2. 上海交通大学医学院瑞金医院皮肤科 上海 200025;
3. 无锡生物制剂(上海)有限公司 上海 201203
2. Department of Dermatology, Ruijin Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai 200025, China;
3. Wuxi Biologics(Shanghai) Co., Ltd., Shanghai 201203, China
皮肤T细胞淋巴瘤(cutaneous T-cell lymphoma, CTCL)是一种由T淋巴细胞克隆浸润引起,以皮肤病变为主要表现的非霍奇金淋巴瘤。临床上CTCL常见的亚型有蕈样肉芽肿、Sezdry综合征和原发性皮肤外周T细胞淋巴瘤[1]。CTCL的发病机制尚不明确,其临床表型与炎症性皮肤病相似,早期诊断困难[2]。目前CTCL的主要诊断方式是组织病理学活检、分子分析和外周血染色体检查,缺少非侵入性检查手段[3]。
代谢组学是“组学领域”的重要组成部分,通过对生物体内代谢物的定性和定量分析来寻找代谢物与生理病理变化的相关关系[4]。近年来,多组学技术已被用于CTCL的诊断,比如利用基因组学分析诊断CTCL[5-6]。LC/MS和GC/MS是代谢组学分析中应用最广泛的两种分析方法,两者识别出的差异代谢物既有区别又有联系。我们基于GC/MS和LC/MS对CTCL人血样进行研究,结果显示CTCL的发病机制与磷脂异常有关[7],进一步考虑进行皮肤组织样本的分析,但由于人病变组织不易获得,因此本研究采用CTCL荷瘤鼠的血浆和肿瘤组织分别进行GC/MS的代谢组学分析,并对LC/MS[8]和GC/MS找出的差异代谢物进行对比、分析和关联,从而为进一步研究CTCL提供依据。
材料和方法试剂和细胞 甲醇、乙腈(HPLC级)、肝素钠购自上海国药控股化学试剂有限公司;正庚烷购自阿拉丁生化科技有限公司(上海);十三酸、吡啶(HPLC级)、甲氧基胺盐酸盐、N-甲基-N-(三甲基硅烷基)三氟乙酰胺(MSTFA)、三甲基氯硅烷(TMCS)、RPMI 1640培养基(ATCC 302001)、HH细胞(T cell Lymphoma ATCC CRL-2105)、青霉素-链霉素溶液(SV30010,10 000 U/mL青霉素:10 000 U/mL链霉素)、胎牛血清(F2442-500 mL)和二甲亚砜(色谱纯)购自美国SigmaAldrich公司。
CTCL异种移植小鼠 HH细胞培养和CTCL异种移植小鼠模型与本课题组前期研究[8]相同。动物实验均符合复旦大学动物实验伦理标准要求。实验动物合格证号:伦药批第(2018-12-YF-YYQ-01)号。对照组:健康小鼠(n=5);实验组:CTCL荷瘤鼠(n=16)。取小鼠眼眶血,加肝素钠抗凝,1 600 ×g离心10 min,留取血浆。
HH细胞皮下注射于雌性裸鼠的右侧腋下,小鼠皮肤组织分别取自对照组小鼠的右腋下皮肤,实验组小鼠皮肤肿瘤组织近端(荷瘤鼠右腋下)、肿瘤组织远端(荷瘤鼠左腋下)和肿瘤组织。
血浆处理 取血浆50 μL至离心管,室温下解冻15 min,加200 μL的-20 ℃甲醇(含40 μg/mL十三烷酸),涡旋5 min,静置10 min,12 000×g下离心15 min,取上清液40 ℃下氮气吹干,残余物衍生化,12 000 ×g下离心5 min,上清液转至GC/MS进行分析。
组织处理 向EP管加入-20 ℃甲醇和小鼠组织(1 mg:50 μL),涡旋5 min,超声10 min,电子组织匀浆器(60 Hz,300 s)中匀浆,12 000×g下离心15 min,取600 μL上清,加入400 μL甲醇(40 μg/mL十三烷酸),涡旋5 min,静置10 min,12 000×g下离心15 min,取900 μL上清液40 ℃下氮气吹干,衍生化3 h,12 000× g下离心5 min,上清液转至GC/MS进行分析。
GC/MS分析 Agilent 7890B气相色谱仪(含7693自动进样器)和5977A四极质量分析仪。色谱柱:DB-5 MS熔融石英毛细管柱(30 m×250 μm×0.25 μm,美国Agilent公司)。程序升温:初始70 ℃保持3 min,之后5 ℃/min升至300 ℃保持5 min。氦气载气流量:1.2 mL/min;进样体积:1 μL;分流比:2:1;入口温度:300 ℃;界面温度:280 ℃;离子源温度:230 ℃。
数据采集及处理 通过MassHunter定性分析软件(B.06.00)将原始数据转换为mzData格式,转换后的数据上传到XCMS-Online(https://xcmsonline.scripps.edu)进行数据过滤,峰对齐和保留时间校正。峰强度校正后,使用“80%规则”筛选数据。数据预处理后用SIMCA 13(Umetrics,Sweden)进行统计分析。
将筛选出的代谢物上传至NIST数据库和人类代谢组数据库(HMDB,http://www.hmdb.ca)进行比对,部分采用标准品进行结构验证。使用热图(GraphPad)分析以观察不同组之间差异代谢物的变化。通过Medcalc软件构建接受者操作特征曲线(ROC)以评估差异代谢物的灵敏度和特异性。采用MetaboAnalyst 3.0 (http://www.metaboanalyst.ca/)进行差异代谢物的代谢通路分析。
