2. 上海中医药大学附属光华中西医结合医院麻醉科 上海 200052
2. Department of Anesthesiology, Guanghua Integrative Medicine Hospital, Shanghai University of Traditional Chinese Medicine, Shanghai 200052, China
孕妇反复或长期暴露于麻醉药物可能对发育中的婴儿产生神经毒性, 但其机制仍不清楚[1]。七氟醚是临床上常用的吸入性麻醉剂, 使用方便、麻醉效果佳、呼吸道刺激小, 是目前临床应用最为广泛的吸入麻醉药物。七氟醚可能会对大脑发育产生影响, 从而引起行为障碍, 该过程的病理机制可能涉及氧化应激、神经细胞凋亡、神经炎症、突触性质改变等。我国每年有超过1 000万孕妇接受麻醉, 怀孕期间重复或长期麻醉药物暴露会导致发育神经的损害和子代的认知功能紊乱。长时间吸入麻醉对脑部神经突触有损伤作用[2], 特别是对未成熟的神经元会导致明显的神经损害[3-4]。长时间接触高浓度七氟醚会改变新生大脑中的葡萄糖和氨基酸代谢过程及细胞内的抗氧化剂与渗透物质系统[5], 降低脂质中磷脂酰乙醇胺、磷脂酰丝氨酸和磷脂酰甘油, 并增加4-羟基非烯醇[6], 而磷脂酰丝氨酸参与神经元存活、神经突起生长和突触形成有关的信号通路[7-10]。已知脂质的异常变化可能会打破磷脂的平衡, 大部分吸入麻醉药都是脂溶性的, 易透过胎盘屏障。探讨药物诱导损伤模型中新的生物标志物, 特别是从脑和血清样本中识别出新的生物标志物, 可能有助于早期发现麻醉药物暴露的潜在神经毒性。目前对于吸入七氟醚后子代神经发育的影响存在争议, 孕期大鼠持续吸入七氟醚后, 有些子代神经细胞发生了凋亡和行为学的改变[11], 但也有一些实验为阴性结果。因此, 母代孕晚期接触七氟醚是否会引起子代神经发育的障碍需要进一步的科学探索。本研究首次通过脂质组学和转录组学探讨七氟醚对子代发育神经的潜在毒性机制。利用超高效液相色谱质谱(ultra performance liquid chromatography/time-of-flight mass spectrometry, UPLC/MS)技术从脂质组学方面来明确哪一种脂质代谢参与神经潜在损伤的发生, 通过RNA-Seq进一步筛选差异表达基因, 并使用RT-qPCR在损伤模型上进一步验证损伤过程中对应基因的变化。
材料和方法麻醉和试剂 动物研究(包括大鼠安乐死程序)按照上海交通大学实验动物中心标准管理规程执行(编号:SMP-ADM-000-A), 我校实验动物中心的许可证号(编号:A 2016077), 遵循国际实验动物评估及认可委员会(AAALAC)和动物保护和使用机构委员会(IACUC)的规定进行动物护理。
孕15~17天的SD大鼠(体质量400~500 g)购自上海斯莱克实验动物有限责任公司, 无病毒、细菌和寄生虫病原体。将孕大鼠单独放入标准箱房中饲养, 喂食颗粒饲料(江苏医药生物工程有限公司)和高温杀菌纯净水。饲养条件:温度(23±2)℃, 相对湿度50%±15%, 12 h光暗轮替, 空气压力100 kPa, 15次/h空气完全过滤一次。试剂:HPLC级乙腈和甲酸(美国Merck公司), TOF-MS校准液(美国AB SCIEX公司), 超纯水来源于Milli-Q纯净水系统(美国米利波公司)。
实验分组 将28只孕大鼠随机分为实验组(S组)和对照组(C组), 每组14只。饲养1~3天后, S组孕大鼠在麻醉箱内接受2%七氟醚+98%氧气6 h; C组孕大鼠在麻醉箱内以相同的流速吸入100%氧气6 h, 总气体流量为400 mL/min, 用气体分析仪(德国Drager公司)测定氧、二氧化碳和七氟醚的浓度。七氟醚麻醉终止后, 每组随机取8只孕大鼠, 腹主动脉血样采集40 μL, 用I-STAT 1分析仪(MN:300-G, 美国Abbott Park公司)分析血气。其余孕大鼠回饲养箱内等待分娩, S组子代43只, C组子代48只, 每组随机抽出12只出生后7天的子代大鼠, 七氟醚麻醉, 2组新生大鼠断头后采集血清标本, 即刻在冰面上取出海马和皮层组织。血清标本、海马及皮层组织于-80 ℃冷冻保存备用。
