乳腺癌是常见的恶性肿瘤之一, 全球每年新增病例约150万, 严重危害女性的健康。早期乳腺癌预后较好, 而晚期乳腺癌发生淋巴结转移和血性转移, 预后往往较差, 因此早诊断、早治疗可提高乳腺癌疗效及改善预后[1]。活组织检测有侵入性, 且存在一定的取样误差, 患者依从性不高。由于乳腺癌缺乏敏感的生物标记物, 简便易行的乳腺癌早期诊断指标仍在探寻中。随着对肿瘤机制的研究越来越深入, 微小RNA(miRNA)已经成为新型基因水平的肿瘤标记物, 可用于肿瘤精确诊断, 但组织标本获取有侵入性, 限制了其在临床的应用[2]。血清miRNA相对较易获得, 并且稳定性较好, 在反复冻融、酸碱和煮沸等恶劣环境下不易降解, 故循环miRNA受到越来越多的关注[3]。本研究通过检测血清miR-141和miR-195-5p, 观察其与临床指标、增殖和迁移的相关性, 并评估其在乳腺癌早期诊断中的价值。
资料和方法临床资料 选择2015年1月至2018年6月在复旦大学附属华山医院诊断为乳腺癌的患者126例, 作为乳腺癌组, 平均年龄(52.37±6.37)岁(35~79岁); 术后病理TNM分期:Ⅰ期22例, Ⅱ期43例, Ⅲ期50例和Ⅳ期11例; 组织分级:Ⅰ级25例, Ⅱ级57例和Ⅲ级44例。选择同期在我院活组织检测为乳房良性病变患者75例, 作为良性病变组, 平均年龄(53.19±7.19)岁(45~76岁); 选择同期在我院行健康体检者45例, 为健康对照组, 平均年龄(52.49±7.16)岁(45~73岁)。纳入标准:健康对照组和乳腺良性病变组无合并其他肿瘤; 乳腺癌患者术前未行化疗、放疗等其他治疗(其中包括11例Ⅳ期术前不愿接受化疗和放疗的患者); 所有患者均签订知情同意书。排除标准:非乳腺癌; 肝炎、结核和急性肺炎等急慢性感染; 脏器功能不全(如肝脏、肾脏和肺等); 有疾病相关用药史。各组一般资料差异无统计学意义, 各组比较的基线相同。
血液标本的抽取 来院第1天和手术后1周抽取空腹肘静脉血约5 mL, 室温静置20 min, 3 000 r/min离心10 min (离心半径15 cm), 分离出血清约3 mL, -80 ℃保存待检测。
血清miR-141和miR-195-5p的检测 采用苯酚氯仿法提取血清RNA。按照说明书进行miRNA的逆转录反应, 其引物由上海昕浩生物合成。miRNA-141引物序列, 正向:5’-ACACTCCAGCTGGGC-ATCTTCCAGTACAGT-3’, 反向5’-CTCAACT-GGTGTCGTGGAGTCGCCAATTCAGTTGAGT-CCAACAC-3’; miR-195-5p引物序列, 正向:5’-G-ATAGCAGCACAGAAATATTGGC-3’, 反向:5’-TGCGGGTGCTCGCTTCGGCAAGT-3’; 以U6作为内参的引物序列, 正向:CTCGCTTCGGCAG-CACA, 反向:AACGCTTCACGAATTT-GCGT。qRT-PCR的总反应体系为20 μL, 主要组成为MgCl2 1.2 μL, PCR缓冲液2 μL, dNTPs (10 mmol/L) 0.4 μL, Taq酶0.3 μL, ddH2O 14.77 μL, 探针和引物共0.33 μL, cDNA 1 μL。循环参数:95 ℃ 5 min, 95 ℃ 15 s, 60 ℃ 1 min, 共40个循环。每个样品设置3个复孔, 并以DEPC处理的ddH2O为阴性对照。检测结束后采用SDS2.0软件分析获得Ct值(图 1)。并用前期合成iRNA的cDNA 1 μmmol/L, 按10-2、10-3、10-4、10-5和10-6稀释后作为标准品, 以浓度的对数为X轴, 对应每个浓度的Ct值为Y轴, 绘制成标准曲线, 根据Ct值通过标准曲线计算出miR-141和miR-195-5p的绝对浓度。
|