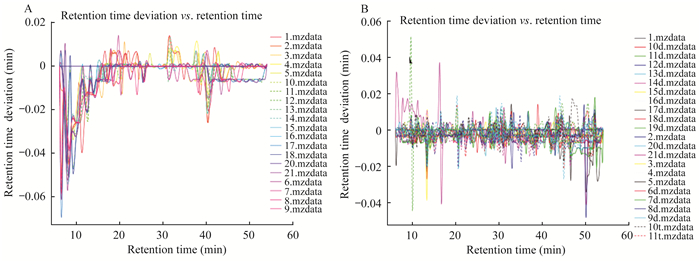
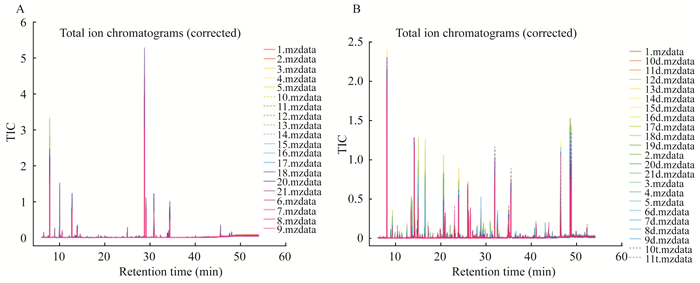
结果原始数据的质量控制和TIC图 所有血浆样品和组织样品的保留时间偏差分别为4 s和1.2 s,说明所采用的分析方法(包括色谱条件)具有良好的重现性(图 1、2)。原始数据包括含有1 704个特征离子的21个血浆样本,含有1 016个特征离子的50个组织样本,数据过滤后,得到1 047个和653个特征离子。
|
| A:Plasma samples; B:Tissue samples. 图 1 对照组与CTCL组小鼠血浆样品和组织样品的保留时间偏差 Fig 1 Retention time deviation plots of plasma and tissue samples of mice in control group and CTCL group |
|
| A:Plasma samples; B:Tissue samples. 图 2 CTCL组小鼠血浆和组织样品的重叠TIC Fig 2 The overlapping TICs of plasma and tissue samples in mice of CTCL group |
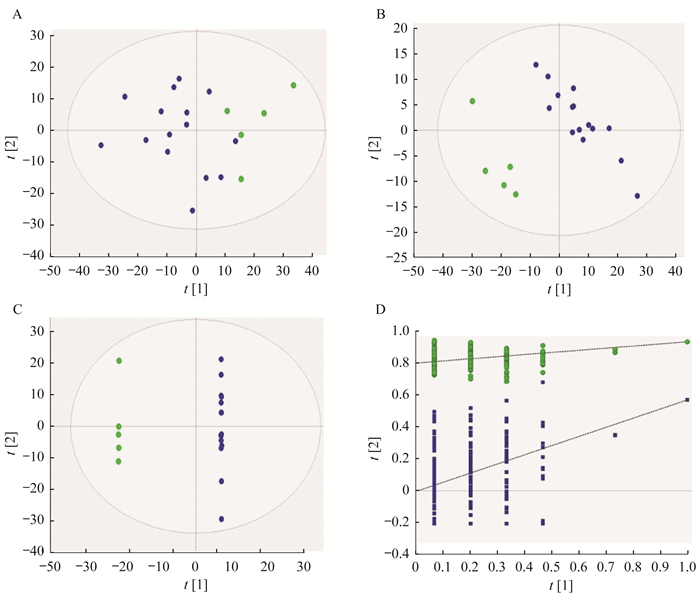
数据的多变量统计分析 在血浆样品的CTCL组和对照组间,PCA分离不明显,其R2X=0.474,Q2=0.204(图 3A);PLS-DA分离明显,其R2X=0.314, R2Y=0.944, Q2=0.576(图 3B);OPLS-DA分离明显,其R2X=0.630, R2Y=1.000, Q2=0.672(图 3C)。200次交叉排列验证结果表明,PLS-DA分析模型具有可靠的预测能力(R2:0, 0.809;Q2:0,-0.003 73) (图 3D)。
|
| Control group was labeled by green circle, CTCL group was labeled by blue circle.A:Scores plot of PCA; B:Scores plot of PLS-DA; C:Scores plot of OPLS-DA; D:Permutation validation plots of PLS-DA:R2 labeled by green triangles and Q2 labeled by blue boxes. 图 3 对照组与CTCL组小鼠血浆PCA、PLS-DA及OPLS-DA分析 Fig 3 PCA, PLS-DA and OPLS-DA analysis of plasma of mice in control group and CTCL group |
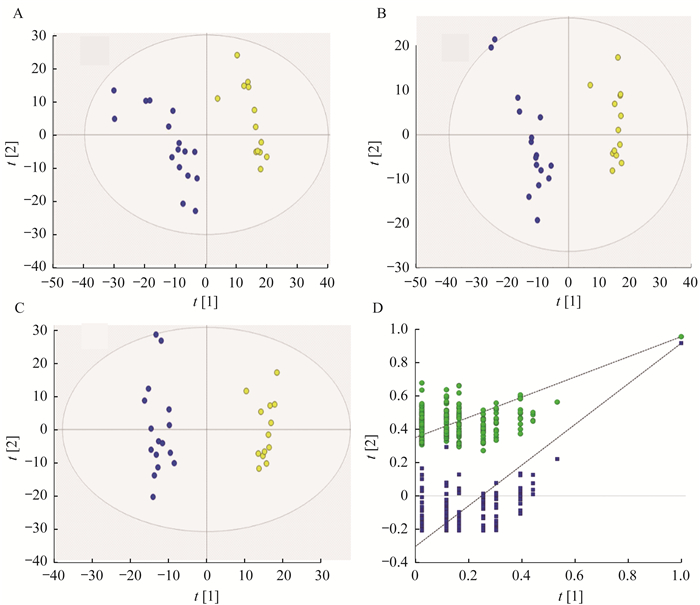
在组织样品中,PCA显示肿瘤组和其他3组间有显著差异(R2X=0.868, Q2=0.721),但是对照组与肿瘤组织远端(荷瘤鼠左腋下)间差异不大。为消除小鼠的个体差异,进一步观察CTCL小鼠自身的肿瘤组织和肿瘤组织远端(荷瘤鼠左腋下)。PCA(R2X=0.748,Q2=0.618)、PLS-DA(R2X=0.531,R2Y=0.955,Q2=0.916)和OPLS-DA(R2X=0.613,R2Y=0.977,Q2=0.939)的得分图和拟合参数均显示出肿瘤组织远端组和肿瘤组有明显分离(图 4)。对PLS-DA进行200次交叉检验验证,R2(0,0.350)和Q2(0,-0.304)表明该模型无过度拟合(图 4)。
|
| Tumor tissue group was labeled by yellow circle, and tumor tissue distal end group was labeled by blue circles.A:Scores plot of PCA; B:Scores plots of PLS-DA; C:Scores plot of OPLS-DA; D:Permutation validation plots of PLS-DA:R2 labeled by green triangles and Q2 labeled by blue boxes. 图 4 CTCL小鼠自身的肿瘤组织和肿瘤组织远端的PCA、PLS-DA及OPLS-DA分析 Fig 4 PCA, PLS-DA and OPLS-DA analysis of tumor tissue and tumor tissue distal end in CTCL mice |
血浆和组织样品中差异代谢物的筛选与结构验证 根据OPLS-DA模型中的VIP值,t检验的P值和FC值(VIP≥1,P≤0.05和FC≥1.5)选择差异代谢物(表 1、2)。在血浆样品中共有29种差异代谢物(其中两种在HMDB中不存在),21种经标准品得以验证,在组织样品中共有33种差异代谢物(其中一种在HMDB中不存在),25种经标准品得以验证。其中13种差异代谢物(L-缬氨酸,L-丝氨酸,L-亮氨酸,甘油,L-异亮氨酸,L-苯丙氨酸,3-磷酸甘油,D-葡萄糖,阿拉伯糖,肌醇,亚油酸,硬脂酸,胆固醇)同时存在于血浆和组织样品中(图 5)。
