近年来,恶性肿瘤的发病率逐年上升,肺癌因高发病率和高致死率而成为公共卫生领域主要问题之一[1]。随着低剂量螺旋CT的普及,肺癌的检出率逐年增高。但由于缺乏特异性症状及有效的早期生物学指标,肺癌的总体救治率依然不理想,大部分患者就诊时已处于中晚期,化疗是目前临床治疗中晚期肺癌的主要手段之一[2-3]。顺铂(cisplatin)是临床常见的化疗药物,在晚期非小细胞肺癌中,顺铂是无EGFR基因突变患者的一线化疗药物[4]。然而,部分化疗患者在使用顺铂一段时间后会出现严重的耐药或不良反应,极大地限制了化疗药物的继续使用。因此,亟需寻找一种增加患者化疗敏感性的方法。
临床调查发现荷瘤状态会增加患者发生抑郁症的概率,主要原因包括:疾病带来的身心压力,随之而来的身体情况逐渐恶化[5],以及抗肿瘤免疫治疗、放化疗、手术等过程[6]。资料显示,所有肿瘤患者中约有15%并发抑郁症[7],而肺癌患者中的抑郁症发病率更高,约为29%[8]。当癌症患者合并抑郁症和其他精神疾病时,会导致生活质量下降[9]、抗癌治疗依从性降低[10]、自杀风险升高[11]等。
氟西汀(fluoxetine)是治疗抑郁症的药物之一,不仅具有抗抑郁作用,还具有抗肿瘤作用。因此,把氟西汀作为肿瘤患者治疗中的辅助药物,在治疗抑郁的同时发挥一定程度的抗肿瘤作用,这是一种非常有前景的治疗方法。然而,关于氟西汀对肺癌细胞的具体作用机制及其与顺铂联合使用的研究报道较少。本研究拟检测氟西汀联合顺铂对肺癌细胞增殖、周期、迁移、侵袭及自噬能力的影响,并深入探讨可能的作用机制,旨在为中晚期肺癌伴抑郁的患者提供临床辅助治疗的新思路和新依据。
材料和方法试剂 肺癌细胞株(H460,上海中乔新舟生物有限公司);顺铂(山东齐鲁制药有限公司);氟西汀、结晶紫染液、细胞培养基(美国Sigma公司);胎牛血清(美国Gibco公司);CCK8试剂盒[日本东仁化学科技(上海)有限公司];细胞周期试剂(美国BD Sciences公司);p21、p27、p62、LC3单克隆抗体(美国Cell Signaling Technology公司);ACTIN、GAPDH及二抗(美国Proteintech公司);ECL试剂盒(美国Millipore公司);小干扰RNA(上海吉玛制药技术有限公司);X-tremeGENE siRNA转染试剂(瑞士Roche公司)。
CCK8实验 取对数生长期的H460细胞,计数后在96孔板中按比例(5000个/孔)加入细胞悬液,按不同的实验目的加入相应浓度的氟西汀(5、10、20、30和40 μmol/L)和顺铂(2.5、5、10和15 μmol/L),每组设3个复孔,孵育24 h后弃去培养基,避光加入100 μL/孔的检测试剂(CCK8试剂∶无血清培养基=1∶9),继续孵育1 h,检测光密度值。
Transwell迁移实验 取对数生长期的H460细胞,计数后在6孔板中按每孔(20×104个/孔)加入细胞悬液,设空白对照组和给药组(20 μmol/L氟西汀、10 μmol/L顺铂、20 μmol/L氟西汀+10 μmol/L顺铂)。细胞培养24 h后用无血清RMPI-1640培养基重悬细胞,在Transwell小室上室加入细胞悬液(8×104个细胞),下室加入800 μL完全培养基后继续培养24 h。用4%多聚甲醛(800 μL/孔)固定0.5 h后,PBS洗涤1~2遍,再用结晶紫染液染色15 min,用湿棉签轻擦滤过膜至菌落清晰可辨,每组随机在镜下(×200)取3个视野拍照并保存,ImageJ计数后取平均值进行统计分析。
Transwell侵袭实验 解冻Matrigel胶,然后用不完全培养基稀释基质胶至终浓度为250 μg/mL(94.6 μL不完全培养基+5.6 μL Matrigel胶),在Transwell小室的上室中加入100 μL配好的胶,置于培养箱中至胶凝固。余步骤同迁移实验。
细胞周期实验 取对数生长期的H460细胞,计数后在6孔板中按比例(20×104个/孔)加入细胞悬液,药物处理方法同Transwell迁移实验,细胞培养24 h后消化转移入离心管中,233×g离心5 min,洗涤2~3遍,加入70%乙醇重悬细胞,-20 ℃静置过夜,次日233×g离心5 min,去除乙醇,洗涤2~3遍,加入PI染料(500 μL/管)重悬细胞,避光室温孵育15 min,上机检测。