样品制备和UPLC/TOF-MS 12 000×g离心(4 ℃) 10 min获得子代血清样本, 取10 μL转移到无菌的EP管中, 加入10 μL优质等级纯水及5 μL异丙醇(10 μg/mL), 用40 μL冷异丙醇+1 %甲酸(v/v)进行旋涡混合, 沉淀蛋白质。-20 ℃静置20 min, 13 000×g离心10 min, 取10 μL上清液转移到玻璃式HPLC小瓶(96孔板)中进行衍生化, 采用HPLC-MS联用技术进行分析。用电喷雾电离(electrospray ionization, ESI)在正离子模式下进行LC-MS检测, 用多重反应监测(multiple reaction monitoring, MRM)对每种化合物进行定量分析。色谱分析采用ACQUITY超高效液相色谱系统(美国Waters公司)分析, 水同步高清晰度飞行时间质谱(TOF-MS)系统(美国Waters公司)阴极ESI源在UPLC上以正负模式显示, IDA模型用于正负离子模式, 由TOF-测量扫描(m/z=100)和紧随6次TOF-MS/MS扫描(m/z=100~1 500)组成, 累积时间分别为0.08 s和0.1 s, 以确保测量质量的精确性。
PLS-DA分析 在代谢组学分析之前, 使用Marker View软件(美国AB SCIEX公司)对原始数据进行预处理, 并将其归一化为总面积, 以纠正每个样本的浓度偏差。采用偏最小二乘判别分析(PLS-DA)方法进行检测, 比较样本间差异, 确定关键化合物, 并对数据进行处理。获取包含样本变量的数据矩阵, 以分析下一个代谢物。在PLS-DA模型中可变影响的化合物预测值(Vip)>1.0和P < 0.05获得潜在生物标记物[12]。
HE染色 对6只新生大鼠的海马和大脑皮质神经元进行HE染色, 检测细胞凋亡。切片去蜡, 水合, HE染色。切片脱水、包埋, 400倍显微镜(日本奥林巴斯公司)下计数CA1锥体细胞数目并拍照, 仅包括细胞核和核仁明显的细胞。
TUNEL染色 将6只新生大鼠海马和皮层组织制成冰冻切片, 浸泡在含3% H2O2的PBS中, 消除内源性过氧化物酶反应。PBS(pH=7.4)洗涤3次、每次5 min, 加入20 % FBS和3 %FBS蛋白15 min。加入与荧光素连接的TUNEL反应液, 切片置于37 ℃加湿箱中1 h, PBS洗涤3次, 每次5 min。切片在室温下与终止液反应10 min, 在37 ℃下与抗地高辛过氧化物酶抗体反应30 min, PBS洗涤3次, 每次5 min。再用DAB(3, 3’-二氨基联苯胺)显影, 二甲苯透明后用中性树脂密封。切片置于荧光显微镜下拍照, 并观察TUNEL阳性染色结果。
RNA提取 将6只新生大鼠的皮层组织块与少量液氮混合后迅速研磨, 重复3次。将皮层组织细胞培养皿置于冰上, 加入Trizol裂解5~10 min。4 ℃下12 000×g离心15 min, 吸取上层水相, 移至另一离心管中, 按Trizol:异戊醇=1:0.6 (v/v)混匀, 室温放置5~10 min。4 ℃下12 000×g离心10 min, 弃上清, 按Trizol:75 %乙醇=1:1(v/v)温和振荡, 悬浮沉淀。室温晾干或真空干燥5~10 min, 检测260 nm下吸光度(D)值, 定量RNA浓度。
RNA-seq实验流程 提取的总RNA样品经琼脂糖电泳和Nanodrop质检及定量后, 用oligo(dT)磁珠富集mRNA, 若RNA为降解样品或为原核样品则直接用rRNA去除试剂盒进行处理; RNA测序文库均由试剂盒完成, 包括RNA片段化后用随机引物反转生成第一链cDNA, 加入dUTP合成第二链cDNA, 双链cDNA末端修复加A后连接Illumina匹配接头, PCR扩增得到最终文库; 构建好的文库用Agilent 2100进行质检, 并由qPCR方法进行文库定量, 使用Illumina Hiseq 4000测序仪进行测序。
统计学分析 采用Graph Prism 5.0软件进行统计分析。计量资料以x±s表示, Vcan基因RNA-seq变化采用独立样本t检验或单因素方差分析。检查方差的正态性和同质性假设。KEGG通路出现在相应富集条目中的差异表达基因数目用Fisher’s精确检验计算P值。Benjamini-Hochberg错误发现率(false discovery rate, FDR)校正P值。P < 0.05为差异有统计学意义。