| NO. | HMDB ID | RT | Metabolitesa | Matching score | Validated by standard samples |
| 1 | HMDB00190 | 7.79 | L-Lactic acid | 960 | Y |
| 2 | HMDB00161 | 8.91 | L-alanine | 812 | Y |
| 3 | HMDB00357 | 10.54 | 3-Hydroxybutyric acid | 943 | N |
| 4 | HMDB00162 | 10.73 | L-Proline1 | 719 | Y |
| 5 | HMDB00883 | 12.10 | L-Valine | 919 | Y |
| 6 | HMDB00294 | 12.68 | Urea | 930 | Y |
| 7 | HMDB00187 | 13.13 | L-Serine | 879 | Y |
| 8 | HMDB00687 | 13.67 | L-Leucine | 930 | Y |
| 9 | HMDB02142 | 13.72 | Phosphoric acid | 910 | Y |
| 10 | HMDB00131 | 13.88 | Glycerol | 936 | Y |
| 11 | HMDB00162 | 14.24 | L-Proline2 | 698 | Y |
| 12 | HMDB00172 | 14.25 | L-Isoleucine | 894 | Y |
| 13 | HMDB00167 | 16.87 | L-Threonine | 919 | Y |
| 14 | HMDB00159 | 22.44 | L-Phenylalanine | 860 | Y |
| 15 | HMDB00666 | 23.99 | Heptanoic acid | 443 | N |
| 16 | HMDB00126 | 25.82 | Glycerol 3-phosphate | 666 | N |
| 17 | HMDB29942 | 26.19 | Arabinose | 766 | N |
| 18 | HMDB00806 | 27.01 | Myristic acid | 817 | Y |
| 19 | HMDB00122 | 28.76 | D-Glucose | 925 | Y |
| 20 | HMDB00625 | 30.30 | Gluconic acid | 622 | N |
| 21 | HMDB00220 | 30.83 | Palmitic acid | 952 | Y |
| 22 | HMDB29942 | 31.29 | Arabinose | 676 | N |
| 23 | HMDB00211 | 32.27 | Myo-inositol | 881 | Y |
| 24 | HMDB00673 | 33.78 | Linoleic acid | 745 | N |
| 25 | HMDB00207 | 33.87 | Oleic acid | 800 | N |
| 26 | HMDB00929 | 34.19 | L-Tryptophan | 869 | Y |
| 27 | HMDB00827 | 34.34 | Stearic acid | 947 | Y |
| 28 | HMDB31074 | 40.06 | Glycerol 1-hexadecanoate | 874 | Y |
| 29 | HMDB00067 | 47.56 | Cholesterol | 727 | Y |
| aThere was more than 1 derivatized product from amino acids and saccharides.The products derived from the same metabolites were distinguished with number. Y:Validated by standard; N:standard samples were not available. | |||||
| NO. | HMDB ID | RT | Metabolitesa | Matching score | Validated by standard samples |
| 1 | HMDB00123 | 6.55 | Glycine | 607 | Y |
| 2 | HMDB00883 | 8.87 | L-Valine1 | 946 | Y |
| 3 | HMDB00161 | 9.26 | L-Alanine | 920 | Y |
| 4 | HMDB00687 | 10.82 | L-Leucine | 872 | Y |
| 5 | HMDB00883 | 12.48 | L-Valine2 | 909 | Y |
| 6 | HMDB00187 | 13.67 | L-Serine | 925 | Y |
| 7 | HMDB00687 | 14.08 | L-Leucine | 731 | Y |