| 图 1 miR-141 (A)和miR-195-5p (B)的溶解曲线 Fig 1 Dissolution curves of miR-141 (A) and miR-195-5p (B) |
慢性病毒转染构建 对人乳腺癌细胞MX-1进行慢病毒包装, 取对数生长期MX-1细胞与250 μL无血清DMEM高糖培养基和0.5 μg PMD2.G包装载体加入到3支1.5 mL无菌Eppendorf管中, 分别加入1.5 μg miR-141、miR-195-5p全长载体和相应的空白载体, 另取无菌Eppendorf管加入无血清DMEM高糖培养基, 并与20 μL转染脂质体混合, 室温静置20 min, 胰蛋白酶消化, 将细胞稀释至1×106/L, 取1 mL细胞悬液, 接种于6孔板, 加入DNA和脂质体, 培养过夜后更换培养液, 48和72 h后分别收集病毒悬液并过滤。病毒悬液和10%DMEM培养基按1:1混合后培养MX-1细胞3天, 筛选出细胞株, 转染miR-141和miR-195-5p全长载体的分别为miR-141组和miR-195-5p组, 转染空白载体的为对照组。
细胞增殖能力检测 胰酶消化的MX-1细胞以2×106/L接种于96孔板, 检测时间点为0、24、48和72 h, CKK-8试剂与10%DMEM高糖培养液1:9混合培养细胞1 h, 检测波长为450 nm, 以各时点的吸光度(D)值与D0值(0 h时)的比值衡量细胞的相对增殖能力。
细胞迁移能力检测 无血清DMEM高糖培养液按1:8稀释Matrigel基质胶, 共200 μL。取3只Transwell小室放置于24孔板中, 迁移细胞密度调整为1×106/L, 下室放置700 μL含10% FBS的高糖培养基, 培养24 h后用10%甲醛固定细胞20 min, 上室加入0.5%结晶紫溶液50 μL, 拍照。用棉签擦去上室未穿出的细胞, 200倍光镜下计数, 取四周及中央5个视野, 求平均值。
观察指标 观察各组血清miR-141和miR-195-5p水平变化, 乳腺癌患者术后血清miR-141和miR-195-5p水平变化, 乳腺癌患者血清miR-141和miR-195-5p水平与临床指标、增殖和迁移的相关性, 及其诊断乳腺癌的灵敏度和特异性。
统计学分析 采用SPSS15.0和MedCalc15.0软件进行统计分析。正态资料的计量资料用x±s表示, 独立因素两组比较采用t检验; 多组比较采用方差分析, 两两比较采用q检验。以血清miR-141和miR-195-5p联合检测作为检验变量进行Logistic回归; 用病理检查结果为金标准, 用ROC曲线预测乳腺癌发生概率、诊断阈值、灵敏度和特异性。检验水准α=0.05, P < 0.05为差异有统计学意义。
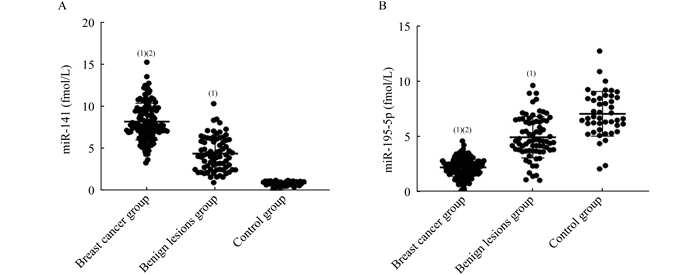
结果各组血清miR-141和miR-195-5p水平比较 血清miR-141水平:乳腺癌组显著高于良性病变组和健康对照组(P < 0.01), 良性病变组较健康对照组显著升高(P < 0.01);血清miR-195-5p水平:乳腺癌组明显低于良性病变组和健康对照组(P < 0.01), 良性病变组较健康对照组明显降低(P < 0.01)(表 1, 图 2)。
| (x±s) | |||
| Groups | Case (n) |
miR-141 (fmol/L) |
miR-195-5p (fmol/L) |
| Control | 45 | 0.88±0.23 | 6.94±2.09 |
| Benign lesions | 75 | 4.43±1.98(1) | 4.93±1.88(1) |
| Breast cancer | 126 | 8.22±2.19(1)(2) | 2.20±0.84(1)(2) |