免疫荧光实验 取对数生长期的H460细胞,计数后在6孔板中按比例(10×104个/孔)加入细胞悬液,药物处理方法同Transwell迁移实验。24 h后吸净培养基,4%多聚甲醛固定,PBST洗涤,5% BSA封闭,4 ℃摇床敷一抗(按1∶300稀释),培养过夜;次日PBST洗脱,室温避光敷二抗(按1∶1 000稀释),弃二抗,加入DAPI染液孵育5 min,再次洗脱,于显微镜下拍摄荧光图片。
Western blot实验 取对数生长期的H460细胞,计数后在6孔板中按比例(20×104个/孔)加入细胞悬液,药物处理方法同Transwell迁移实验。24 h后弃去培养基,加入裂解液(SDS∶PMSF=100∶1,100 μL/孔)提取总蛋白,SDS-PAGE凝胶电泳,80 v恒压转膜,5%脱脂牛奶室温封闭,4℃摇床敷一抗(按1:1 000稀释),培养过夜,室温二抗(按1∶5 000稀释)孵育1 h,曝光显色。
siRNA转染实验 取对数生长期的H460细胞,计数后在6孔板中按比例(10×104个/孔)加入细胞悬液培养24 h。先配制转染体系(5 µL siRNA+8 µL转染试剂+200 µL Opti-MEM培养基),室温孵育15 min,加入相应培养孔中,继续培养6 h,更换培养基,按细胞周期和Western blot实验处理细胞。
统计学处理 所有实验均重复3次,所有数据均用x±s表示。采用GraphPad Prism5和Excel 2013处理数据,采用单因素方差分析和t检验分析两组数据之间的差异,P < 0.05为差异有统计学意义。
结果氟西汀增加顺铂对肺癌细胞增殖的抑制作用 随着药物浓度增加,与对照组相比,经氟西汀和顺铂处理的肺癌细胞(H460)活力逐渐降低(图 1A),当氟西汀(20 μmol/L)和顺铂(10 μmol/L)联合使用24 h后,细胞活力降低较单药更为明显(P=0.000 4)(图 1B)。经两药联合处理的细胞,细胞形态发生显著变化:细胞出现肿胀,胞内可见大量空泡样结构,细胞死亡增加(图 1C)。

|
| A: H460 cell was treated with the increasing concentrations of fluoxetine and cisplatin for 24 h, and cell proliferation was measured by CCK-8 assay. B: H460 cells were treated with fluoxetine (20 μmol/L), cisplatin (10 μmol/L) and fluoxetine plus cisplatin for 24 h, and cell viability was measured by CCK8 kit. C: Morphology of H460 cells treated with fluoxetine (20 μmol/L), cisplatin (10 μmol/L) and fluoxetine plus cisplatin after 24 h (scale bar=50 μm). D: The ability of migration and invasion of H460 cells treated with fluoxetine (20 μmol/L), cisplatin (10 μmol/L), and fluoxetine plus cisplatin was determined by transwell assay (scale bar=100 μm). E: Quantization of D, vs. Control, (1) P < 0.05, (2) P < 0.01. 图 1 氟西汀增加顺铂对肺癌细胞增殖、迁移、侵袭能力的抑制作用 Fig 1 Fluoxetine increased the inhibitory ability of cisplatin on the proliferation, migration and invasion of H460 cells |