结果动脉血气分析 为消除低氧血症或二氧化碳蓄积等因素干扰, 对实验动物进行动脉血气分析(表 1), 血气分析结果与氧和二氧化碳交换相关的各项数据都在正常安全范围之内, 两组间pH值和动脉血二氧化碳的差异无统计学意义。S组大鼠仅受七氟醚的影响而不受七氟醚麻醉所致动脉血气的影响, 因此动物模型建成。
| Observation item | C group (n=8) | S group (n=8) |
| pH | 7.37±0.03 | 7.41±0.01 |
| PaCO2 (mmHG) | 43.80±3.50 | 46.97±4.25 |
| BE (mmol/L) | -0.33±1.53 | 4±1.0 |
| HCO3 (mmol/L) | 25.57±1.75 | 29.90±1.90 |
| SaO2 (%) | 98.33±0.58 | 100±0 |
| C group:Control group; S group:Sevoflurane prenatal exposure group.pH:Potential of hydrogen; PaCO2:Partial pressure of carbon dioxide in arterial blood; HCO3:Carbonic acid hydrogen radical; BE:Residual value of blood alkali; SaO2:Degree of blood oxygen saturation. | ||
UPLC/TOF-MS分析七氟醚诱导的神经毒性 UPLC/TOF-MS分析分别采用正电离和负电离两种模式, 获得代谢组学图谱(图 1)。分别对每组6个QC样品进行测定, 在负离子和正离子模式下产生10个生物标记的保留时间和6个共峰的峰面积配对保留时间m/z(表 2)。保留时间的RSD < 3.71%, 峰面积的RSD为3.62%~38.58%。
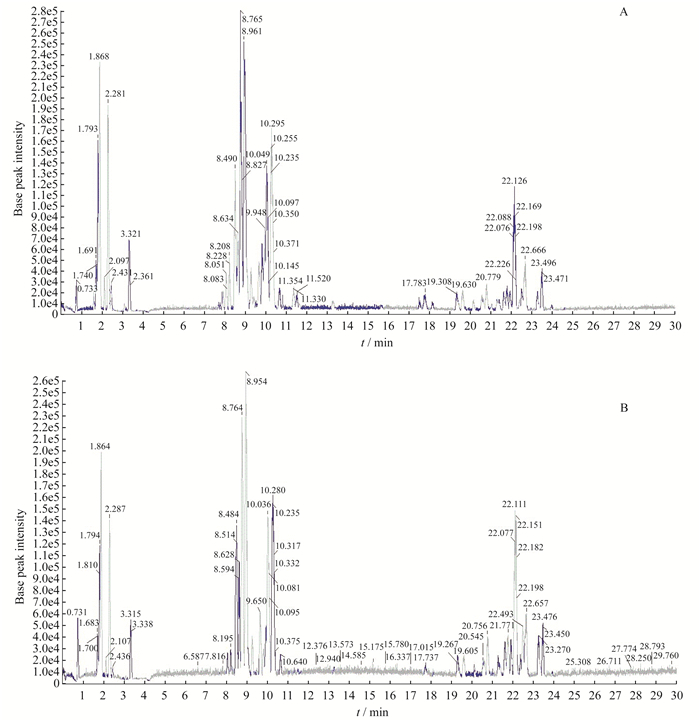
|
| A:ESI positive mode; B:ESI negative mode.The UPLC/TOF-MS analysis was performed using an Acquity TMUPLC system coupled to a SynaptTMG2 high-definition time-of-flight mass spectrometry system with ESI positive and negative modes.In both positive and negative ion modes, basic peak chromatogram profiles displayed no difference between S and C groups.ESI:Electrospray ionization. 图 1 基于UPLC/TOF-MS分析新生大鼠血清阳性和阴性ESI的典型基峰强度色谱图 Fig 1 Typical base peak intensity chromatogram of the rat serum obtained in ESI positive and negative mode based on UPLC/TOF-MS analysis |
| No. | m/z | RT (min) |
Ion | Metabolite | Related pathways | Related diseases | Change trend |
| 1 687 | 496.341 0 | 2.27 | [M+H]+ | LysoPC (16:0) | Glycerophospho lipid metabolism | Pancreatic cancer, Barth sgl, Atherosclerosis | ↑ |
| 2 918 | 707.499 1 | 2.96 | [M+K]+ | DG (20:1n9/0:0/20:5n3) | Pancreatic cancer, Barth sgl, Atherosclerosis | ↑ | |
| 3 488 | 774.563 8 | 7.86 | [M+H]+ | PE (22:6(4Z, 7Z, 10Z, 13Z, 16Z, 19Z)/P-18:1(11Z)) | Glycerophospho lipid metabolism | Pancreatic cancer, Barth sgl | ↓ |
