2. 广东省人民医院麻醉科-广东医学科学院 广州 510080
2. Department of Anesthesiology, Guangdong General Hospital-Guangdong Academy of Medical Sciences, Guangzhou 510080, Guangdong Province, China
皮肤黑色素瘤(skin cutaneous melanoma,SKCM)是来源于皮肤黑色素细胞的恶性肿瘤,在皮肤肿瘤中恶性程度最高[1]。BRAF基因编码RAF家族丝氨酸/苏氨酸蛋白激酶,其在SKCM患者中突变率约为50%突变[2-3]。该基因的突变将活化MEK蛋白,导致MEK/ERK通路被激活,促进细胞增殖、存活与转移[4]。因此,BRAF是SKCM的重要驱动基因,其突变往往预示着临床预后较差。目前,抗程序性死亡蛋白-1(programmed death-1,PD-1)单抗单一疗法或与ipilimumab联合疗法已然成为不可手术的BRAF野生型SKCM患者的一线治疗方案;而BRAF抑制剂+MEK抑制剂双靶治疗是不可手术的BRAF突变SKCM患者的一线治疗方案。但是仅根据BRAF突变情况确定治疗方案可能不利于提高治疗反应率。因此,本研究将寻找一个SKCM治疗的标志物,联合BRAF突变分析后可为SKCM患者选择更合适的治疗方案,提高免疫治疗反应率。
研究发现,肿瘤细胞转移是不同器官通过趋化作用吸引特定类型肿瘤细胞归巢的结果。趋化因子受体4(chemokine receptor,CXCR4)在众多肿瘤组织中表达升高,当CXCR4大量存在于肿瘤细胞上时,会促进癌细胞迁移,为肿瘤转移奠定了基础。此外,CXCR4也参与炎症,具有趋化免疫细胞的生物学作用[5]。CXCR4在肿瘤生长、侵袭、血管生成、转移和治疗抵抗中起关键作用,CXCR4高表达往往预示肿瘤预后不良[6]。本研究在SKCM中探索CXCR4表达对肿瘤微卫星不稳定性(microsatellite instability,MSI)和免疫检查点分子表达的影响,并根据CXCR4表达联合BRAF突变情况评估患者免疫检查点阻断(immune checkpoint blockade,ICB)治疗的反应性。
资料和方法数据资料 SKCM患者(n=470)数据来源于癌症基因组图谱(The Cancer Genome Atlas,TCGA)数据库。本研究纳入470名于1978年至2013年确诊为SKCM的患者,均获得知情同意。研究对象以白种人为主,男性290人,女性180人,其中BRAF野生(BRAFWT)患者230例(原发性SKCM患者204例,转移性SKCM 26例),突变(BRAFMUT)患者235例(原发性SKCM患者209例,转移性SKCM 26例)。另外5名患者无BRAF是否突变的数据,将其剔除。
基因突变分析 分析上述SKCM患者基因突变数据包,利用Oncoplot展示患者中体细胞突变情况,并按照基因突变频率排序选出突变频率前十的突变基因,水平直方图显示各基因的突变频率。
筛选差异基因和GO富集分析 用R软件的Limma软件包分析BRAFWT和BRAFMUT组mRNA的差异表达,筛选出Adjusted P < 0.05且log2(倍数变化) > 1或log2(倍数变化) < -1的差异基因。通过metascape平台将筛选出的差异性表达基因进行富集分析。
基因表达分析 利用GEPIA2分析TCGA、GTEx数据库中SKCM患者癌组织和配对的正常组织中CXCR4表达情况,以log2(TPM+1)的数值为横坐标。通过Wilcox检验2组样本显著性,Kruskal-Wallis检验3组样本显著性。
MSI与基因表达的相关性分析 使用TCGA数据库中SKCM患者mRNA数据,通过Spearman的相关分析描述MSI得分与基因表达之间的相关性。Pspearman表示两者之间的相关性,取值范围在[-1, 1],Pspearman为负数代表MSI得分与基因表达呈现负相关性,正值代表呈现正相关性,绝对值越接近1,二者相关性越强;越接近0,则二者相关性越弱。P < 0.05为差异有统计学意义。
