2. 上海市影像医学研究所 上海 200032
2. Shanghai Institute of Medical Imaging, Shanghai 200032, China
胰腺癌恶性程度较高,预后较差,患者5年总体生存率低于5%,中位生存期小于6个月[1]。外科手术切除是目前唯一有可能治愈胰腺癌的治疗手段。约80%的患者在确诊时已属晚期,因肿瘤侵犯周围重要解剖结构或发生远处转移,无法接受手术切除[2]。全身化疗仍是晚期胰腺癌的主要治疗方法,但是以吉西他滨为基础的传统方案的中位生存期仅为6.8个月[3]。新型FOLFIRINOX方案(奥沙利铂、伊立替康、氟尿嘧啶和亚叶酸钙)使中位生存期延长至11.1个月,但是较重的毒性反应和副作用限制了该方案的广泛应用[4]。因此,我们仍需不断探索新的治疗策略,提高晚期胰腺癌的疗效。
射频消融(radiofrequency ablation, RFA)通过射频电流引起离子震荡后产热,可以使肿瘤组织产生凝固性坏死,作为肿瘤热消融的一种,在肝、肺、肾等实体肿瘤治疗中取得了良好的治疗效果[5-6]。由于胰腺解剖学位置和结构的特殊性,目前胰腺癌的RFA临床应用明显落后于其他实质脏器肿瘤[7-8]。针对胰腺消融特殊性设计出的Habib EUS RFA针(英国EMcision公司),比传统消融针更细,并发症可能相对较少[7]。目前Haibib EUS RFA针尚无国内应用的报道。本研究拟在CT引导下使用Haibib EUS RFA针治疗晚期胰腺癌,评价Haibib EUS RFA治疗晚期胰腺癌的可行性和安全性,并初步观察其治疗效果,为RFA治疗晚期胰腺癌提供临床依据。
资料和方法病例资料 选择2016年5月至2019年1月于复旦大学附属中山医院介入治疗科接受CT引导下RFA治疗的晚期胰腺癌患者。纳入标准:(1)已完成1~3次动脉灌注化疗的晚期胰腺癌患者;(2)获得病理学依据;(3)年龄18~75岁;(4)胰腺病灶未接受过放疗、碘125粒子植入等局部治疗;(5)ECOG评分≤2分;(6)凝血酶原时间延长<3 s;(7)白细胞计数>4×109/L,血小板计数>85×109/L;(8)血肌酐<115 μmol/L。排除标准:(1)有胰腺穿刺禁忌证;(2)无法控制的感染;(3)患者有其他肿瘤病史;(4)预计生存期<3个月。本研究获得复旦大学附属中山医院伦理委员会批准(R2019-136),所有患者均签署知情同意书。
所有患者治疗前行肝、肾功能、血常规、凝血功能、血清淀粉酶及CA-199的检查。同时术前行增强CT或增强MRI确定胰腺内肿瘤位置、大小及周围脉管侵犯情况。
主要器材Habib EUS RFA针(英国EMcision公司)直径1Fr (0.33 mm),长度190 cm,工作(消融)段长度8 mm(图 1A); 21G超声穿刺针(美国Cook公司)(图 1A); Rita 1500 RF发生器(美国Mountain View公司)(图 1B)。

|
| A:The Habib EUS RFA probe comprises a 1 Fr wire (0.33 mm) with a working length of 190 cm and the length of electrically active part of the probe is 8 mm (arrow); the Habib EUS RFA probe can be safely passed through the biopsy channel (related 21G fine needle) (circle) into the tumor lesion with CT guidance.B:A Rita 1500 RF-generator (RITA Medical systems, Mountain View, CA, USA) for RFA. 图 1 胰腺消融所用的Habib EUS RFA针及相关的21G穿刺针 Fig 1 The Habib EUS RFA probe and related 21G FNA needle for pancreatic cancer RFA |