| 8 | HMDB00131 | 14.17 | Glycerol | 930 | Y |
| 9 | HMDB00172 | 14.66 | L-Isoleucine | 760 | Y |
| 10 | HMDB00123 | 14.97 | Glycine | 934 | Y |
| 11 | HMDB00187 | 16.50 | L-Serine | 946 | Y |
| 12 | HMDB00696 | 20.51 | L-Methionine | 765 | Y |
| 13 | HMDB00267 | 20.53 | Pyroglutamic acid | 930 | Y |
| 14 | HMDB00191 | 20.59 | L-Aspartic acid | 936 | Y |
| 15 | HMDB00168 | 22.41 | L-Asparagine | 804 | Y |
| 16 | HMDB00148 | 22.96 | L-Glutamic acid | 899 | Y |
| 17 | HMDB00159 | 23.00 | L-Phenylalanine | 898 | Y |
| 18 | HMDB00126 | 26.08 | Glycerol 3-phosphate | 927 | N |
| 19 | HMDB00224 | 26.46 | O-Phosphoethanolamine | 890 | Y |
| 20 | HMDB00094 | 27.28 | Citric acid | 735 | Y |
| 21 | HMDB00446 | 27.94 | N-Alpha-acetyllysine | 740 | N |
| 22 | HMDB00122 | 28.80 | D-Glucose | 843 | Y |
| 23 | HMDB00182 | 29.36 | L-Lysine | 840 | Y |
| 24 | HMDB00158 | 29.68 | L-Tyrosine | 908 | Y |
| 25 | HMDB00289 | 30.84 | Uric acid | 912 | N |
| 26 | HMDB00292 | 31.23 | Xanthine | 680 | N |
| 27 | HMDB00211 | 32.47 | Myo-inositol | 927 | Y |
| 28 | HMDB00673 | 34.82 | Linoleic acid | 904 | N |
| 29 | HMDB00827 | 35.42 | Stearic acid | 936 | Y |
| 30 | HMDB01043 | 37.45 | Arachidonic acid | 843 | N |
| 31 | HMDB00944 | 41.76 | Behenic acid | 879 | N |
| 32 | HMDB02003 | 44.60 | Tetracosanoic acid | 622 | N |
| 33 | HMDB00067 | 48.50 | Cholesterol | 931 | Y |
| aThere was more than 1 derivatized product from amino acids and saccharides.The products derived from the same metabolites were distinguished with number.Y:Validated by standard; N:Standard samples were not available. | |||||

|
| 图 5 CTCL小鼠血浆样品和肿瘤组织样品中检测到的差异代谢物数据的Venn图 Fig 5 Venn diagrams presenting data for differential metabolites detected in plasma samples and tumor tissue samples of CTCL mice detected by GC/MS and LC/MS respectively |
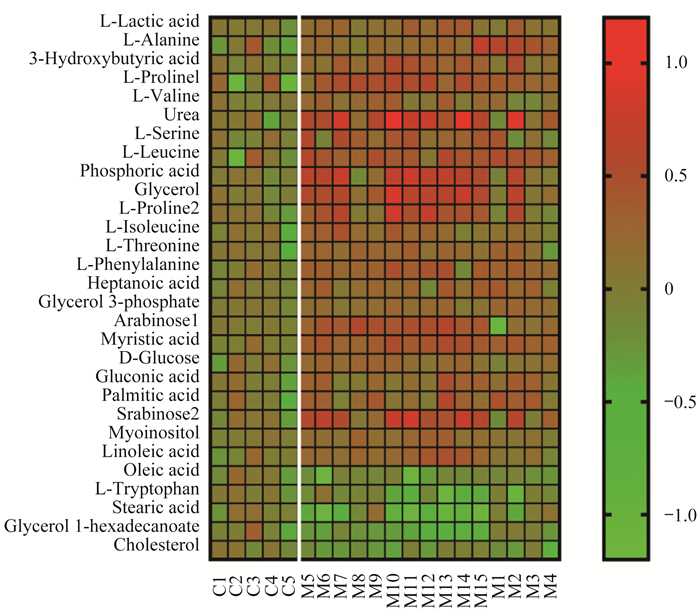
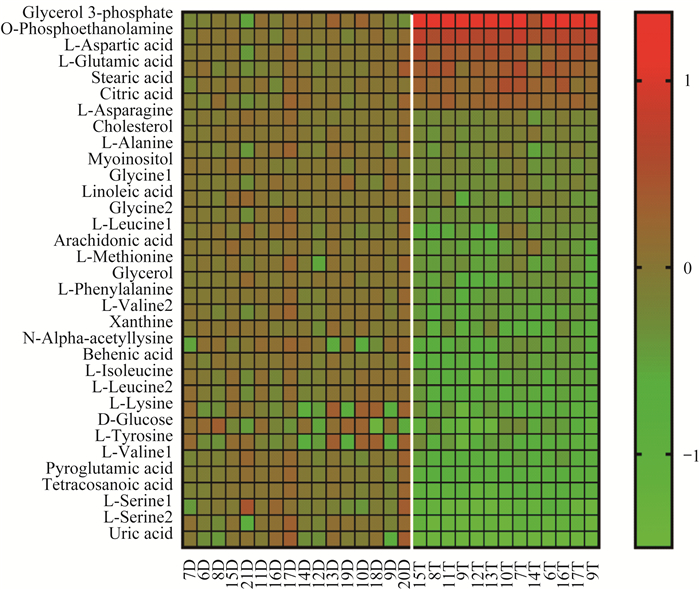
在血浆样品中CTCL组的乳酸和大部分氨基酸增加;油酸、硬脂酸、1-十六烷酸酯甘油和胆固醇明显减少(图 6)。在组织样品中肿瘤组的3-磷酸甘油,邻磷酸乙醇胺和L-天冬氨酸显著增加;大部分氨基酸含量减少(图 7)。
|
| C:Control group; M:CTCL group. 图 6 对照组与CTCL组小鼠血浆样品中代谢物的热图分析 Fig 6 Heatmap of metabolites in plasma samples of mice in control group and CTCL group |
|
| D:Tumor tissue distal end group; T:Tumor group. 图 7 CTCL小鼠自身的肿瘤组织和肿瘤组织远端组中代谢物的热图 Fig 7 Heatmap of metabolites in tumor tissue group and tumor tissue distal end group in CTCL mice |