| F | 311.428 | 227.994 | |
| P | 0.000 | 0.000 | |
| (1)vs.control group, P < 0.01;(2)vs.benign lesions group, P < 0.01. | |||
|
| (1)vs.control group, P < 0.01;(2)vs.benign lesions group, P < 0.01. 图 2 各组血清miR-141(A)和miR-195-5p(B)表达的比较 Fig 2 Comparison of serum miR-141 (A) and miR-195-5p (B) expressions in different groups |
良性病变组和乳腺癌组术后血清miR-141和miR-195-5p水平表达 乳腺癌组术后血清miR-141水平为(1.37±0.57) fmol/L, 明显低于术前的(8.22±2.19) fmol/L(t=33.978, P < 0.01);乳腺癌组术后血清miR-195-5p水平为(5.77±1.62) fmol/L, 明显高于术前的(2.20±0.84) fmol/L (t=21.960, P < 0.01)。乳腺良性病组术后血清miR-141水平为(1.25±0.86) fmol/L, 明显低于术前的(4.43±1.98) fmol/L(t=12.867, P < 0.01);乳腺良性病组术后血清miR-195-5p水平为(6.26±1.86) fmol/L, 明显高于术前的(4.93±1.88) fmol/L (t=4.355, P < 0.01)。
乳腺癌患者血清miR-141和miR-195-5p水平与临床参数的关系 乳腺癌组血清miR-141和miR-195-5p水平与年龄和肿瘤直径无明显相关性, 而与淋巴结转移、雌激素受体(estrogen receptor, ER)、孕激素受体(progesteron receptor, PR)、人类表皮因子受体2 (human epidermal growth factor receptor 2, HER-2)、TNM分期、分子分型和组织分型具有明显的相关性(P < 0.01, 表 2), 通过多元线性回归发现, 血清miR-141和miR-195-5p水平与淋巴结转移、PR、TNM分期和分子分型呈线性相关(P < 0.05, 表 3)。
| (x±s) | |||||||
| Clinical indexes | Cases (n) | miR-141 (fmol/L) | t or F | P | miR-195-5p (fmol/L) | t or F | P |
| Age (y) | 0.128 | 0.899 | 0.626 | 0.533 | |||
| ≤50 | 52 | 8.19±2.06 | 2.23±0.38 | ||||
| >50 | 74 | 8.24±2.23 | 2.18±0.48 | ||||
| Tumor diameter (cm) | 0.289 | 0.773 | 1.376 | 0.171 | |||
| ≤2 | 25 | 8.34±2.13 | 2.09±0.39 | ||||
| >2 | 101 | 8.19±2.37 | 2.23±0.47 | ||||
| Lymph node metastasis | 5.981 | < 0.001 | 9.490 | < 0.001 | |||
| + | 51 | 9.68±2.38 | 1.76±0.32 | ||||
| - | 75 | 7.23±2.17 | 2.50±0.49 | ||||
| ER | 3.157 | 0.002 | 6.707 | < 0.001 | |||
| + | 81 | 7.75±2.26 | 2.38±0.35 | ||||