| 3 037 | 728.521 7 | 8.57 | [M+Na]+ | SM (d18:0/16:0) | Sphingolipid metabolism | Atherosclerosis | ↓ |
| 3 929 | 808.589 7 | 8.85 | [M+H]+ | PC (16:0/22:5(7Z, 10Z, 13Z, 16Z, 19Z)) | Glycerophospho lipid metabolism | ↓ | |
| 3 897 | 806.572 9 | 9.08 | [M+Na]+ | Galactosylceramide (d18:1/22:0) | Sphingolipid metabolism | Hidradenitis suppurativa | ↓ |
| 3 535 | 780.550 5 | 9.13 | [M+Na]+ | CerP (d18:1/26:0) | ↓ | ||
| 3 238 | 756.555 9 | 9.8 | [M+Na]+ | PC (16:0/16:0) | Glycerophospho lipid metabolism | Adolescent idiopathic scoliosis | ↓ |
| 5 586 | 923.747 6 | 22.92 | [M+H]+ | TG (20:5(5Z, 8Z, 11Z, 14Z, 17Z)/18:2(9Z, 12Z)/20:5(5Z, 8Z, 11Z, 14Z, 17Z)) | ↑ | ||
| 4 776 | 857.756 8 | 23.98 | [M+K]+ | TG (14:0/20:1(11Z)/15:0) | ↓ | ||
| RT:Retention time (min); m/z:Mass-to-charge ratio.The independent t test was verified by Graph Prism 5.0 in serum of group S and group C.P < 0.05.↑:Up regulation.↓:Down regulation. | |||||||
偏最小二乘判别分析(PLS-DA) 两组采用UPLC/TOF-MS样品剖面进行主成分分析(principal component analysis, PCA)和PLS-DA检测。在正离子模式下获得更好的分离效果(图 2)。采用3个参数对P的性能进行评价。LS-DA模型在正离子模式下:R2X=0.665, R2Y=0.996, Q2=0.962, 因此选择PLS-DA模型。

|
| C:PLS-DA in negative ion mode.S:PLS-DA in positive ion mode. 图 2 新生大鼠血清PLS-DA分析图 Fig 2 The serum of neonatal rats analyzed by PLS-DA |
七氟醚治疗脂质代谢途径分析 用Metabo Analyst(http://www.metaboanalyst.ca)进一步分析鉴定出的生物标志物(图 3)。发现6条代谢途径:甘油磷脂代谢、糖基磷脂酰肌醇(glycosylphos phatidylinositol, GPI)-锚定生物合成、亚油酸代谢、α-亚麻酸代谢, 鞘脂代谢和花生四烯酸代谢, 其中甘油磷脂代谢是最重要的代谢途径。
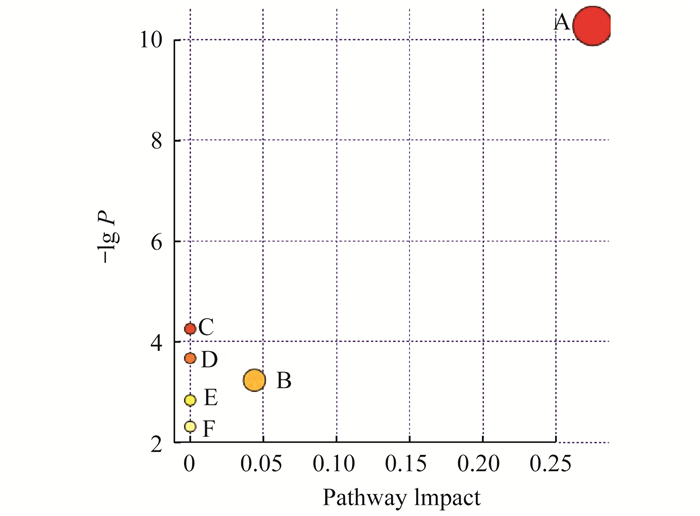
|
| Pathway analysis on biomarkers of sevoflurane-induced neurogenerative disease model.All matched pathways were acquired according to P values from pathway enrichment analysis and pathway impact values from pathway topology analysis, using pathway library of Rattus norvegicus (rat).A:Glycerophospholipid metabolism; B:Glycosylphos phatidylinositol (GPI)-anchor biosynthesis Linoleic acid metabolism; C:Alpha-linolenic acid metabolism; D:Sphingolipid metabolism; E:Arachidonic acid metabolism. 