治疗方法 患者仰卧位,CT扫描后根据病灶所在位置和周围解剖结构确定穿刺点和进针角度。穿刺点局部麻醉,用21G超声穿刺针经皮穿刺进入病灶中央部位,并接近病灶底部,距病灶底部约5 mm(图 2B)。撤出21G超声穿刺针针芯,送入Habib EUS RFA针,消融针头端和21G超声穿刺针齐平。固定Habib EUS RFA针,回撤21G超声穿刺针约2 cm,CT扫描确认RFA导管位置(图 2C)。连接Rita 1500 RF发生器,功率5 W,消融时间90 s。60 s后同时撤除RFA导管和超声穿刺针,再次CT扫描观察有无并发症(图 2D)。

|
| A: A locally advanced pancreatic cancer (white arrow) detected on pancreatic body by the enhanced abdominal CT scan before RFA; B: Introduce the 21G FNA needle (white arrow) by CT guidance; C:The Habib EUS RFA probe (white arrow) exposed directly at the treatment area and verify positioning by CT scan; D:No bleeding was verified by CT scan after RFA. 图 2 CT引导下Habib EUS RFA治疗胰腺癌的过程 Fig 2 The procedure of CT-guided Habib EUS RFA for pancreatic cancer |
如病灶直径>3 cm,则行单针道两点消融。第一点消融结束后,同时回撤RFA导管和超声穿刺针约8 mm,用同样参数进行第二点消融。
治疗后处理及观察指标 术后1天复查肝、肾功能、血常规、血清淀粉酶。为预防感染和急性胰腺炎,所有患者均接受抗生素输注(手术前3 h口服头孢曲松1 g,每日2次,连续3天)和预防(术前3 h奥曲肽0.1 mg,每日3次,连续3天)。
主要观察指标:Habib EUS RFA治疗晚期胰腺癌的技术成功率及相关并发症。次要观察指标:Habib EUS RFA的治疗效果。
随访 所有患者术后1个月复查肝肾功能、血常规、CA-199以及腹部增强CT/MRI(图 2A),观察病灶消融区域治疗后改变。
统计学方法 计量资料采用x±s表示,用独立样本或配对样本t检验进行分析;计数资料用百分比(%)表示,用χ2检验进行比较。通过Fisher精确检验比较分类变量组。使用传统的线性最小二乘回归进行相关。使用SPSS统计软件19.0进行分析,P<0.05为差异有统计学意义。
结果基本信息 从2016年5月到2019年1月,6名(4名男性,2名女性)晚期胰腺癌患者纳入本研究,平均年龄58.2岁(43~66岁)。患者的基本资料见表 1。3例病灶位于胰头部,2例位于胰体部,1例位于胰尾部。胰腺肿瘤长轴的平均长度为32.7 mm(23~55 mm)。所有患者病理检查均证实为胰腺导管腺癌。所有患者RFA治疗之前,均接受1~3次以吉西他滨为基础的动脉灌注化疗。RFA治疗后,所有患者继续接受以吉西他滨为基础的动脉灌注化疗。
| Patient | Gender | Age(y) | Site | Size (mm) | Application time (s) | Ablation points |
| 1 | Female | 43 | Tail | 35×30 | 90 | 2 |
| 2 | Male | 66 | Body | 28×24 | 90 | 1 |
| 3 | Female | 65 | Body | 23×21 | 90 | 1 |
| 4 | Male | 56 | Head | 55×52 | 90 | >2 |
| 5 | Male | 62 | Head | 30×25 | 90 | 2 |
| 6 | Male | 57 | Head | 25×15 | 90 | 1 |
可行性与安全性 6例患者均在CT引导下成功实施了RFA。3例患者行单针道单点消融,2例患者行单针道两点消融,1例患者行多针道多点消融。在手术期间和术后,所有患者的生命参数(血压、心率、氧合)均稳定。所有患者治疗后均无严重的治疗相关不良事件(如急性胰腺炎、出血,胰瘘等)发生。2例患者出现RFA后腹痛,2天后自行缓解。1例患者消融后淀粉酶水平升高,无任何严重急性胰腺炎的迹象,对症处理后2天内恢复到正常范围。其他血生化指标较术前无明显变化(表 2)。