| - | 45 | 9.06±2.18 | 1.88±0.48 | ||||
| PR | 5.241 | < 0.001 | 6.451 | < 0.001 | |||
| + | 62 | 7.17±2.16 | 2.46±0.39 | ||||
| - | 64 | 9.24±2.27 | 1.95±0.49 | ||||
| HER-2 | 5.299 | < 0.001 | 4.303 | < 0.001 | |||
| + | 49 | 6.90±2.15 | 2.42±0.47 | ||||
| - | 77 | 9.06±2.28 | 2.06±0.45 | ||||
| TNM staging | 35.208 | < 0.001 | 90.297 | < 0.001 | |||
| Ⅰ | 22 | 5.46±1.86 | 3.43±0.49 | ||||
| Ⅱ | 43 | 7.28±2.06 | 2.35±0.47 | ||||
| Ⅲ | 50 | 9.31±2.27 | 1.74±0.42 | ||||
| Ⅳ | 11 | 12.46±2.34 | 1.24±0.46 | ||||
| Molecular typing | 41.888 | < 0.001 | 186.188 | < 0.001 | |||
| Luminal A | 47 | 6.16±1.86 | 3.26±0.51 | ||||
| Luminal B | 43 | 8.42±1.96 | 2.03±0.45 | ||||
| HER-2(+) | 16 | 9.64±2.29 | 1.38±0.43 | ||||
| Basal-like | 20 | 11.50±2.37 | 0.73±0.44 | ||||
| Tissue typing | 16.126 | < 0.001 | 112.078 | < 0.001 | |||
| Ⅰ | 25 | 6.19±1.86 | 3.16±0.49 | ||||
| Ⅱ | 57 | 8.38±2.18 | 2.27±0.45 | ||||
| Ⅲ | 44 | 9.17±2.37 | 1.56±0.41 | ||||
| ER:Estrogen receptor; PR:Progesteron receptor; HER-2:Human epidermal growth factor receptor 2. | |||||||
| Dependent variable | B | SD | t | P | 95%CI of B |
| miR-141 | |||||
| Tissue typing | -0.050 | 0.215 | -0.231 | 0.818 | -0.475-0.375 |
| Molecular typing | 0.608 | 0.121 | 5.009 | 0.000 | 0.367-0.848 |
| TNM staging | 1.152 | 0.148 | 7.765 | 0.000 | 0.858-1.445 |
| HER-2 | -0.270 | 0.185 | -1.460 | 0.147 | -0.637-0.096 |
| PR | -0.456 | 0.159 | -2.875 | 0.005 | -0.771-(-0.142) |
| ER | -0.384 | 0.242 | -1.583 | 0.116 | -0.863-0.096 |
| Lymph node metastasis | 0.638 | 0.276 | 2.307 | 0.023 | 0.090-1.185 |
| miR-195-5p | |||||