图 3 七氟醚诱导神经退行性病变疾病模型生物标志物的通路分析 Fig 3 The pathway analysis of biological markers of the neurodegeneration disease model induced by sevoflurane |
HE染色和TUNEL染色 TUNEL染色分析各子代大鼠海马和大脑皮质神经元凋亡(图 4A、4B)。S组染色质致密、深染, 形成致密团块, HE染色的组织学切片中可能出现断裂。细胞体积缩小, 胞浆致密, 嗜酸性。HE染色和TUNEL染色结果显示两组细胞凋亡差异无统计学意义。
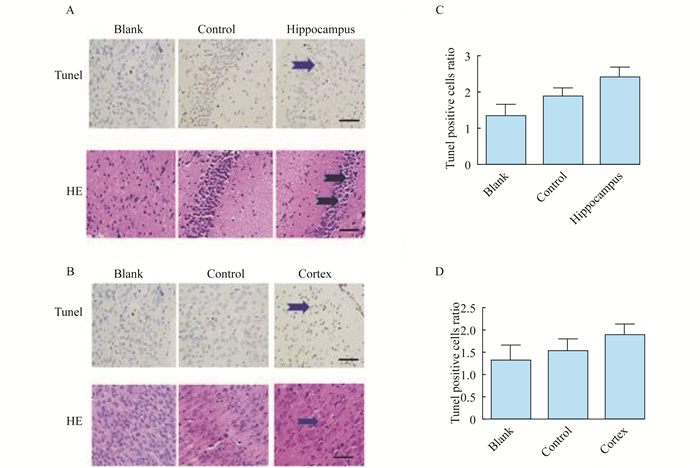
|
| The experiment was repeated 3 times.Apoptosis in hippocampus and cerebral cortex was detected by TUNEL and HE staining.Blank:Blank group; Control:Control group; Hippocampus:Hippocampus group; Cortex:Cortex group.A and B:The black arrows indicate apoptotic cells.Observation of HE and TUNEL staining for the hippocampus and cerebral cortex tissue in morphological changes in each group (scale bar=20 μm).C and D:The statistical analysis of neural cell apoptosis was detected by TUNEL and HE staining from hippocampi and cerebral cortex tissue of each offspring rat. 图 4 各组海马和大脑皮层组织HE染色和TUNEL染色的形态变化(×400) Fig 4 HE and TUNEL staining for the hippocampus and cerebral cortex tissue in morphological changes of each group (×400) |
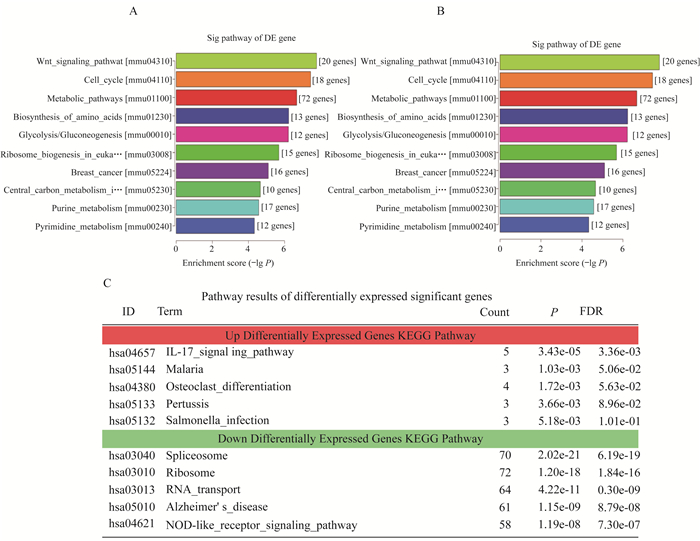
RNA-seq技术 利用RNA-seq技术对S组和C组试验前后转录本中的差异表达基因进行全面分析(图 5), Vcan基因确实有表达上调的趋势, 两组差异有统计学意义, 并发现相关差异表达显著基因的通路(图 6):红色为上调, 绿色为下调, RT-qPCR验证Vcan基因表达上调明显。
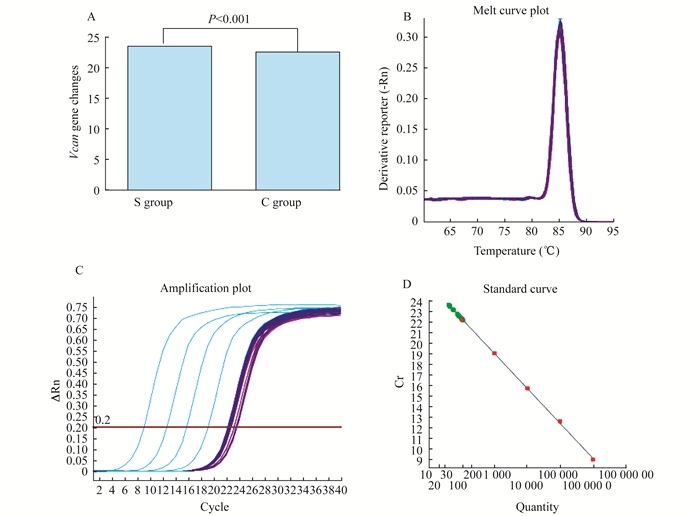
|
| A:The change of Vcan gene by RT-PCR; B:The melting point curve for drawing; C:The expansion region Vcan gene.D:As the standard curve. 图 5 Vcan基因RT-PCR变化及熔融和扩增曲线 Fig 5 Vcan gene PCR RT-PCR changes, melting and amplification curves |
|