| Patient | Hemoglobin (g/dL) | White blood cells (109/L) | Amylase (U/L) | IL-6 (pg/mL) | |||||||
| Before | After | Before | After | Before | After | Before | After | ||||
| 1 | 11.5 | 11.3 | 5.6 | 6.4 | 36 | 42 | 4.5 | 28 | |||
| 2 | 9.5 | 9.6 | 6.5 | 6.3 | 26 | 28 | 3.6 | 27 | |||
| 3 | 12.5 | 12.6 | 4.8 | 5.0 | 38 | 66 | 3.2 | 3.2 | |||
| 4 | 13.1 | 13.0 | 6.6 | 7.6 | 56 | 112 | 31 | 38 | |||
| 5 | 9.8 | 10.1 | 7.2 | 5.2 | 59 | 32 | 3.7 | 13 | |||
| 6 | 10.6 | 10.2 | 6.9 | 3.8 | 25 | 28 | 28 | 18 | |||
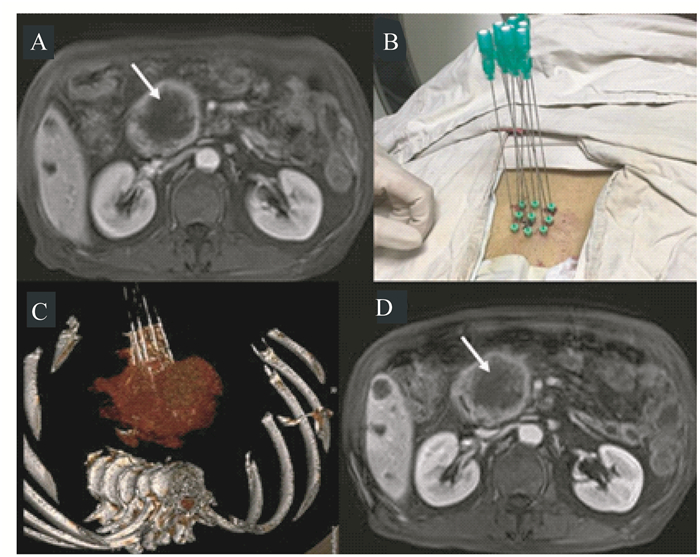
术后1个月影像学随访显示,Habib EUS RFA针在晚期胰腺癌病灶中形成低密度坏死区(图 3~4)。其中1例患者的胰头癌直径达到5.5 cm(图 4A),我们采用CT引导下多点进针消融肿块(图 4B、4C),1个月后MRI随访,病灶中心区域也出现了明显的坏死区(图 4D)。

|
| A:CT showed a low-density mass (white arrow) at the pancreatic tail before RFA; B:CT-guided RFA was performed with 5 W of ablation power for 90 s; C:After RFA, CT scan showed air bubbles (white arrow) in the lesions; D:1 month after RFA, CT showed a decrease of the low-density mass (white arrow). 图 3 1例胰尾癌行单点消融及1个月的随访结果 Fig 3 Single-point RFA and one-month follow-up for a patient with pancreatic cancer at the tail of the pancreas |
|
| A:MRI showed mild enhancement of lesions at the head of the pancreas (white arrow) before RFA; B:CT-guided RFA was performed at multiple points in sequence, with 5 W of ablation power for 90 s; C:3D CT reconstruction for multi-point ablation; D:1 month after RFA, MRI showed necrosis in ablation area (white arrow). 图 4 1例胰头癌行多点消融以及1个月的随访结果 Fig 4 Multi-point RFA and one-month follow-up for a patient with pancreatic cancer at the head of the pancreas |
RFA治疗晚期胰腺癌不能起到根治效果,但是具有以下治疗作用[9-12]:(1)通过肿瘤局部坏死,减轻肿瘤负荷,延长患者生存期;(2)具有一定的镇痛效果,减少镇痛药物摄入量;(3)激活机体抗肿瘤免疫。但是,胰腺是一个解剖上非常特殊的脏器, 体积较小,位于腹腔深部,周围有胃肠道、胆道、腹腔干、肠系膜上动脉和门静脉等重要组织结构。胰腺组织极易损伤,这些因素导致晚期胰腺癌RFA穿刺定位困难,容易引起胰腺炎、胰瘘、门脉静血栓形成、十二指肠穿孔、胆瘘等并发症。因此,RFA治疗晚期胰腺癌的临床应用远远落后于其他实体肿瘤。