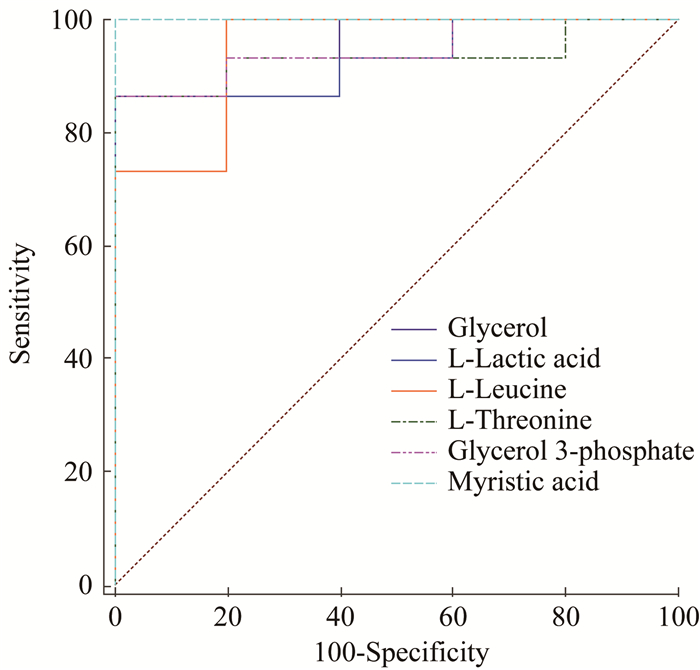
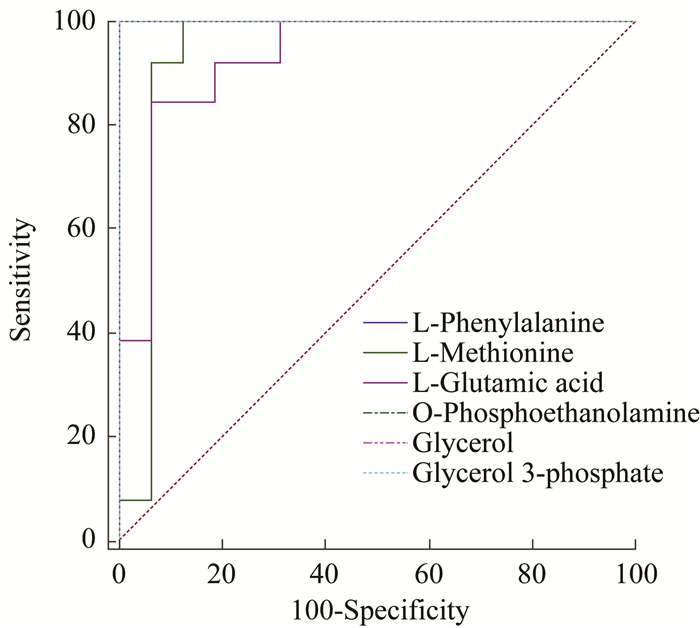
血浆和组织样品中差异代谢物的ROC分析 对62种差异代谢物作ROC分析(表 3、4):在血浆样品中,肉豆蔻酸的AUC值为1,表明肉豆蔻酸具有最高的特异性和灵敏度(图 8);在组织样品中,多种差异代谢物的AUC值为1(图 9)。
| Metabolites | RT | AUC | Standard error | 95% CI | Sensitivity | Specificity |
| L-Lactic acida | 7.79 | 0.907 | 0.069 | 0.692-0.990 | 73.33 | 100.00 |
| L-Alanine | 8.91 | 0.840 | 0.147 | 0.609-0.963 | 93.33 | 80.00 |
| 3-Hydroxybutyrica acid | 10.54 | 0.947 | 0.049 | 0.746-0.998 | 86.67 | 100.00 |
| L-Valine | 12.10 | 0.800 | 0.103 | 0.563-0.943 | 80.00 | 80.00 |
| Ureaa | 12.68 | 0.920 | 0.064 | 0.709-0.993 | 80.00 | 100.00 |
| L-Serine | 13.13 | 0.800 | 0.100 | 0.563-0.943 | 66.67 | 100.00 |
| L-Leucinea | 13.67 | 0.947 | 0.058 | 0.746-0.998 | 100.00 | 80.00 |
| Phosphoric acid | 13.72 | 0.853 | 0.088 | 0.625-0.969 | 80.00 | 100.00 |
| Glycerola | 13.88 | 0.947 | 0.049 | 0.746-0.998 | 86.67 | 100.00 |
| L-Proline | 14.24 | 0.800 | 0.105 | 0.563-0.943 | 66.67 | 100.00 |
| L-Isoleucine | 14.25 | 0.827 | 0.103 | 0.594-0.957 | 60.00 | 100.00 |
| L-Threoninea | 16.87 | 0.933 | 0.058 | 0.728-0.996 | 86.67 | 100.00 |
| L-Phenylalaninea | 22.44 | 0.933 | 0.067 | 0.728-0.996 | 93.33 | 100.00 |
| Heptanoic acid | 23.99 | 0.867 | 0.083 | 0.641-0.975 | 73.33 | 100.00 |
| Glycerol 3-phosphatea | 25.82 | 0.947 | 0.048 | 0.746-0.998 | 86.67 | 100.00 |
| Myristic acida | 27.01 | 1.000 | 0.000 | 0.832-1.000 | 100.00 | 100.00 |
| D-Glucose | 28.76 | 0.787 | 0.138 | 0.549-0.935 | 93.33 | 60.00 |
| Gluconic acid | 30.30 | 0.867 | 0.101 | 0.641-0.975 | 80.00 | 80.00 |
| Palmitic acid | 30.83 | 0.867 | 0.100 | 0.641-0.975 | 93.33 | 80.00 |
| Myo-inositola | 32.27 | 0.960 | 0.039 | 0.766-1.000 | 86.67 | 100.00 |
| Linoleic acid | 33.78 | 0.853 | 0.122 | 0.625-0.969 | 93.33 | 80.00 |
| Oleic acid | 33.87 | 0.720 | 0.183 | 0.478-0.894 | 100.00 | 60.00 |
| L-Tryptophan | 34.19 | 0.827 | 0.125 | 0.594-0.957 | 93.33 | 80.00 |
| Stearic acid | 34.34 | 0.813 | 0.098 | 0.578-0.950 | 66.67 | 100.00 |
| Cholesterol | 47.56 | 0.867 | 0.096 | 0.641-0.975 | 66.67 | 100.00 |
| aAUC>0.9. | ||||||
| Metabolites | RT | AUC | Standard error | 95% CI | Sensitivity | Specificity |
| L-Valine1a | 8.87 | 1.000 | 0.000 | 0.881-1.000 | 100.00 | 100.00 |
