质量,可能会减少术中膈肌及膈神经受损发生率;且实验结果提示深度NMB改善患者术后疼痛及胃肠道积气、积液,减少以上原因对术后呼吸运动、肺部排痰能力等方面的影响,间接改善围术期肺损伤。但受限于样本量和实验设计,还需要进一步研究来阐述深度NMB相较中度NMB减少围术期相关肺损伤的机制。
术后肌松残留可能减弱患者术后肺部排痰能力,并引起肺泡有效通气量不足、上呼吸道梗阻及反流误吸的风险升高,导致患者围术期肺损伤发生率增加[32]。肌松拮抗有助于降低术后24 h呼吸系统并发症的发生风险[33]。相对于国内普遍使用的胆碱酯酶抑制剂新斯的明,罗库溴铵特异性拮抗剂舒更葡糖钠的肌松逆转作用更为显著,5 min内便可有效逆转罗库溴铵引起的深度NMB,其临床应用使得深肌松手术后呼吸恢复延迟的风险被降低,大幅减少了肌松残余对患者术后的影响,便于麻醉医师针对不同的病例和手术方案实施个体化肌松管理,在多种手术中获得稳定高质量的手术空间[34-35]。肥胖患者的腹腔手术存在更为严峻的手术空间不足的问题,采用深度NMB联合舒更葡糖钠,能够改善手术空间,同时缩短手术时间,提高手术效率[35]。本研究采用舒更葡糖钠作为拮抗剂,联合围术期肌松监测,确保拔管前患者TOFR > 0.9,以减少肌松残留对患者围术期肺功能的影响。
本研究不足之处在于样本量较少,评估时间较短,且不同外科医师对手术条件的评价可能存在偏倚。后续外科医师如能在手术后回顾手术视频并就整体评价达成一致或可减少评价者偏倚。
综上所述,本研究发现,使用罗库溴铵诱导,舒更葡糖钠拮抗的深度NMB能够改善手术条件及围术期肺氧合功能,减少手术后PPCs。其也可降低术中IAP和减少手术后腹部疼痛,促进术后胃肠道功能恢复,为临床推广使用深度NMB进行腹腔镜结直肠手术提供了理论依据。
| [1] |
VOLZ J, KÖSTER S, SPACEK Z, et al. The influence of pneumoperitoneum used in laparoscopic surgery on an intraabdominal tumor growth[J]. Cancer, 1999, 86(5): 770-774.
[URI]
|
| [2] |
EBIBO LR, DHAHRI A, VERHAEGHE P, et al. What to do when it is technically impossible to perform laparoscopic sleeve gastrectomy[J]. Obes Surg, 24(12): 2069-2074.
[URI]
|
| [3] |
OGURLU M, KUCUK M, BILGIN F, et al. Pressure-controlled vs volume-controlled ventilation during laparoscopic gynecological surgery[J]. Minim Invasive Gynecol, 2010, 17(3): 295-300.
[DOI]
|
| [4] |
NEIRA VM, KOVESI T, GUERRA L, et al. The impact of pneumoperitoneum and Trendelenburg positioning on respiratory system mechanics during laparoscopic pelvic surgery in children:a prospective observational study[J]. Can J Anesth, 2015, 62(7): 798-806.
[URI]
|
| [5] |
KIM MH, LEE KY, LEE KY, et al. Maintaining optimal surgical conditions with low insufflation pressures is possible with deep neuromuscular blockade during laparoscopic colorectal surgery:a prospective, randomized, double-blind, parallel-group clinical trial[J]. Medicine (Baltimore), 2016, 95(9): e2920.
[URI]
|
| [6] |
STAEHR-RYE AK, RASMUSSEN LS, ROSENBERG J, et al. Surgical space conditions during low-pressure laparoscopic cholecystectomy with deep versus moderate neuromuscular blockade:a randomized clinical study[J]. Anesth Analg, 2014, 119(5): 1084-1092.
[URI]
|
| [7] |
BARRIO J, ERRANDO CL, GARCIA-RAMON J, et al. Influence of depth of neuromuscular blockade on surgical conditions during low-pressure pneumoperitoneum laparoscopic cholecystectomy:a randomized blinded study[J]. J Clin Anesth, 2017, 42: 26-30.