氟西汀增加顺铂对肺癌细胞迁移侵袭能力的抑制作用 Transwell实验结果显示(图 1D和1E),与对照组相比,氟西汀组和顺铂组细胞迁移和侵袭数下降(迁移:242.00±22.09 vs. 132.00±40.79和43.70±14.72,P均 < 0.05;侵袭:62.00±16.00 vs. 31.50±11.50和16.00±1.00,P均 < 0.05),且两药联合后细胞数目下降更为明显(迁移:242.00±22.09 vs. 27.00±15.25,P=0.003;侵袭:62.00±16.00 vs. 6.00±1.00,P=0.014)。
氟西汀通过增加G0/G1期细胞阻滞而增加顺铂的敏感性 细胞周期结果显示(图 2A),与对照组相比,氟西汀处理后的肺癌细胞G0/G1期细胞比例增加(47.28%±4.00% vs. 56.43%±1.05%,P=0.026);经顺铂处理后S期细胞比例增加(38.55%±3.96% vs. 66.10%±3.80%,P=0.009)。当氟西汀与顺铂联合应用后,相较于顺铂单药,G0/G1期细胞比例增加(32.96%±4.78% vs. 24.51%±3.61%,P=0.042),S期细胞比例减少(57.36%±5.84% vs. 66.11%±3.80%,P=0.037)。Western blot结果显示(图 2C),氟西汀增加G0/G1期相关蛋白p27和p21的表达,且p21蛋白在联合组中增加更为明显。

|
| A: Representative FACS data of cell cycle analysis determined by flow cytometry in the 4 groups treated with fluoxetine (20 μmol/L), cisplatin (10 μmol/L) and fluoxetine plus cisplatin for 24 h. B: The representative immunofluorescence images of LC3 puncta in H460 cells treated with fluoxetine and fluoxetine plus cisplatin (scale bar=50 μm). C: The expression of p21, p27, p62 and LC3 in H460 cells were measured by immunoblotting analysis. vs. Control, (1) P < 0.05, (2) P < 0.01, (3) P < 0.001; vs. Fluoxetine, (4) P < 0.05, (5) P < 0.01, (6) P < 0.001; vs. Cisplatin, (7) P < 0.001. 图 2 氟西汀通过诱导细胞周期阻滞和自噬的形成增加顺铂对肺癌细胞的敏感性 Fig 2 Fluoxetine enhanced sensitivity to cisplatin in H460 cells via arrested cell cycle and induced autophagy |
氟西汀联合顺铂诱导细胞自噬 免疫荧光结果显示,与对照组相比,顺铂处理后的肺癌细胞未见明显自噬体生成,而当其与氟西汀联合使用后,可见明显的自噬体生成(绿色荧光),融合的荧光图像也证实产生的自噬体位于细胞质(图 2B)。Western blot结果显示(图 2C),使用顺铂后自噬相关蛋白LC3和p62表达未发生明显变化,而当顺铂和氟西汀联合使用时,LC3和p62蛋白明显增加。
氟西汀联合顺铂通过p21相关途径影响细胞的侵袭能力 Transwell迁移实验结果显示(图 3A),在氟西汀和顺铂联合治疗中,与NC-siRNA组相比,p21-siRNA组细胞迁移能力增加,但差异无统计学意义;而在侵袭实验中(图 3B),联合治疗组中p21-siRNA处理后的细胞侵袭能力明显增加(26.00±6.16 vs. 60.33±7.59,P=0.004)。