| L-Alanine | 9.26 | 0.861 | 0.072 | 0.682-0.960 | 92.31 | 75.00 |
| L-Leucine1a | 10.82 | 0.913 | 0.062 | 0.749-0.985 | 84.62 | 100.00 |
| L-Serine a | 13.67 | 1.000 | 0.000 | 0.881-1.000 | 100.00 | 100.00 |
| Glycerola | 14.17 | 1.000 | 0.000 | 0.881-1.000 | 100.00 | 100.00 |
| L-Isoleucinea | 14.66 | 1.000 | 0.000 | 0.881-1.000 | 100.00 | 100.00 |
| Glycine | 14.97 | 0.938 | 0.043 | 0.781-0.993 | 92.31 | 87.5 |
| L-Methioninea | 20.51 | 0.938 | 0.058 | 0.781-0.993 | 100.00 | 87.50 |
| Pyroglutamic acida | 20.53 | 1.000 | 0.000 | 0.881-1.000 | 100.00 | 100.00 |
| L-Aspartic acida | 20.59 | 0.928 | 0.063 | 0.768-0.991 | 84.62 | 100.00 |
| L-Asparaginea | 22.41 | 0.928 | 0.049 | 0.768-0.991 | 76.92 | 100.00 |
| L-Glutamic acida | 22.96 | 0.933 | 0.046 | 0.775-0.992 | 84.62 | 93.75 |
| L-Phenylalaninea | 23.00 | 1.000 | 0.000 | 0.881-1.000 | 100.00 | 100.00 |
| Glycerol 3-phosphatea | 26.08 | 1.000 | 0.000 | 0.881-1.000 | 100.00 | 100.00 |
| O-Phosphoethanolaminea | 26.46 | 1.000 | 0.000 | 0.881-1.000 | 100.00 | 100.00 |
| Citric acida | 27.28 | 0.918 | 0.056 | 0.755-0.987 | 100.00 | 81.25 |
| N-Alpha-acetyllysine | 27.94 | 0.870 | 0.072 | 0.693-0.965 | 100.00 | 75.00 |
| D-Glucose | 28.80 | 0.851 | 0.076 | 0.670-0.955 | 100.00 | 68.75 |
| L-Lysine | 29.36 | 0.856 | 0.072 | 0.676-0.958 | 92.31 | 75.00 |
| L-Tyrosine | 29.68 | 0.856 | 0.072 | 0.676-0.958 | 92.31 | 75.00 |
| Uric acida | 30.84 | 1.000 | 0.000 | 0.881-1.000 | 100.00 | 100.00 |
| Xanthinea | 31.23 | 0.981 | 0.021 | 0.847-1.000 | 100.00 | 93.75 |
| Myo-inositola | 32.47 | 0.976 | 0.023 | 0.839-1.000 | 92.31 | 93.75 |
| Linoleic acida | 34.82 | 0.962 | 0.031 | 0.816-0.999 | 84.62 | 100.00 |
| Stearic acida | 35.42 | 0.966 | 0.028 | 0.824-0.999 | 92.31 | 93.75 |
| Arachidonic acida | 37.45 | 0.933 | 0.068 | 0.775-0.992 | 92.31 | 100.00 |
| Behenic acida | 41.76 | 1.000 | 0.000 | 0.881-1.000 | 100.00 | 100.00 |
| Tetracosanoic acida | 44.60 | 1.000 | 0.000 | 0.881-1.000 | 100.00 | 100.00 |
| Cholesterola | 48.50 | 0.938 | 0.054 | 0.781-0.993 | 92.31 | 93.75 |
| aAUC>0.9. | ||||||
|
| 图 8 对照组与CTCL组小鼠血浆样品中部分AUC>0.9的差异代谢物的ROC Fig 8 The ROC of differential metabolites with AUC > 0.9 in plasma samples of mice in control group and CTCL group |
|