免疫检查点分析 CD274、CTLA4、HAVCR2、LAG3、PDCD1、PDCD1LG2、TIGIT和SIGLEC15基因是免疫检查点相关的转录本,通过R软件包ggplot2和pheatmap评估免疫检查点相关基因的表达情况。通过Kruskal-Wallis检验分析4组之间的显著性,P < 0.05为差异有统计学意义。
结果筛选BRAFWT组和BRAFMUT组的差异性表达基因及相应通路 SKCM患者中高频突变基因的全景瀑布图(图 1A),其中BRAF突变率高达50%,SNV类别的变体以C > T为主(图 1B)。SKCM患者根据BRAF突变与否分为BRAFWT组(n=230)和BRAFMUT组(n=235),并分析两组的差异性表达基因。BRAFMUT组与BRAFWT组相比,15个基因表达显著下调,2个基因表达显著上调(图 1C,差异基因见表 1)。通过metascape平台对上述17个差异基因进行富集分析,结果表明这些差异性表达基因主要涉及p53下游通路和IL-18途径(图 1D)。综上,BRAF是SKCM的高频突变基因,且突变后可能会影响p53及IL-18通路,这两个通路在免疫微环境[7]、细胞生长[8]、转移[9-10]等多个方面都有重要影响。
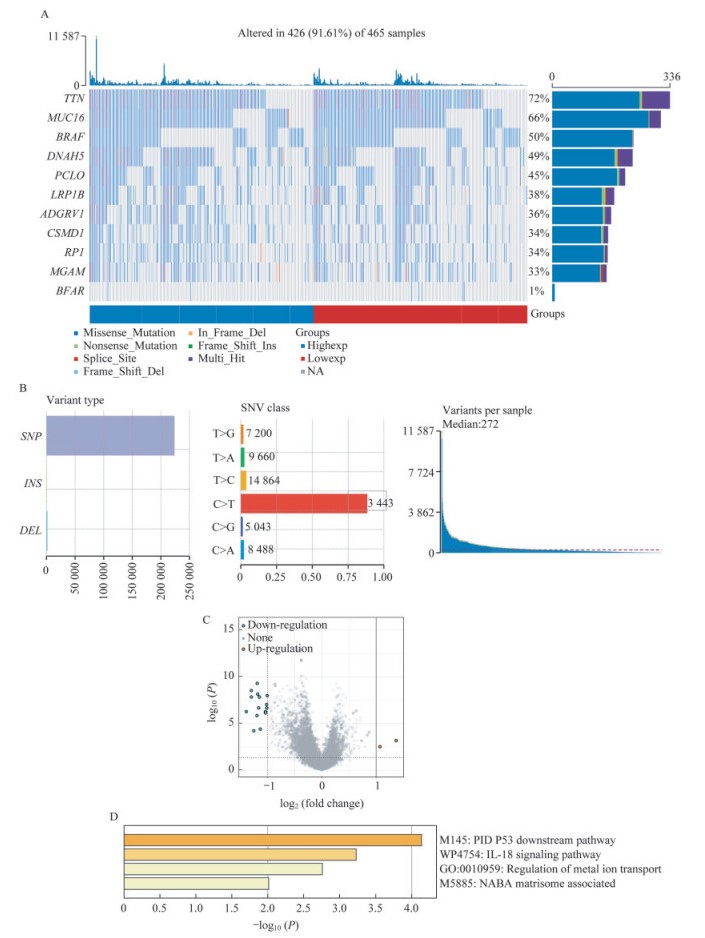
|
| A: Oncoplot shows the somatic landscape of SKCM tumor cohort.Genes are ordered by their mutation frequencies.B: Cohort summary plot shows the distribution of variants according to variant type and SNV class.Bottom part (from left to right) indicates mutation load of variant sample. C: Contrast BRAFMUT to BRAFWT group, 17 differentially expressed genes were screened out. Among these, 15 downregulated genes were indicated by blue dots and two upregulated genes were indicated by red dots. Adjusted P < 0.05 and log2 (fold change) > 1 or log2 (fold change) < -1 were defined as the threshold for the differential expression of mRNAs. D: Seventeen differentially expressed genes were enrichment analyzed by GO term via metascape platform. 图 1 BRAF基因突变景观及差异性表达基因 Fig 1 Landscape of BRAF mutation and differentially expressed genes |
| Gene | logFC | P |
| TYRP1 | 1.37 | 7.54E-4 |
| PMEL | 1.07 | 3.33E-3 |
| KCNN4 | -1.19 | 5.62E-10 |
| SPP1 | -1.30 | 3.31E-9 |
| CHL1 | -1.18 | 2.10E-6 |
| CMBL | -1.01 | 1.14E-8 |
| TF | -1.30 | 1.63E-8 |
| PTPRZ1 | -1.15 | 1.64E-8 |
| CMTM5 | -1.02 | 1.01E-7 |
| ITIH6 | -1.16 | 2.36E-7 |
| LIF | -1.01 | 2.39E-7 |
| LOXL4 | -1.39 | 5.91E-7 |