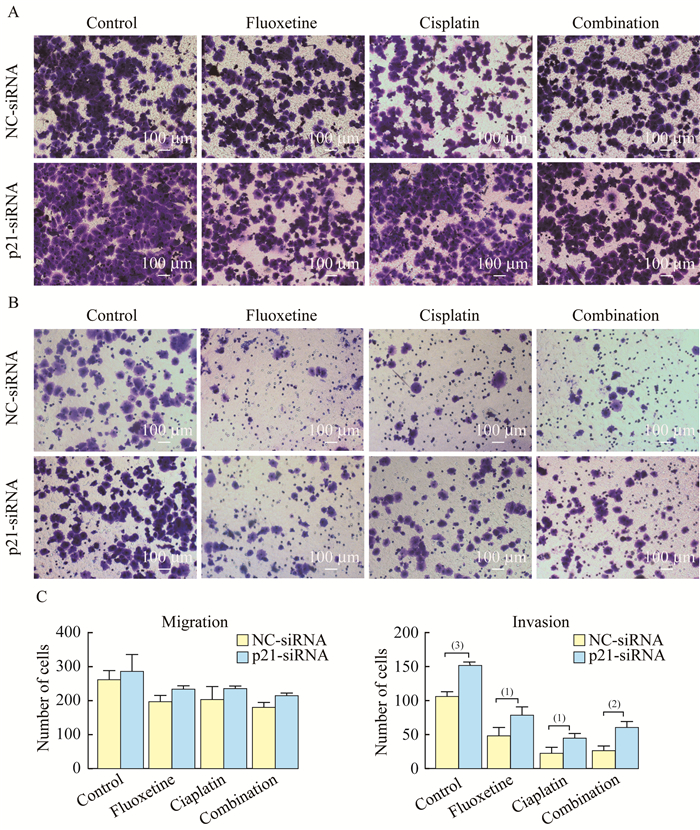
|
| The ability of migration (A) and invasion (B) of H460 cells treated with NC-siRNA and p21-siRNA was determined by transwell assay (scale bar=100 μm). C: The number of migration and invasion cells in each group. vs. NC-siRNA, (1) P < 0.05, (2) P < 0.01, (3) P < 0.001. 图 3 氟西汀联合顺铂通过p21相关途径影响细胞侵袭 Fig 3 Fluoxetine enhanced the sensitivity of cisplatin to H460 cells by inhibiting invasion via the p21-dependent signaling pathway |
氟西汀联合顺铂通过p21相关途径阻滞细胞周期及诱导细胞自噬 细胞周期结果显示(图 4A),在氟西汀和顺铂联合治疗中,与NC-siRNA组相比,p21-siRNA组G0/G1期细胞比例减少(52.46%±2.53% vs. 44.34%±3.32%,P=0.043),S期细胞比例增加(23.42%±4.98% vs. 33.84%±0.75%)。免疫荧光结果显示(图 4B),在氟西汀和顺铂联合治疗中,与NC-siRNA组相比,p21-siRNA组中自噬体未发现明显变化(绿色荧光)。Western blot结果显示(图 4C),在氟西汀和顺铂联合治疗中,与NC-siRNA组相比,p21-siRNA组p27、p21和p62蛋白均显著减少,而LC3蛋白未发生明显变化。
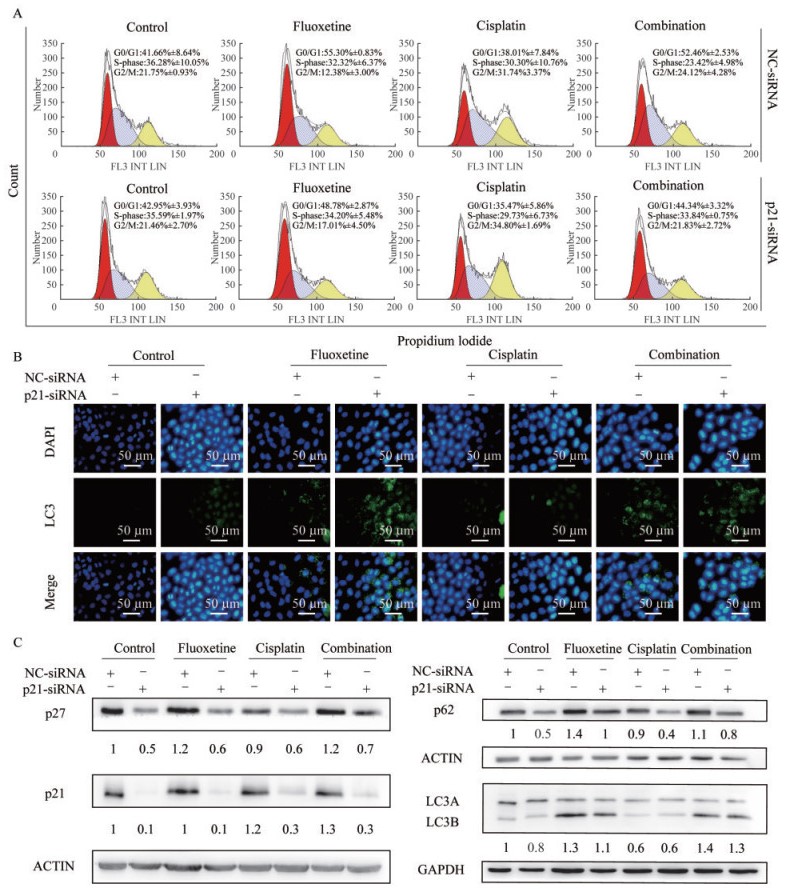
|
| A: Cell cycle distributions were determined by flowcytometry. B: Representative images of LC3 puncta in H460 cells treated with NC-siRNA and p21-siRNA (scale bar=50 μm). C: The protein level of p21, p27, p62 and LC3 in H460 cells were measured by Western blot. 图 4 氟西汀联合顺铂通过p21相关途径阻滞细胞周期及诱导细胞自噬 Fig 4 Fluoxetine enhanced the sensitivity of cisplatin to H460 cells by cell cycle arrest and autophagy via the p21-dependent signaling pathway |
作为高度恶性的肿瘤之一,肺癌约占肿瘤相关性死亡的30%[12]。肺癌的治疗面临诸多难题,如药物毒性、放化疗抵抗等,严重影响肺癌患者的预后[13]。确诊癌症对患者和家属都会造成巨大的心理负担,甚至可能导致患者由抑郁状态发展成抑郁症[14]。当肿瘤患者合并抑郁症时,不仅会增加肿瘤治疗的复杂性,也会影响治疗效果,使预后更差。虽然有文献报道氟西汀可以改善患者的化疗抵抗,但是关于氟西汀联合顺铂的具体作用机制却鲜少研究。因此,深入了解氟西汀与顺铂联合后能否产生抗肿瘤的作用及其具体机制成为亟待解决的问题。
恶性肿瘤具有向周围正常组织侵袭以及向远处组织转移的趋势,迁移能力表示细胞随着淋巴管或血管向远处移动的能力,而侵袭能力则是细胞通过破坏基质,进入血管或淋巴管的能力。肿瘤细胞迁移和侵袭能力的增强是导致肿瘤复发和加速进展的原因之一。本研究显示,氟西汀联合顺铂可以同时抑制细胞的迁移和侵袭能力,说明两种药物联合应用可以在一定程度上缓解肿瘤细胞的远处转移,减慢肿瘤病程。