| A and B:The KEGG pathway analyzed the top 10 item bar diagrams.In P-value order from low to high, the ordinate represents P-value(-log10 transformation); C:Pathway analysis of the first 5 Pathway of up and down significantly differentially expressed genes KEGG Pathway.Pathway analysis is a functional analysis mapping genes to KEGG pathways.The P-value (EASE-score, Fisher-P value or Hypergeometric-P value) denotes the significance of the pathway correlated to the conditions.Lower the P-value, more significant is the pathway.The recommend P-value cut-off is 0.05.Upregulated differential expression gene KEGG pathway analysis results folder; Down-regulated differential expression gene KEGG pathway analysis results folder.DE:Differentially expressed. 图 6 调节基因的信号通路和差异表达显著基因的通路 Fig 6 Signal pathway of differentially expressed genes and pathway results of differentially expressed significant genes |
本研究基于脂质组学, 在七氟醚诱导子代潜在神经损伤的过程中, 发现6种代谢途径, 包括甘油磷脂代谢途径、GPI-锚定生物合成、亚油酸代谢、α-亚麻酸代谢、鞘脂代谢和花生四烯酸代谢, 其中甘油磷脂代谢是最重要的代谢途径。脂质代谢的改变被认为是导致中枢神经系统损伤的关键因素[13], 血脂异常与阿尔茨海默症、帕金森症和亨廷顿症等疾病有关。吸入麻醉药异氟醚低浓度时表现为神经保护, 高浓度时诱导细胞凋亡[14]。母体妊娠过程中暴露于七氟醚可能会对其后代的神经发育产生风险[3]。本研究中, MetaboAnalyst系统用于进一步分析已鉴定的生物标志物, 在S组中发现潜在的内源性代谢产物甘油磷脂和鞘磷脂。将以上3种方法与稳定同位素标记相结合, 可用于分析脂类代谢(脂肪酸、甘油磷脂和鞘脂代谢)[15]。
甘油磷脂是参与哺乳动物细胞膜中的主要脂类。在许多神经退行性疾病中磷脂酰胆碱稳态被破坏[16], 当溶血磷脂酰胆碱水平升高, 可导致神经鞘脱髓鞘及不同程度的轴突变性[17]。细胞内磷脂酰胆碱含量减少, 可能有助于预防或治疗阿尔茨海默症[18], 在内质网中磷脂稳态破坏后, 磷脂失衡可导致许多神经疾病。
鞘脂代谢改变最终导致神经损伤。抑制PKC的溶血磷脂会产生神经毒性, 而且这些脂类会导致细胞膜结构的变化, 特别是细胞膜上脂肪通道的功能改变[19]。膜脂(如神经节苷脂和鞘脂)与可溶性Aβ和不溶性形式相互作用[20-22], 并影响Aβ神经毒性。鞘磷脂是由游离鞘氨醇基及其磷酸盐、神经酰胺、鞘氨酰肌醇和复杂的鞘磷脂组成的一类不同的脂质群类, 可在神经疾病等各种疾病中检测到这类脂质群[23-25]。因此, 鞘脂代谢与神经系统疾病密切相关, 可能与神经疾病的发病机制有关。本研究中, 脂质组学的发现进一步证实产前接触七氟醚对子代的潜在神经毒性。
在转录组学方面, 本研究首次揭示七氟醚子代神经毒性的转录表达规律, 发现相关差异表达显著基因的通路, 进一步RT-qPCR验证发现, 发现孕大鼠暴露于七氟醚后, 新生大鼠皮层Vcan基因表达明显上调, Vcan基因与一种名为versican的蛋白质有关。根据KEGG数据库(Kyoto Encyclopedia of Genes and Genomes)中的生物学通路分类条目和基因表达情况, 过表达对理解Vcan如何调控包括神经发育、功能和修复在内的大脑皮层潜在毒性具有重要意义[26]。