| ELOVL2 | -1.04 | 6.05E-7 |
| CTXND1 | -1.04 | 8.03E-7 |
| FXYD3 | -1.19 | 1.59E-6 |
| MAGEA3 | -1.13 | 4.43E-5 |
| MAGEC2 | -1.25 | 6.57E-5 |
CXCR4在SKCM中高表达,且突变组表达高于野生组 CXCR4与肿瘤细胞的关系较为密切,在介导肿瘤定向迁移、侵袭和转移中发挥重要的作用,同时也是趋化免疫细胞的关键分子。String数据库分析出CXCR4与p53、IL-18蛋白均存在相互作用关系(图 2A)。相关研究也报道了CXCR4的启动子区域存在p53的作用位点[11],并且CXCR4可以有效调控IL-18的表达[12]。我们进一步探索发现CXCR4 mRNA在SKCM癌组织(T)表达明显高于正常组织(N)(图 2B),且BRAFMUT组的表达水平明显高于BRAFWT组(图 2C)。综上,CXCR4与p53、IL-18通路密切相关,且在SCKM患者中高表达。
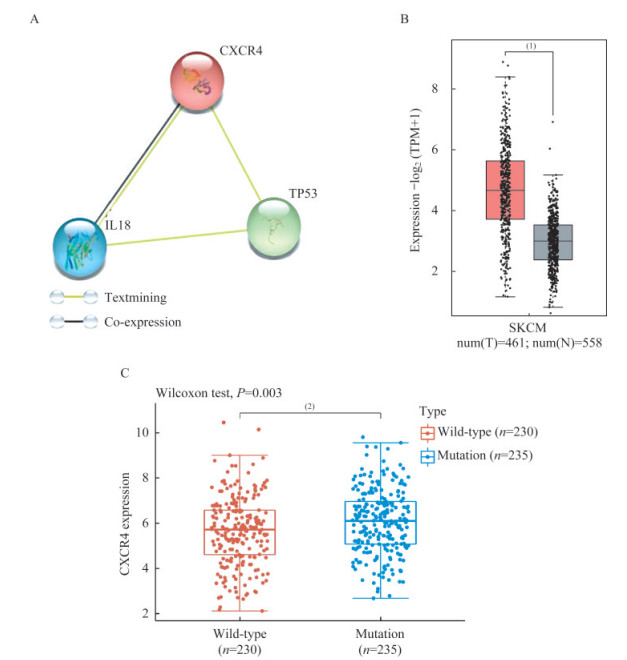
|
| A: Relationship of CXCR4, TP53 and IL18 were analyzed by String database. B: mRNA expressions of CXCR4 of SKCM (n=461) and normal (n=558) tissues from TCGA and GETx database were analyzed by GEPIA2. C: RNA-sequencing expression (level 3) profiles were downloaded from the TCGA dataset. The difference of expression of BRAFWT and BRAFMUT group were through the Wilcoxon test.(1)P < 0.05, (2)P < 0.01. 图 2 CXCR4在SKCM中表达升高,在BFAF突变患者中的表达更高 Fig 2 CXCR4 is elevated in SKCM and shows even higher expression in patients with mutated BRAF |
CXCR4表达与MSI和免疫检查点表达存在相关性 近年,SKCM的免疫治疗方面取得了突破性进展,尤其是抗PD-1/PD-L1治疗[13]。随着研究深入,发现抗PD-1治疗法在SKCM中的总体响应率不到40%[14]。因此,通过选择有效的标志物筛选出适合使用免疫治疗的患者显得至关重要。如今肿瘤治疗的标志物众多,MSI和免疫检查点是常用的免疫治疗生物标志物。据相关研究报道,高度MSI(MSI-H)的患者更适合进行抗PD-1治疗[15-16]。因此,我们分别在BRAFWT和BRAFMUT患者中分析了MSI与CXCR4表达的相关性。结果表明,在BRAFWT组中,CXCR4表达水平与MSI呈负相关性;而在BRAFMUT组中,两者无显著相关性(图 3A)。免疫检查点是抑制免疫系统的“刹车”,常在免疫细胞上表达,导致机体无法有效产生抗肿瘤免疫应答,进而促进肿瘤免疫逃逸。据报道,SIGLEC15、TIGIT、CD274、HAVCR2、PDCD1、CTLA4、LAG3和PDCD1LG2是T细胞衰竭相关的免疫检查点,也是比较重要的免疫靶点[17]。于是,我们进一步分析了CXCR4与上述免疫检查点之间的表达相关性,结果表明CXCR4与多个免疫检查点(HAVCR2、PDCD1、PDCD1LG2、TIGIT)表达呈正相关性(图 3B)。综上,我们认为CXCR4低表达的SKCM患者免疫检查点分子呈低表达,可能更适合进行免疫治疗。
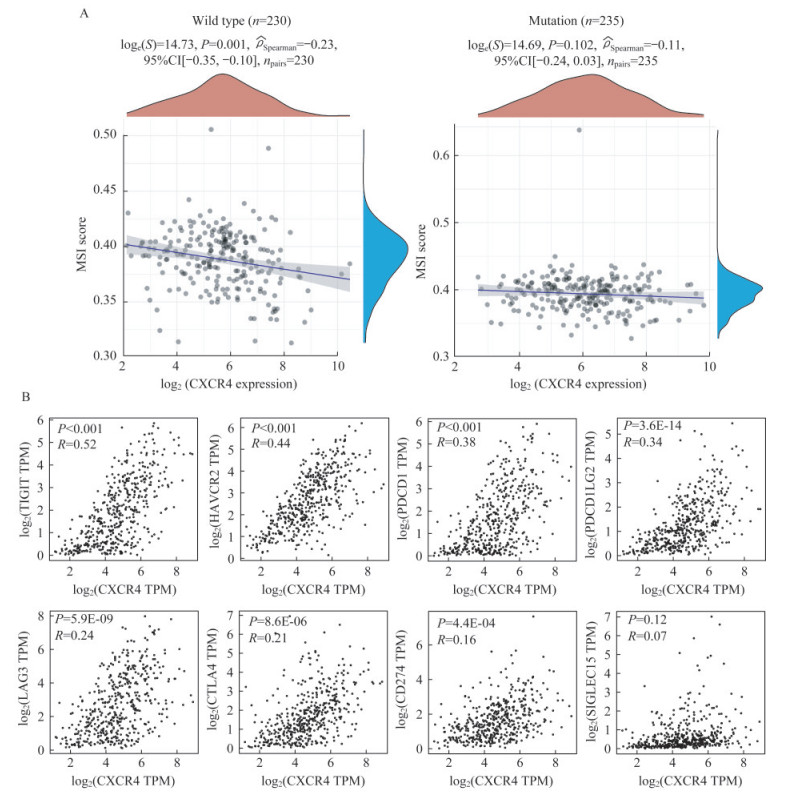
|