[URI]
|
| [8] |
BAETE S, VERCRUYSSE G, VANDER LAENEN M, et al. The effect of deep versus moderate neuromuscular block on surgical conditions and postoperative respiratory function in bariatric laparoscopic surgery:a randomized, double blind clinical trial[J]. Anesth Analg, 2017, 124(5): 1469-1475.
[URI]
|
| [9] |
KOO BW, OH AY, SEO KS, et al. Randomized clinical trial of moderate versus deep neuromuscular block for lowpressure pneumoperitoneum during laparoscopic cholecystectomy[J]. World J Surg, 2016, 40(12): 2898-2903.
[DOI]
|
| [10] |
ÖZDEMIR-VAN BRUNSCHOTDMD, SCHEFFERGJ, JAGTMVAN DER, 等. Quality of recovery after low-pressure laparoscopic DONOR nephrectomy facilitated by deep neuromuscular blockade:a randomized controlled study[J]. World J Surg, 2017, 41(11): 2950-2958. [DOI]
|
| [11] |
KOPMAN AF, NAGUIB M. Laparoscopic surgery and muscle relaxants:is deep block helpful?[J]. Anesth Analg, 2015, 120(1): 51-58.
[URI]
|
| [12] |
PARK JS, AHN EJ, KO DD, et al. Effects of pneumoperitoneal pressure and position changes on respiratory mechanics during laparoscopic colectomy[J]. Korean J Anesthesiol, 2012, 63(5): 419-424.
[URI]
|
| [13] |
DUGGAN M, KAVANAGH BP. Pulmonary atelectasis:a pathogenic perioperative entity[J]. Anesthesiology, 2005, 102(4): 838-854.
[URI]
|
| [14] |
GULDNER A, KISS T, SERPA NETO A, et al. Intraoperative protective mechanical ventilation for prevention of postoperative pulmonary complications:a comprehensive review of the role of tidal volume, positive end-expiratory pressure, and lung recruitment maneuvers[J]. Anesthesiology, 2015, 123(3): 692-713.
[URI]
|
| [15] |
FARTOUKH M, MAITRE B, HONORE S, et al. Diagnosing pneumonia during mechanical ventilation the clinical pulmonary infection score revisited[J]. Am J Respir Crit Care Med, 2003, 168(2): 173-179.
[URI]
|
| [16] |
NEUDECKER J, SAUERLAND S, NEUGEBAUER E, et al. The European Association for Endoscopic Surgery clinical practice guideline on the pneumoperitoneum for laparoscopic surgery[J]. Surg Endosc, 2002, 16(7): 1121-1143.
[URI]
|
| [17] |
HUA J, GONG J, YAO L, et al. Low-pressure versus standard-pressure pneumoperitoneum for laparoscopic cholecystectomy:a systematic review and meta-analysis[J]. Am J Surg, 2014, 208(1): 143-150.
[URI]
|
| [18] |
KOC M, ERTAN T, TEZ M, et al. Randomized, prospective comparison of postoperative pain in low-versus high-pressure pneumoperitoneum[J]. ANZ J Surg, 2005, 75(8): 693-696.
[URI]
|
| [19] |
BOGANI G, UCCELLA S, CROMI A, et al. Low vs standard pneumoperitoneum pressure during laparoscopic hysterectomy:prospective randomized trial[J]. J Minim Invasive Gynecol, 2014, 21(3): 466-471.
[URI]
|
| [20] |
AMMAR AS, MAHMOUD KM, KASEMY ZA. A comparison of sugammadex and neostigmine for reversal of rocuronium-induced neuromuscular blockade in children[J]. Acta Anaesthesiol Scand, 2017, 61(4): 374-380.
[URI]
|
| [21] |
FAGERLUND MJ, FINK H, BAUMÜLLER E, et al. Postanaesthesia pulmonary complications after use of muscle relaxants in Europe:Study protocol of the POPULAR study[J]. Eur J Anaesthesiol, 2016, 33(5): 381-383.