细胞周期决定了细胞分裂的过程,而细胞周期紊乱是肿瘤的标志之一[15]。持续性应激会导致细胞周期调控机制紊乱,并最终导致肿瘤进展[16]。G0/G1期是细胞周期中的敏感期,受到外部信号干扰后就会导致细胞进程停滞[17]。本研究发现氟西汀和顺铂都可以影响细胞周期,相较于顺铂组,联合组中G0/G1期细胞占比增加。周期蛋白(cyclins)、周期蛋白依赖性激酶(cyclin-dependent kinases,CDKs)、周期依赖性激酶抑制因子(cyclin-dependent kinase inhibitors,CKIs)联合调控细胞周期,这些因子的异常表达常常导致周期紊乱,进而导致肿瘤的发生发展。p27和p21是细胞周期负向调节因子,它们分别结合并抑制CDK4/6-Cyclin D1和Cyclin A2/ CDK2复合物,阻止细胞由G1期进入S期。相较于顺铂单药组,联合用药组中p21和p27蛋白上调更为明显,因此推测虽然两种药物作用于不同的细胞周期,但联合应用后主要使细胞阻滞在G0/G1期,从而影响细胞分裂。
自噬是一把双刃剑,可以促进肿瘤的进展,也可以在肿瘤发生过程中杀伤吞噬肿瘤细胞而发挥抗肿瘤的作用[18]。有文献报道,抗抑郁药物可以通过免疫相关途径介导自噬的产生从而治疗患者的抑郁症[19]。有研究证实舍曲林和厄洛替尼联合应用可以通过诱导自噬来抑制肿瘤发生发展[20]。p62(sequestosome-1,SQSTM1)和LC3是参与自噬的重要调节剂之一[21]。激活自噬后,p62被转运至溶酶体降解同时使胞质型LC3(LC3-A)转变为膜型LC3(LC3-B),因此,可以通过定量分析p62和LC3-B/LC3-A的比值来间接评估细胞内自噬水平的高低。本研究中观察到p62和LC3蛋白表达升高,由此认为氟西汀联合顺铂可以诱导自噬,但同时也在一定程度上破坏了溶酶体的功能,从而使p62蛋白表现为反常性的表达增高。
p21负性调节CDKs,从而阻滞细胞周期于G0/G1而发挥作用[22]。p21在多种肿瘤中表达降低,且与肿瘤的预后密切相关。前期实验中调控p21相关通路可以增加药物对肿瘤细胞的杀伤作用[23]。而且,也有报道证实p21与肿瘤细胞的迁移和侵袭有一定的关系[24]。部分研究认为肿瘤细胞的迁移和侵袭能力增强与p21表达增加之间存在正相关性[25],也有实验发现p21表达上调之后抑制肿瘤细胞的迁移和侵袭[26]。这些现象使我们开始思考:p21相关通路是否参与氟西汀联合顺铂的抗肿瘤作用?如果参与,p21发挥何种作用?我们通过Transwell实验发现敲除p21能缓解氟西汀联合顺铂对肺癌细胞侵袭能力的抑制作用,分析p21敲除后的细胞周期发现,联合组中G0/G1期细胞比例下降,S期细胞比例升高。通过研究p21与自噬的关系发现,与NC-siRNA相比,p21敲除的联合组中虽然LC3表达量变化不大,但p62表达明显下调,而在氟西汀处理后,p62表达有所升高。综合这些数据我们认为氟西汀能通过p21信号通路增强顺铂的抗肿瘤作用。
综上所述,氟西汀联合顺铂在体外能通过p21依赖的途径抑制细胞侵袭能力,阻滞细胞周期,诱导细胞自噬,且作用效果优于单药,为临床治疗无EGFR基因突变的晚期肺癌化疗患者提供了一种新的治疗思路,但现有的实验基础对于临床的治疗指导作用仍然略显薄弱。本研究主要集中在细胞层面研究氟西汀和顺铂的药物作用机制,缺乏更为复杂及可靠的体内实验证据,因此需要通过进一步的研究来确定氟西汀联合顺铂是否也可以在体内发挥抗肿瘤的作用以及其在体内发挥抗肿瘤作用的具体机制是否也是通过p21相关通路。后续研究可进一步分析氟西汀联合顺铂在临床患者中的作用,并筛选联合用药在临床使用中的最佳配比。
作者贡献声明 邵莎莉 论文构思、撰写和修订,实验实施,数据采集。庄喜兵 数据采集,图表制作,论文撰写。乔田奎 论文构思和审校。
利益冲突声明 所有作者均声明不存在利益冲突。
| [1] |