| A: Correlation analysis between CXCR4 expression and MSI in BRAFWT and BRAFMUT group was performed using Spearmans method. The abscissa represents CXCR4 expression distribution, and the ordinate represents MSI score distribution. The density curve on the right represents the distribution trend of MSI score; the upper density curve represents the distribution trend of CXCR4 expression. B: Pearson correlation analysis between CXCR4 expression and immune checkpoints genes (SIGLEC15, TIGIT, CD274, HAVCR2, PDCD1, CTLA4, LAG3 and PDCD1LG2) in SKCM tissues was performed by GEPIA. 图 3 CXCR4表达与MSI评分 Fig 3 CXCR4 expression and MSI score |
CXCR4低表达的SKCM患者更适合选择ICB治疗 虽然ICB治疗法改变了转移性SKCM患者的临床治疗方式,但是仍有大多数患者ICB治疗后无反应或治疗后会复发。我们选取了8个与免疫检查点相关的转录本(SIGLEC15、TIGIT、CD274、HAVCR2、PDCD1、CTLA4、LAG3和PDCD1LG2),并检测上述免疫检查点分子在G1(BRAFWT/CXCR4高表达)、G2(BRAFWT/CXCR4低表达)、G3(BRAFMUT/CXCR4高表达)和G4(BRAFMUT/CXCR4低表达)组中的表达情况。结果表明,上述8个基因在G2、G4组中的表达水平显著低于G1、G3组,即CXCR4低表达的患者免疫检查点表达水平低(图 4A)。另外,我们再通过肿瘤免疫功能障碍和排斥(tumor immune dysfunction and exclusion,TIDE)算法预测G1~G4组对免疫检查点抑制剂响应性。结果显示,G2组的TIDE评分在上述4组中最低(图 4B)。TIDE评分低,ICB疗效好,接受ICB治疗后生存期长。因此,CXCR4低表达的SKCM患者更适合ICB治疗法,尤其是BRAFWT的SKCM患者ICB治疗效果可能更好。

|
| A: Expression distribution of 8 immune checkpoints gene in groups G1-G4 was analyzed by R software. The ordinate represents the expression distribution of gene. The significance difference of four groups was tested with Kruskal-Wallis test. B: Potential ICB response of groups G1-G4 was predicted with TIDE algorithm. The statistical table of immune response of samples in different groups in the prediction results (upper). The distribution of immune response scores in different groups in the prediction results (below). The significance difference of four groups was tested with Kruskal-Wallis test. (1)P < 0.001, (2)P < 0.05, ns: No significance. 图 4 CXCR4低表达的患者免疫检查点表达低,更适合选择ICB治疗 Fig 4 Patients with lower expression of CXCR4 have lower expression of immune checkpoints and are more suitable for ICB therapy |