| 图 9 CTCL小鼠自身的肿瘤组织和肿瘤组织远端组样品中部分AUC>0.9的差异代谢物的ROC Fig 9 The ROC of differential metabolites with AUC > 0.9 in CTCL mice of tumor tissue and tumor tissue distal end |
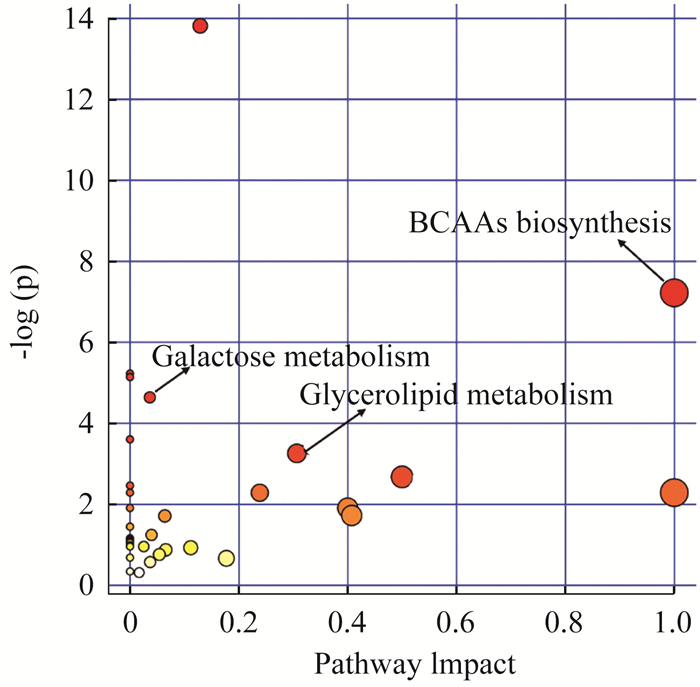
血浆和组织样品中差异代谢物的Pathway分析 支链氨基酸(BCAA)和甘油磷脂代谢被认为是血浆和组织样品中最相关的代谢途径(图 10)。
|
| 图 10 BCAAs与甘油磷脂代谢是最相关的代谢途径 Fig 10 BCAAs and glycerolipid metabolism were the most relevant metabolic pathways |
多平台多样本数据相互补充和印证 GC/MS和LC/MS是目前代谢组学分析的主流技术平台。本研究采用GC/MS分析平台,对CTCL荷瘤鼠的血浆和肿瘤组织样品进行代谢组学分析,并将研究结果和课题组之前的研究结果[8]进行比较(图 5),结果表明:血浆中的胆固醇同时在LC/MS和GC/MS中出现,肿瘤组织中黄嘌呤和7种氨基酸(L-天冬酰胺、L-赖氨酸、L-苯丙氨酸、L-酪氨酸、L-色氨酸、L-亮氨酸、L-谷氨酸)同时在LC/MS和GC/MS分析平台中出现。LC/MS和GC/MS两种分析平台对该类差异代谢物的分析均能达到较高的灵敏度,表明这两种分析方法不仅可以互补,还可以进行有效的相互验证。此外,由于代谢组学涉及的化合物性质差异较大,不同的分析平台检出的种类不同,如GC/MS可检出乳酸和葡萄糖,LC/MS可检出PC类和MG类,采用多平台技术可以增加差异代谢物的覆盖面,因此在以后的研究中可以考虑将两种分析方法结合以获得更多的信息。
由于代谢途径不同,在血浆和肿瘤组织中会出现不同的差异代谢物,我们又将血浆和肿瘤组织样品的分析结果进行了比较,共同差异代谢物有13种,分别占血浆差异代谢物和肿瘤组织差异代谢物的48.3%和42.3%;涉及的共同通路有BCAA代谢、半乳糖代谢、亚油酸通路和甘油磷脂代谢。说明血浆和肿瘤组织样品不仅可以单独反映CTCL的局部代谢,两种样品之间的差异代谢物还较为相关。不同样本得到的差异代谢物在种类和数量上不完全相同,如在GC/MS平台中单甘脂和棕榈酸等15种差异代谢物只在血浆样本中出现,尿酸和磷酸乙醇胺等19种差异代谢物只在肿瘤组织样本中出现。
CTCL小鼠血浆和肿瘤组织中BCAA异常 BCAA(包括缬氨酸,亮氨酸和异亮氨酸)在血浆中上调并在肿瘤组织中下调,这表明BCAA在CTCL的恶性T细胞增殖过程中被转运和消耗,以适应更高的蛋白质合成速率。异常BCAA是CTCL小鼠血浆和肿瘤组织最重要的代谢特征。
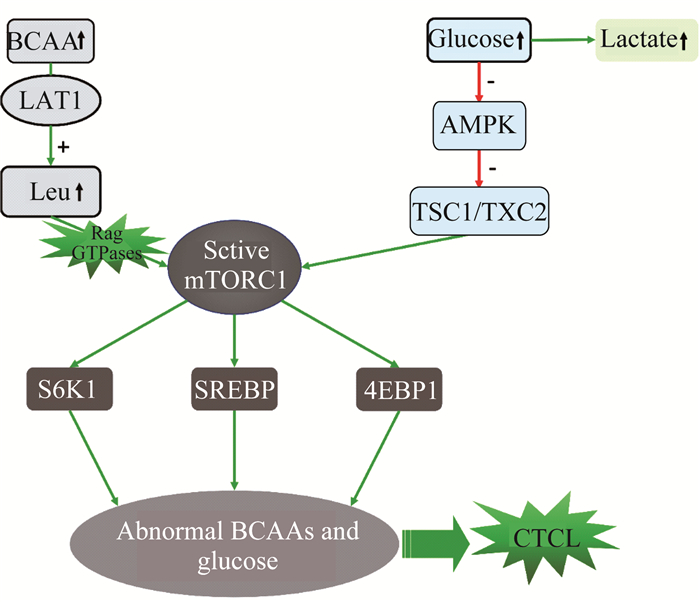
BCAA(尤其是亮氨酸)可通过激活mTORC1[9]刺激骨骼肌蛋白形成。同时亮氨酸会降低AMP激酶(AMPK)活性, 从而引发胰岛素抵抗和癌症[10]。在本研究中,亮氨酸在血浆中明显升高,在组织中明显减少,表明BCAA可能激活mTORC1并引发CTCL(图 11)。
|
| 图 11 基于CTCL小鼠血浆和肿瘤组织中BCAA异常假设的CTCL小鼠代谢通路 Fig 11 Hypothetical metabolic pathways based on BCAA abnormalities in plasma and tumor tissues of CTCL mice |
CTCL小鼠血浆和肿瘤组织中3-磷酸甘油上调 血浆和肿瘤组织中增加的3-磷酸甘油可能导致细胞膜降解或恶性T细胞形态变化。小鼠血浆中3-磷酸甘油的升高与课题组之前的研究结果[8]相一致。
CTCL小鼠血浆中葡萄糖上调 体内葡萄糖含量也会影响mTORC1[10]的活性。血浆中葡萄糖的升高会抑制AMPK通路,从而减轻TCS1/TSC2复合物的抑制功能并激活mTORC1[11]。同样,葡萄糖也可以通过降低亮氨酸等AMPK激酶的活性来激活mTORC1[12]。在本研究中,血浆中葡萄糖含量增加,意味着葡萄糖激活mTORC1, 同时参与癌细胞的Warburg效应,而血浆中乳酸含量增加也验证了这一点[13];肿瘤组织中葡萄糖含量降低,可认为葡萄糖通过糖酵解被消耗并转化为乳酸(图 11)。
CTCL小鼠血浆o-磷酸乙醇胺被降解 我们之前的研究已显示:组织中鞘氨醇-1-磷酸酯(S1P)增加会促使磷酸戊糖途径与核苷酸代谢相互作用,且与CTCL肿瘤组织大小显著相关(Pearson相关系数ρ=0.64)[8]。本研究显示, 血浆中S1P降解的o-磷酸乙醇胺含量也增加,不同样本(血浆和皮肤)的研究结果表明S1P和o-磷酸乙醇胺的异常可能与CTCL关系密切。
| [1] |
VAKEVA L, NⅡTTYVUOPIO R, LEPPA S, et al. Allogeneic haematopoietic stem cell transplantation for patients with cutaneous T-cell lymphoma[J]. Acta Derm-Venereol, 2016, 96(6): 816-817.
[URI]
|
| [2] |