外科手术中,RFA可以选择更为有利的消融穿刺途径;超声显像更为清晰;术中可以采取相应措施防止热损伤;可以即时处理热消融引起的并发症,目前已成为晚期胰腺癌RFA最常用的治疗途径[13]。2014年之前公开发表的胰腺癌RFA临床报道,病例数大于5例、随访资料较为完整的研究均采用手术途径[14]。由于外科手术本身创伤较大,在降低RFA相关并发症的同时,增加了外科手术相关并发症。据报道,手术途径RFA治疗晚期胰腺癌的RFA并发症发生率约占总体并发症的50%[14-15]。
超声内镜(endoscopic ultrasound, EUS)是将内镜和超声相结合的消化道检查技术,近年来超声内镜下的介入性治疗已经是国内外内镜技术的热点之一。EUS导引RFA治疗晚期胰腺癌时,消融针通过内窥镜途径送达胃腔或十二指肠内,经胃壁或肠壁穿刺胰腺病灶,损伤小于外科术中RFA。超声探头和胰腺仅间隔一层胃壁或肠壁,可以清晰显示胰腺肿瘤,而且超声导引可以实时监测消融进展情况,根据消融范围的变化及时调整消融参数[16]。多项小样本的前瞻性临床研究均证实了EUS导引RFA治疗晚期胰腺癌的可行性和安全性[17-21]。EUS导引RFA可以安全地在胰腺病灶内产生凝固性坏死区,术后并发症通常较轻且具有自限性,无需临床干预,例如轻度腹痛、少量腹水、一过性血淀粉酶升高等[20-21]。
考虑到EUS引导下RFA操作距离较长(Haibib EUS RFA针长达190 cm),操作技术较为复杂,通常只能进行一个针道消融[20]。而Haibib EUS RFA针直径仅0.33 mm,可以通过21G的穿刺针,理论上穿刺通过除动脉以外的组织脏器,均不会产生并发症,为经皮穿刺提供了安全性和便捷性。因此,我们尝试改变穿刺途径,在CT导引下将Haibib EUS RFA针经皮穿刺治疗晚期胰腺癌。
本研究CT引导下经皮穿刺RFA的成功率为100%,所有患者治疗后均无严重的治疗相关不良事件(如急性胰腺炎、出血,胰瘘等)发生。本研究中使用的Habib EUS RFA针直径仅为1F,相当于0.33 mm,直径小于常用的传统射频针(18~19G,约1.07~1.27 mm)。其可以通过22G穿刺针,穿刺系统直径较外科手术中的消融针小,因此可以明显减少穿刺相关并发症[14-15],为经皮穿刺提供了安全性和便捷性。此外胰腺组织对热损伤敏感,动物实验表明,Haibib EUS RFA针在正常胰腺组织中的消融范围(直径6~8 mm)[19, 22]也较普通消融针小,对正常胰腺组织损伤更小,所有动物均未出现消化系统症状和行为异常[19]。本研究中,胰腺癌患者RFA治疗后仅出现血淀粉酶一过性升高,术后5天即降至正常,本研究结果和动物实验保持一致,仅是一过性淀粉酶升高。Pai等[23]使用Habib EUS RFA针在EUS引导下治疗8例胰腺肿瘤患者,其中6例为囊性肿瘤,2例为神经内分泌肿瘤。治疗后3~6个月,2例囊性肿瘤病灶完全消失,4例直径缩小48.4%。2例神经内分泌肿瘤血供明显减少并伴有中央坏死。所有患者均未出现严重并发症。表明Habib EUS消融针在不同的引导途径中安全性都较传统RFA针好。
CT导引穿刺可以同时建立多个针道,使有效范围较小的治疗方法应用于体积较大的肿瘤,例如碘125粒子植入术[24],这是EUS导引无法达到的。虽然RFA治疗晚期胰腺癌并不要求达到肿瘤完全坏死的目的,但8 mm的消融直径是远远不够的。本研究中1例患者胰头癌的直径达到5.5 cm,我们采用多点进针的方法(图 4),扩大了Haibib EUS RFA针的消融范围,使其可以用于体积较大的瘤体。一方面拓展了Haibib EUS RFA针的适用范围,另一方面也为体积较大的胰腺癌提供了安全有效的局部治疗手段。
本研究中的6例病例术后1个月的影像随访资料显示,在患者的病灶消融区域观察到低密度坏死区,证明RFA对晚期胰腺肿瘤有减轻肿瘤负荷的效果。虽然本研究对患者进行了后期随访,但是由于病例数较少且部分发生远处转移,因此需要入组更多的病例,进一步研究患者的生存获益。局部消融治疗可以缓解部分患者因胰腺肿瘤引起的疼痛。6例患者中有4例RFA后疼痛明显缓解,但是维持时间不到1个月,之后患者是否需要再次接受RFA治疗,需要进一步研究。