SKCM属于高侵袭性的恶性肿瘤,晚期患者使用化疗药物治疗后缓解率较低[18],总生存期不足6个月[14]。BRAF基因在SKCM中广泛突变,可驱动肿瘤发生发展,其突变往往意味着预后更差。随着人们对细胞信号转导和新型靶向药物的深入研究,特异性BRAF抑制剂的发现标志着SKCM治疗进入了新的时代。
免疫治疗改变了SKCM的治疗方式,起始于靶向表达在T细胞表面的CTLA-4分子。Ipilimumab可通过靶向CTLA-4,增强T细胞的活化与增殖,刺激抗肿瘤免疫应答,进而提高SKCM的总生存期[19]。近年来,除了研究MSI与肿瘤发生的关系外,MSI在肿瘤免疫治疗的响应预测方面的应用也是一项热门研究。MSI会产生大量的移码突变,产生“移码肽”,更容易刺激肿瘤浸润淋巴细胞的免疫应答,可能是MSI-H肿瘤对免疫治疗敏感的原因之一。CXCR4是人体肿瘤中表达最为广泛的趋化因子受体[20],我们发现CXCR4在SKCM中表达升高,且在BRAF突变的患者中升高更为明显,在BRAFWT患者中CXCR4表达水平与MSI负荷呈负相关。据报道,CXCR4/CXCL12轴能够促进肿瘤细胞增殖[21]和血管生成[5],促进免疫逃逸。BRAF突变往往预示着更差的临床结局。于是,我们认为CXCR4有望成为联合BRAF突变分析的合适标志物,CXCR4低表达的患者可能对免疫治疗更敏感。
近年来,ICB等免疫治疗法在肿瘤治疗领域取得了突破性进展,如抗CLTA-4[22]、PD-1[23-24]、PD-L1[25]在治疗转移性SKCM中均有很好的应用。尽管如此,并非所有的SKCM患者经过ICB治疗都会获益,部分患者会发生耐药,部分患者治疗无效,其机制尚不明确。CXCR4在肿瘤微环境中免疫细胞的转运、浸润及激活等方面发挥着关键的作用。我们也发现在SKCM中CXCR4与免疫检查点HAVCR2(TIM-3)、PDCD1(PD-1)、PDCD1LG2(PD-L2)和TIGIT呈正相关。T细胞介导的细胞免疫受到多个免疫检查点的严格调控,上述4个分子属于这样的免疫检查点。随着恶性肿瘤的进展,肿瘤细胞表面高表达上述免疫检查点分子,促进T细胞衰竭以及其他天然免疫细胞功能异常[26-27]。据相关研究报道,CXCR4刺激肿瘤转移到CXCL12大量表达的区域,CXCL12可通过排除T细胞,阻止T细胞发挥抗肿瘤活性,从而对ICB产生耐药性[28]。由此,我们认为CXCR4低表达的SKCM患者免疫检查点表达低,更适合选用ICB治疗法。我们进一步将CXCR4结合BRAF突变分析对SKCM患者进行了分组,并预测出CXCR4低表达尤其BRAFWT的患者ICB治疗效果更好。本研究为筛选适合使用ICB治疗的SKCM人群提供了理论基础,将CXCR4表达与BRAF突变分析联合应用,可提高ICB治疗的反应率,避免不合理的治疗。
本研究仅停留在分子表达及生物信息学模型评估层面,且患者数据来源于美国的TCGA数据库,纳入样本数量相对有限,后期我们将会入组本院治疗的SKCM患者以获取更加真实的结果,使得本文提出的治疗方案更具实际应用意义。
作者贡献声明 戴赛林 论文构思、撰写和修订,数据采集。邵甲云 可行性分析,数据统计和分析,模型运算。雪燕 图表制作,论文修订。郑洁 论文修订。
利益冲突声明 所有作者均声明不存在利益冲突。
| [1] |
DE LA FUENTE-GARCÍA A, OCAMPO-CANDIANI J. Cutaneous melanoma[J]. Gac Med Mex, 2010, 146(2): 126-135.
|
| [2] |