基于UPLC/TOF-MS的脂肪组学和RNA-seq测序为探讨脂质失衡导致发育神经发生相关潜在毒性提供了全面的信息。本研究中, RNA-seq技术应用于孕晚期大鼠暴露于七氟醚对其子代的潜在神经毒性, 对精确调控七氟醚的浓度和时间有重要的意义。本研究提示异常的甘油磷脂和鞘脂代谢可能诱导潜在神经毒性, 产前接触七氟醚所致子代潜在发育神经毒性的病理过程还需要更多的证据, 调节甘油磷脂和鞘脂代谢可作为孕晚期吸入麻醉药长时间暴露后潜在神经损伤的治疗手段。本研究为今后进一步探讨孕晚期模式动物七氟醚暴露的脂质代谢组学、转录组学和相关潜在神经毒理学分子机制提供了理论依据。
| [1] |
PINYAVAT T, SARAIYA IVR, CHEN J, et al. Anesthesia exposure in children:practitioners respond to the 2016 FDA drug safety communication[J]. Neurosurg Anesthesiol, 2019, 31(1): 129-133.
[DOI]
|
| [2] |
LIU J, WANG P, ZHANG X, et al. Effects of different concentration and duration time of isoflurane on acute and long-term neurocognitive function of young adult C57BL/6 mouse[J]. Int J Clin Exp Pathol, 2014, 7(9): 5828-5836.
[PubMed]
|
| [3] |
CATTANO D, VALLEGGI S, ABRAMO A, et al. Nitrous oxide discretely up-regulates nNOS and p53 in neonatal rat brain[J]. Minerva Anestesio, 2010, 76(6): 420-424.
[PubMed]
|
| [4] |
LIU F, RAINOSEK SW, FRISCH-DAIELLO JL, et al. Potential adverse effects of prolonged sevoflurane exposure on developing monkey brain:from abnormal lipid metabolism to neuronal damage[J]. Toxicol Sci, 2015, 147(2): 562-572.
[DOI]
|
| [5] |
LIU B, GU Y, XIAO H, et al. Altered metabolomic profiles may be associated with sevoflurane-induced neurotoxicity in neonatal rats[J]. Neurochem Res, 2015, 40(4): 788-799.
[DOI]
|
| [6] |
WILLIAMS T, LYNN B, MARKESBERY W, et al. Increased levels of 4-hydroxynonenal andacrolein, neurotoxic markers of lipid peroxidation, in the brain in mild cognitive impairment and early Alzheimer's disease[J]. Neurobiol Aging, 2006, 27(8): 1094-1099.
[DOI]
|
| [7] |
AKBAR M, CALDERON F, Wen Z, et al. Docosahexaenoic acid:A positive modulator of Akt signaling in neuronal survival[J]. Proc Natl Acad Sci USA, 2005, 102(31): 10858-10863.
[DOI]
|
| [8] |
HUANG BX, AKBAR M, KEVALA K, et al. Phosphatidylserine is a critical modulator for Akt activation[J]. J Cell Biol, 2011, 192(6): 979-992.