BRAY F, FERLAY J, SOERJOMATARAM I, et al. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries[J]. CA Cancer J Clin, 2018, 68(6): 394-424.
[DOI]
|
| [2] |
HEIST RS, ENGELMAN JA. SnapShot: non-small cell lung cancer[J]. Cancer Cell, 2012, 21(3): 442-448.
|
| [3] |
ROSTI G, BEVILACQUA G, BIDOLI P, et al. Small cell lung cancer[J]. Ann Oncol, 2006, 17(Suppl 2): 5-10.
|
| [4] |
KANDA S, OHE Y, GOTO Y, et al. Five-year safety and efficacy data from a phase Ib study of nivolumab and chemotherapy in advanced non-small-cell lung cancer[J]. Cancer Sci, 2020, 111(6): 1933-1942.
[DOI]
|
| [5] |
IRWIN MR. Depression and insomnia in cancer: prevalence, risk factors, and effects on cancer outcomes[J]. Curr Psychiatry Rep, 2013, 15(11): 404.
[DOI]
|
| [6] |
ONITILO AA, NIETERT PJ, EGEDE LE. Effect of depression on all-cause mortality in adults with cancer and differential effects by cancer site[J]. Gen Hosp Psychiatry, 2006, 28(5): 396-402.
[DOI]
|
| [7] |
MITCHELL AJ, CHAN M, BHATTI H, et al. Prevalence of depression, anxiety, and adjustment disorder in oncological, haematological, and palliative-care settings: a meta-analysis of 94 interview-based studies[J]. Lancet Oncol, 2011, 12(2): 160-174.
[DOI]
|
| [8] |
HOPWOOD P, STEPHENS RJ. Depression in patients with lung cancer: prevalence and risk factors derived from quality-of-life data[J]. J Clin Oncol, 2000, 18(4): 893-903.
[DOI]
|
| [9] |
ARRIETA O, ANGULO LP, NUNEZ-VALENCIA C, et al. Association of depression and anxiety on quality of life, treatment adherence, and prognosis in patients with advanced non-small cell lung cancer[J]. Ann Surg Oncol, 2013, 20(6): 1941-1948.
[DOI]
|
| [10] |
COLLEONI M, MANDALA M, PERUZZOTTI G, et al. Depression and degree of acceptance of adjuvant cytotoxic drugs[J]. Lancet, 2000, 356(9238): 1326-1327.