GUAN J, GUPTA R, FILIPP FV. Cancer systems biology of TCGA SKCM: efficient detection of genomic drivers in melanoma[J]. Sci Rep, 2015, 5: 7857.
[DOI]
|
| [3] |
LI Y, UMBACH DM, LI L. Putative genomic characteristics of BRAF V600K versus V600E cutaneous melanoma[J]. Melanoma Res, 2017, 27(6): 527-535.
[DOI]
|
| [4] |
DAVIES H, BIGNELL GR, COX C, et al. Mutations of the BRAF gene in human cancer[J]. Nature, 2002, 417(6892): 949-954.
[DOI]
|
| [5] |
MOUSAVI A. CXCL12/CXCR4 signal transduction in diseases and its molecular approaches in targeted-therapy[J]. Immunol Lett, 2020, 217: 91-115.
[DOI]
|
| [6] |
SUSEK KH, KARVOUNI M, ALICI E, LUNDQVIST A. The role of CXC chemokine receptors 1-4 on immune cells in the tumor microenvironment[J]. Front Immunol, 2018, 9: 2159.
[DOI]
|
| [7] |
ZHOU T, DAMSKY W, WEIZMAN OE, et al. IL-18BP is a secreted immune checkpoint and barrier to IL-18 immunotherapy[J]. Nature, 2020, 583(7817): 609-614.
[DOI]
|
| [8] |
DUPAUL-CHICOINE J, ARABZADEH A, DAGENAIS M, et al. The Nlrp3 inflammasome suppresses colorectal cancer metastatic growth in the liver by promoting natural killer cell tumoricidal activity[J]. Immunity, 2015, 43(4): 751-763.
[DOI]
|
| [9] |
SUN Q, FAN G, ZHUO Q, et al. Pin1 promotes pancreatic cancer progression and metastasis by activation of NF-κB-IL-18 feedback loop[J]. Cell Prolif, 2020, 53(5): e12816.
|
| [10] |
LI K, WEI L, HUANG Y, et al. Leptin promotes breast cancer cell migration and invasion via IL-18 expression and secretion[J]. Int J Oncol, 2016, 48(6): 2479-2487.
[DOI]
|
| [11] |
MEHTA SA, CHRISTOPHERSON KW, BHAT-NAKSHATRI P, et al. Negative regulation of chemokine receptor CXCR4 by tumor suppressor p53 in breast cancer cells: implications of p53 mutation or isoform expression on breast cancer cell invasion[J]. Oncogene, 2007, 26(23): 3329-3337.