[DOI]
|
| [9] |
KIM HY. Novel metabolism of docosahexaenoic acid in neural cells[J]. J Biol Chem, 2007, 282(26): 18661-18665.
[DOI]
|
| [10] |
KIM HY, AKBAR M, KIM YS. Phosphatidylserine-dependent neuroprotective signaling promoted by docosahexaenoic acid[J]. Prostaglandins Leukot Essent Fatty Acids, 2010, 82(4-6): 165-172.
[DOI]
|
| [11] |
王宇恒, 成永霞, 焦晶华, 等. 大鼠妊娠前后吸入异氟烷或七氟烷对子代脑发育的影响[J]. 中国医科大学学报, 2015, 44(2): 143-147. [DOI]
|
| [12] |
WU L, LIANG W, CHEN W, et al. Screening and analysis of the marker components in ganoderma lucidum by HPLC and HPLC-MSn with the aid of chemometrics[J]. Molecules, 2017, 22(4): 584.
[DOI]
|
| [13] |
RAO MA, HATCHER JF. Role of lipids in brain injury and diseases[J]. Future Lipidol, 2007, 2(4): 403-422.
[DOI]
|
| [14] |
YANG Z, LV J, LI X, et al. Sevoflurane decreases self-renewal capacity and causes c-Jun N-terminal kinase-mediated damage of rat fetal neural stem cells[J]. Sci Rep, 2017, 7: 46304.
[DOI]
|
| [15] |
SHEVCHENKO A, SIMONS K. Lipidomics:coming to grips with lipid diversity[J]. Nat Rev Mol Cell Biol, 2010, 11(8): 593-598.
[DOI]
|
| [16] |
MORGAN NV, WESTAWAY SK, MORTON JE, et al. PLA2G6, encoding a phospholipase A2, is mutated in neurodegenerative disorders with high brain iron[J]. Nat Genet, 2006, 38(7): 752-754.
[DOI]
|
| [17] |
HIRATSUKA D, KURGANOV E, FURUBE E, et al. VEGF- and PDGF-dependent proliferation of oligodendrocyte progenitor cells in the medulla oblongata after LPC-induced focal demyelination[J]. J Neuroimmunol, 2019, 332: 176-186.
[DOI]
|
| [18] |
KO M, HATTORI T, ABDULLAH M, et al. Yamane T2, Michikawa M4.Phosphatidylcholine protects neurons from toxic effects of amyloid β-protein in culture[J]. Brain Res, 2016, 1642: 376-383.
[DOI]
|
| [19] |
HAWKINS-SALSBURY J, PARAMESWAR A, JIANG X, et al. Sands MSPsychosine, the cytotoxic sphingolipid that accumulates in globoid cell leukodystrophy, alters membrane architecture[J]. Lipid Res, 2013, 54(12): 3303-3311.
[DOI]
|
| [20] |
SOYON H, BETH LO, TING Y, et al. Soluble Abeta oligomers are rapidly sequestered from brain ISF in vivo and bind GM1 ganglioside on cellular membranes[J]. Neuron, 2014, 82(2): 308-319.
[DOI]
|
| [21] |
RUIZ A, JOSHI P, MASTRANGELO R, et al. Testing Abeta toxicity on primary CNS cultures using drug-screening microfluidic chips[J]. Lab Chip, 2014, 14(15): 2860-2866.
[DOI]
|
| [22] |
MARTINS IC, KUOERSTEIN I, WILKINSON H, et al. Lipids revert inert Abeta amyloid fibrils to neurotoxic protofibrils that affect learning in mice[J]. Embo J, 2008, 27(1): 224-233.
[DOI]
|
| [23] |
VAN EDG, WALTER J. Sphingolipids:critical players in Alzheimer's disease[J]. Prog Lipid Res, 2012, 51: 378-393.
[DOI]
|
| [24] |
HORRES CR, HANNUN YA. The roles of neutral sphingomyelinases in neurological pathologies[J]. Neurochem Res, 2012, 37(6): 1137-1149.
[DOI]
|
| [25] |
HIA T, DNNENBERG AJ. Sphingolipid signaling in metabolic disorders[J]. Cell Metab, 2012, 16(4): 420-434.
[DOI]
|
| [26] |
WU Y, SHENG W, LIWEN C, et al. Versican V1 isoform induces neuronal differentiation and promotes neurite outgrowth[J]. Mol Biol Cell, 2004, 15(5): 2093-2104.
[DOI]
|
 2019, Vol. 46
2019, Vol. 46