[URI]
|
| [22] |
MARTINI CH, BOON M, BEVERS RF, et al. Evaluation of surgical conditions during laparoscopic surgery in patients with moderate vs deep neuromuscular block[J]. Br J Anaesth, 2014, 112(3): 498-505.
[URI]
|
| [23] |
BLOBNER M, FRICK CG, STÄUBLE RB, et al. Neuromuscular blockade improves surgical conditions(NISCO)[J]. Surg Endosc, 2015, 29(3): 627-636.
[URI]
|
| [24] |
BRUINTJES M, HELDEN EVVAN, BRAAT AE, et al. Deep neuromuscular block to optimize surgical space conditions during laparoscopic surgery:a systematic review and meta-analysis[J]. Br J Anaesth, 2017, 118(6): 834-842.
[URI]
|
| [25] |
BOON M, MARTINI C, YANG HK, et al. Impact of high-versus low-dose neuromuscular blocking agentadmini-stration on unplanned 30-day readmission rates in retroperitoneal laparoscopic surgery[J]. PLoS One, 2018, 13(5): e0197036.
[DOI]
|
| [26] |
NOJIRI T, INOUE M, YAMAMOTO K, et al. Inhaled tiotropium to prevent postoperative cardiopulmonary complications in patients with newly diagnosed chronic obstructive pulmonary disease requiring lung cancer surgery[J]. Surg Today, 2014, 44(2): 285-290.
[URI]
|
| [27] |
IMBERGER G, MCLLROY D, PACE NL, et al. Positive end-expiratory pressure (PEEP) during anaesthesia for the prevention of mortality and postoperative pulmonary complications[J]. Cochrane Database Syst Rev, 2010, 9: CD007922.
[URI]
|
| [28] |
NGUYEN NT, ANDERSON JT, BUDD M, et al. Effects of pneumoperitoneum on intraoperative pulmonary mechanics and gas exchange during laparoscopic gastric bypass[J]. Surg Endosc, 2004, 18(1): 64-71.
[URI]
|
| [29] |
PARK JS, AHN EJ, KO DD, et al. Effects of pneumoperitoneal pressure and position changes on respiratory mechanics during laparoscopic colectomy[J]. Korean J Anesthesiol, 2012, 63(5): 419-424.
[URI]
|
| [30] |
GU WJ, WANG F, LIU JC. Effect of lung-protective ventilation with lower tidal volumes on clinical outcomes among patients undergoing surgery:a meta-analysis of randomized controlled trial[J]. CMAJ, 2015, 187(3): E101-E109.
[PubMed]
|
| [31] |
吴新民. 麻醉过程中使用肌松药及其拮抗剂的必要性[J]. 临床药物治疗杂志, 2019, 17(6): 32-34. [URI]
|
| [32] |
MCLEAN DJ, DIAZ-GIL D, FARHAN HN, et al. Dose-dependent association between intermediate-acting neuromuscular-blocking agents and postoperative respiratory complications[J]. Anesthesiology, 2015, 122(6): 1201-1213.
[URI]
|
| [33] |
DE SOUZA CM, TARDELLI MA, TEDESCO H, et al. Efficacy and safety of sugammadex in the reversal of deep neuromuscular blockade induced by rocuronium in patients with end-stage renal disease:A comparative prospective clinical trial[J]. Eur J Anaesthesiol, 2015, 32(10): 681-686.
[URI]
|
| [34] |
RAHE-MEYER N, BERGER C, WITTMANN M, et al. Recovery from prolonged deep rocuronium-induced neuromuscular blockade:A randomized comparison of sugammadex reversal with spontaneous recovery[J]. Anaesthesist, 2015, 64(7): 506-512.
[URI]
|
| [35] |
EL-RAHMAN AMA BD, OTHMAN AH, SHERIF FAEL, et al. Comparison of three different doses sugammadex based on ideal body weight for reversal of moderate rocuronium-induced neuromuscular blockade in laparoscopic bariatric surgery[J]. Minerva Anestesiol, 2017, 83(2): 138-144.
[URI]
|
 2020, Vol. 47
2020, Vol. 47