[DOI]
|
| [12] |
HOU J, WANG C, MA D, et al. The cardioprotective and anxiolytic effects of Chaihujialonggumuli granule on rats with anxiety after acute myocardial infarction is partly mediated by suppression of CXCR4/NF-κB/GSDMD pathway[J]. Biomed Pharmacother, 2021, 133: 111015.
[DOI]
|
| [13] |
EGGERMONT AM, SPATZ A, ROBERT C. Cutaneous melanoma[J]. Lancet, 2014, 383(9919): 816-827.
[DOI]
|
| [14] |
WU X, WANG X, ZHAO Y, et al. Granzyme family acts as a predict biomarker in cutaneous melanoma and indicates more benefit from anti-PD-1 immunotherapy[J]. Int J Med Sci, 2021, 18(7): 1657-1669.
[DOI]
|
| [15] |
GREEN AK, FEINBERG J, MAKKER V. A review of immune checkpoint blockade therapy in endometrial cancer[J]. Am Soc Clin Oncol Educ Book, 2020, 40: 1-7.
|
| [16] |
GOODMAN AM, SOKOL ES, FRAMPTON GM, et al. Microsatellite-stable tumors with high mutational burden benefit from immunotherapy[J]. Cancer Immunol Res, 2019, 7(10): 1570-1573.
[DOI]
|
| [17] |
HU J, CHEN Z, BAO L, et al. Single-cell transcriptome analysis reveals intratumoral heterogeneity in ccrcc, which results in different clinical outcomes[J]. Mol Ther, 2020, 28(7): 1658-1672.
[DOI]
|
| [18] |
MISHRA H, MISHRA PK, EKIELSKI A, et al. Melanoma treatment: from conventional to nanotechnology[J]. J Cancer Res Clin Oncol, 2018, 144(12): 2283-2302.
[DOI]
|
| [19] |
VAN ALLEN EM, MIAO D, SCHILLING B, et al. Genomic correlates of response to CTLA-4 blockade in metastatic melanoma[J]. Science, 2015, 350(6257): 207-211.
[DOI]
|
| [20] |
SINGH AK, ARYA RK, TRIVEDI AK, et al. Chemokine receptor trio: CXCR3, CXCR4 and CXCR7 crosstalk via CXCL11 and CXCL12[J]. Cytokine Growth Factor Rev, 2013, 24(1): 41-49.
[DOI]
|
| [21] |
BIANCHI ME, MEZZAPELLE R. The chemokine receptor CXCR4 in cell proliferation and tissue regeneration[J]. Front Immunol, 2020, 11: 2109.
[DOI]
|
| [22] |
AGARWALA SS. Novel immunotherapies as potential therapeutic partners for traditional or targeted agents: cytotoxic T-lymphocyte antigen-4 blockade in advanced melanoma[J]. Melanoma Res, 2010, 20(1): 1-10.
[DOI]
|
| [23] |
GIDE TN, QUEK C, MENZIES AM, et al. Distinct immune cell populations define response to anti-PD-1 monotherapy and anti-PD-1/anti-CTLA-4 combined therapy[J]. Cancer Cell, 2019, 35(2): 238-255.e6.
[DOI]
|
| [24] |
HAMID O, ROBERT C, DAUD A, et al. Safety and tumor responses with lambrolizumab (anti-PD-1) in melanoma[J]. NEJM, 2013, 369(2): 134-144.
[DOI]
|
| [25] |
RIBAS A, ALGAZI A, ASCIERTO PA, et al. PD-L1 blockade in combination with inhibition of MAPK oncogenic signaling in patients with advanced melanoma[J]. Nat Commun, 2020, 11(1): 6262.
[DOI]
|
| [26] |
DIXON KO, TABAKA M, SCHRAMM MA, et al. TIM-3 restrains anti-tumour immunity by regulating inflammasome activation[J]. Nature, 2021, 595(7865): 101-106.
[DOI]
|
| [27] |
KALBASI A, RIBAS A. Tumour-intrinsic resistance to immune checkpoint blockade[J]. Nat Rev Immunol, 2020, 20(1): 25-39.
[DOI]
|
| [28] |
LU G, QIU Y, SU X. Targeting CXCL12-CXCR4 signaling enhances immune checkpoint blockade therapy against triple negative breast cancer[J]. Eur J Pharm Sci, 2021, 157: 105606.
[DOI]
|
 2023, Vol. 50
2023, Vol. 50