[DOI]
|
| [11] |
SHIM EJ, PARK JH. Suicidality and its associated factors in cancer patients: results of a multi-center study in Korea[J]. Int J Psychiatry Med, 2012, 43(4): 381-403.
[DOI]
|
| [12] |
CHEN W, ZHENG R, BAADE PD, et al. Cancer statistics in China, 2015[J]. CA Cancer J Clin, 2016, 66(2): 115-132.
[DOI]
|
| [13] |
TSUBURAYA A, MORITA S, KODERA Y, et al. A randomized phase Ⅱ trial to elucidate the efficacy of capecitabine plus cisplatin (XP) and S-1 plus cisplatin (SP) as a first-line treatment for advanced gastric cancer: XP ascertainment vs. SP randomized PII trial (XParTS Ⅱ)[J]. BMC Cancer, 2012, 12: 307.
[DOI]
|
| [14] |
GOPALAN MR, KARUNAKARAN V, PRABHAKARAN A, et al. Prevalence of psychiatric morbidity among cancer patients - hospital-based, cross-sectional survey[J]. Indian J Psychiatry, 2016, 58(3): 275-280.
[DOI]
|
| [15] |
URREGO D, TOMCZAK AP, ZAHED F, et al. Potassium channels in cell cycle and cell proliferation[J]. Philos Trans R Soc Lond B Biol Sci, 2014, 369(1638): 20130094.
[DOI]
|
| [16] |
STEPULAK A, RZESKI W, SIFRINGER M, et al. Fluoxetine inhibits the extracellular signal regulated kinase pathway and suppresses growth of cancer cells[J]. Cancer Biol Ther, 2008, 7(10): 1685-1693.
[DOI]
|
| [17] |
MASSAGUE J. G1 cell-cycle control and cancer[J]. Nature, 2004, 432(7015): 298-306.
[DOI]
|
| [18] |
RUBINSZTEIN DC, CODOGNO P, LEVINE B. Autophagy modulation as a potential therapeutic target for diverse diseases[J]. Nat Rev Drug Discov, 2012, 11(9): 709-730.
[DOI]
|
| [19] |
ALCOCER-GOMEZ E, CASAS-BARQUERO N, WILLIAMS MR, et al. Antidepressants induce autophagy dependent-NLRP3-inflammasome inhibition in major depressive disorder[J]. Pharmacol Res, 2017, 121: 114-121.
[DOI]
|
| [20] |
JIANG X, LU W, SHEN X, et al. Repurposing sertraline sensitizes non-small cell lung cancer cells to erlotinib by inducing autophagy[J]. JCI Insight, 2018, 3(11): e98921.
[DOI]
|
| [21] |
LIU WJ, YE L, HUANG WF, et al. p62 links the autophagy pathway and the ubiqutin-proteasome system upon ubiquitinated protein degradation[J]. Cell Mol Biol Lett, 2016, 21: 29.
[DOI]
|
| [22] |
YOON MK, MITREA DM, OU L, et al. Cell cycle regulation by the intrinsically disordered proteins p21 and p27[J]. Biochem Soc Trans, 2012, 40(5): 981-988.
[DOI]
|
| [23] |
XU S, HUANG H, CHEN YN, et al. DNA damage responsive miR-33b-3p promoted lung cancer cells survival and cisplatin resistance by targeting p21(WAF1/CIP1)[J]. Cell Cycle, 2016, 15(21): 2920-2930.
[DOI]
|
| [24] |
KREIS NN, LOUWEN F, YUAN J. The multifaceted p21 (Cip1/Waf1/CDKN1A) in cell differentiation, migration and cancer therapy[J]. Cancers (Basel), 2019, 11(9): 1220.
[DOI]
|
| [25] |
KREIS NN, FRIEMEL A, RITTER A, et al. Function of p21 (Cip1/Waf1/CDKN1A) in migration and invasion of cancer and trophoblastic cells[J]. Cancers (Basel), 2019, 11(7): 989.
[DOI]
|
| [26] |
SANG Y, ZHANG R, SUN L, et al. MORF4L1 suppresses cell proliferation, migration and invasion by increasing p21 and E-cadherin expression in nasopharyngeal carcinoma[J]. Oncol Lett, 2019, 17(1): 294-302.
[URI]
|
 2023, Vol. 50
2023, Vol. 50