SIDIROPOULOS KG, MARTINEZ-ESCALA ME, YELAMOS O, et al. Primary cutaneous T-cell lymphomas:a review[J]. J Clin Pathol, 2015, 68(12): 1003-1005.
[DOI]
|
| [3] |
BENJAMIN CHASE A, MARKEL K, TAWA MC. Optimizing care and compliance for the treatment of mycosis fungoides cutaneous T-cell lymphoma with mechlorethamine gel[J]. Clin J Oncol Nurs, 2015, 19(6): 131-139.
[DOI]
|
| [4] |
NICHOLSON JK, TIMBRELL JA, SADLER PJ. Proton NMR spectra of urine as indicators of renal damage.Mercury-induced nephrotoxicity in rats[J]. Mol Pharmacol, 1985, 27(6): 644-651.
[URI]
|
| [5] |
LITVINOV IV, NETCHIPOROUK E, CORDEIRO B, et al. The use of transcriptional profiling to improve personalized diagnosis and management of cutaneous T-cell lymphoma (CTCL)[J]. Clin Cancer Res, 2015, 21(12): 2820-2829.
[DOI]
|
| [6] |
ION A, POPA IM, PAPAGHEORGHE LM, et al. Proteomic approaches to biomarker discovery in cutaneous T-cell lymphoma[J]. Dis Markers, 2016, 2016: 9602472.
|
| [7] |
JIANG G, SHEN X, KANG H, et al. Serum metabolite profiling of cutaneous T-cell lymphoma based on a multiplatform approach[J]. J Chromatography B Anal Technol Biomed Life Sci, 2018, 1077-1078: 71-76.
[DOI]
|
| [8] |
LE Y, SHEN X, KANG H, et al. Accelerated, untargeted metabolomics analysis of cutaneous T-cell lymphoma reveals metabolic shifts in plasma and tumor adjacent skins of xenograft mice[J]. J Mass Spectrom, 2018, 53(2): 172-182.
[DOI]
|
| [9] |
KIMBALL SR, JEFFERSON LS. Signaling pathways and molecular mechanisms through which branched-chain amino acids mediate translational control of protein synthesis[J]. J Nutr, 2006, 136(1 Suppl): 227S-231S.
[URI]
|
| [10] |
LONG W, SAFFER L, WEI L, et al. Amino acids regulate skeletal muscle PHAS-I and p70 S6-kinase phosphorylation independently of insulin[J]. Am J Physiol Endocrinol Metab, 2000, 279(2): 301-306.
[DOI]
|
| [11] |
INOKI K, ZHU T, GUAN KL. TSC2 mediates cellular energy response to control cell growth and survival[J]. Cell, 2003, 115(5): 577-590.
[DOI]
|
| [12] |
GWINN DM, SHACKELFOR DB, EGAN DF, et al. AMPK phosphorylation of raptor mediates a metabolic checkpoint[J]. Mol Cell, 2008, 30(2): 214-226.
[DOI]
|
| [13] |
WARBURG O. On respiratory impairment in cancer cells[J]. Science, 1956, 124(3215): 269-270.
[URI]
|
 2020, Vol. 47
2020, Vol. 47