综上所述,CT引导的Habib EUS RFA可用于晚期胰腺癌的治疗。RFA术为不能切除的胰腺癌患者提供了一种新的治疗方法,并有望成为其综合治疗的一部分。
| [1] |
HIDALGO M. Pancreatic cancer[J]. N Engl J Med, 2010, 362(17): 1605-1617.
[DOI]
|
| [2] |
ARCIDIACONO PG, CARRARA S, RENI M, et al. Feasibility and safety of EUS-guided cryothermal ablation in patients with locally advanced pancreatic cancer[J]. Gastrointest Endosc, 2012, 76(6): 1142-1151.
[DOI]
|
| [3] |
GILLEN S, SCHUSTER T, MEYER ZUM BUSCHENFELDE C, et al. Preoperative/neoadjuvant therapy in pancreatic cancer: a systematic review and meta-analysis of response and resection percentages[J]. PLoS Med, 2010, 7(4): e1000267.
[DOI]
|
| [4] |
CONROY T, DESSEIGNE F, YCHOU M, et al. FOLFIRINOX versus gemcitabine for metastatic pancreatic cancer[J]. N Engl J Med, 2011, 364(19): 1817-1825.
[DOI]
|
| [5] |
HARA K, TAKEDA A, TSURUGAI Y, et al. Radiotherapy for hepatocellular carcinoma results in comparable survival to radiofrequency ablation: a propensity score analysis[J]. Hepatology, 2019, 69(6): 2533-2545.
[URI]
|
| [6] |
HYUN MH, CHUNG SM. Randomized, controlled trial of percutaneous cryoablation versus radiofrequency ablation in hepatocellular carcinoma[J]. Hepatology, 2015, 62(3): 980-987.
[URI]
|
| [7] |
TATLI S, TAPAN U, MORRISON PR, et al. Radiofrequency ablation: technique and clinical applications[J]. Diagn Interv Radiol, 2012, 18(5): 508-516.
[URI]
|
| [8] |
GIRELLI R, FRIGERIO I, SALVIA R, et al. Feasibility and safety of radiofrequency ablation for locally advanced pancreatic cancer[J]. Br J Surg, 2010, 97(2): 220-225.
[URI]
|
| [9] |
VARSHNEY S, SEWKANI A, SHARMA S, et al. Radiofrequency ablation of unresectable pancreatic carcinoma: feasibility, efficacy and safety[J]. JOP, 2006, 7(1): 74-78.
[URI]
|
| [10] |
HAEN SP, PEREIRA PL, SALIH HR, et al. More than just tumor destruction:immunomodulation by thermal ablation of cancer[J]. Clin Dev Immunol, 2011, 20(11): 160-165.
[URI]
|
| [11] |
YANG W, WANG W, LIU B, et al. Immunomodulation characteristics by thermal ablation therapy in cancer patients[J]. Asia Pac J Clin Oncol, 2018, 14(5): e490-e497.
[DOI]
|
| [12] |
高珊珊, 王小林. RFA治疗局部晚期胰腺癌的研究进展[J]. 复旦学报(医学版), 2018, 45(1): 87-92. [DOI]
|
| [13] |
BARTOS A, BREAZU C, SPÂRCHEZ Z, et al. Radiofrequency ablation of locally advanced pancreatic tumors[J]. J Gastrointestin Liver Dis, 2018, 27(1): 102-104.
[URI]
|
| [14] |
ROMBOUTS SJ, VOGEL JA, VAN SANTVOORT HC, et al. Systematic review of innovative ablative therapies for the treatment of locally advanced pancreatic cancer[J]. Br J Surg, 2015, 102(3): 182-193.
[URI]
|
| [15] |
CASADEI R, RICCI C, PEZZILLI R, et al. A prospective study on radiofrequency ablation locally advanced pancreatic cancer[J]. Hepatobiliary Pancreat Dis Int, 2010, 9(3): 306-311.
[URI]
|
| [16] |
CRINÒ SF, D'ONOFRIO M, BERNARDONI L, et al. EUS-guided radiofrequency ablation (EUS-RFA) of solid pancreatic neoplasm using an 18-gauge needle electrode:feasibility, safety, and technical success[J]. J Gastrointestin Liver Dis, 2018, 27(1): 67-72.
[URI]
|
| [17] |
GAIDHANE M, SMITH I, ELLEN K, et al. Endoscopic ultrasound-guided radiofrequency ablation (EUS-RFA) of the pancreas in a porcine model[J]. Gastroenterol Res Pract, 2012, 2012: 431-451.
[URI]
|
| [18] |
SEICEAN A, TEFAS C, UNGUREANU B, et al. Endoscopic ultrasound guided radiofrequency ablation in pancreas[J]. Hepatogastroenterology, 2014, 61(134): 1717-1721.
[URI]
|
| [19] |
SILVIU UB, DANIEL P, CLAUDIU M, et al. Endoscopic ultrasound-guided radiofrequency ablation of the pancreas:an experimental study with pathological correlation[J]. Endosc Ultrasound, 2015, 4(4): 330-335.
[DOI]
|
| [20] |
SONG TJ, SEO DW, LAKHTAKIA S, et al. Initial experience of EUS-guided radiofrequency ablation of unresectable pancreatic cancer[J]. Gastrointest Endosc, 2016, 83(2): 440-453.
[DOI]
|
| [21] |
SCOPELLITI F, PEA A, CONIGLIARO R, et al. Technique, safety, and feasibility of EUS-guided radiofrequency ablation in unresectable pancreatic cancer[J]. Surg Endosc, 2018, 32(9): 4022-4028.
[DOI]
|
| [22] |
YOON WJ, DAGLILAR ES, KAMIONEK M, et al. Evaluation of radiofrequency ablation using the 1-Fr wire electrode in the porcine pancreas, liver, gallbladder, spleen, kidney, stomach, and lymph nodes:A pilot study[J]. Dig Endosc, 2015, 19(5): 355-366.
|
| [23] |
PAI M, HABIB N, SENTURK H, et al. Endoscopic ultrasound guided radiofrequency ablation, for pancreatic cystic neoplasms and neuroendocrine tumors[J]. World J Gastrointest Surg, 2015, 27, 7(4): 52-59.
[URI]
|
| [24] |
DEYUE Y, ZHEN G, JING Z, et al. 125I seed implantation for hepatocellular carcinoma with portal vein tumor thrombus:a systematic review and meta-analysis[J]. Brachytherapy, 2019, 18(4): 521-529.
[DOI]
|
 2020, Vol. 47
2020, Vol. 47