| Tissue typing | -0.074 | 0.081 | -0.905 | 0.367 | -0.235-0.087 |
| Molecular typing | -0.227 | 0.046 | -4.926 | 0.000 | -0.318-(-0.135) |
| TNM staging | -0.452 | 0.056 | -8.040 | 0.000 | -0.563-(-0.341) |
| HER-2 | 0.106 | 0.070 | 1.513 | 0.133 | -0.033-0.245 |
| PR | 0.169 | 0.060 | 2.801 | 0.006 | 0.049-0.288 |
| ER | 0.027 | 0.092 | 0.297 | 0.767 | -0.155-0.209 |
| Lymph node metastasis | -0.216 | 0.105 | -2.060 | 0.042 | -0.423-(-0.008) |
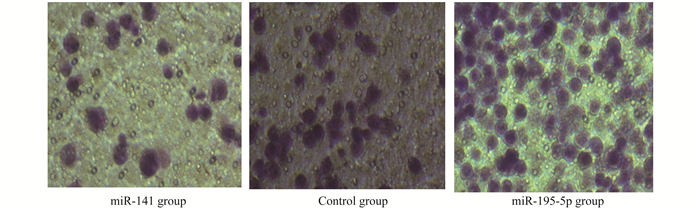
各组乳腺癌细胞在各时点相对增殖能力和迁移能力的比较 乳腺癌细胞转染miR-141和miR-195-5p质粒后, 与对照组比较, 随着培养时间的延长, miR-141组增殖能力明显升高, 而miR-195-5p组增殖能力明显降低(表 4)。各组培养24 h后, 通过Transwell迁移实验在200倍光镜下(图 3)发现, 对照组下室细胞数为(76.37±10.55)个, miR-141组下室细胞数为(138.34±15.67)个, miR-195-5p组下室细胞数为(65.46±9.42)个, miR-141组细胞迁移能力明显高于对照组(P < 0.01), 而miR-195-5p组迁移能力明显低于对照组(P < 0.01)。
| (x±s) | |||
| Groups | 24 h | 48 h | 72 h |
| Control | 1.68±0.35 | 3.16±0.36 | 6.75±0.53 |
| miR-141 | 1.83±0.28 | 3.95±0.42 | 7.89±0.68 |
| miR-195-5p | 1.43±0.22 | 2.34±0.32 | 4.64±0.46 |
| F | 2.502 | 23.542 | 43.382 |
| P | 0.130 | < 0.001 | < 0.001 |
|
| 图 3 细胞培养24 h后上室细胞的光镜观察(×200) Fig 3 Light microscopic observation of the upper chamber cells after 24 h of cell culture (×200) |
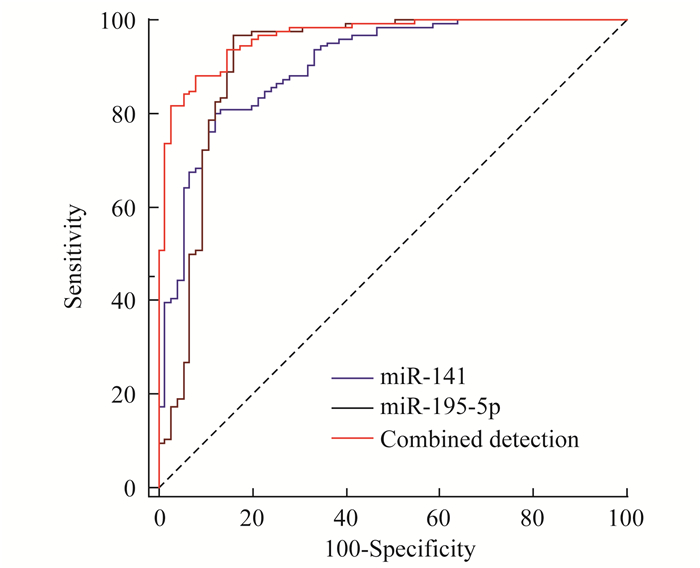
乳腺良性病变与乳腺癌的ROC曲线分析 以血清miR-141 (x1)和miR-195-5p (x2)水平根据乳腺良性疾病是否合并乳腺癌进行二元回归得出方程:y=1.093x1-1.886x2-0.245, 代入方程后得出联合检测(miR-141+miR-195-5p)变量。以联合检测作为检验变量, 以发生乳腺癌为状态变量绘制ROC曲线(图 4)。联合检测诊断乳腺癌的灵敏度为88.1%, 特异性为92.0%, AUC为0.964。联合检测的AUC明显优于miR-141(Z=3.413, P < 0.01)和miR-195-5p(Z=3.426, P < 0.01)单独检测, 而血清miR-141和miR-195-5p的AUC比较差异无统计学意义(表 5)。
|
| 图 4 miR-141和miR-195-5p在诊断乳腺癌的ROC曲线分析 Fig 4 ROC curve analysis of miR-141 and miR-195-5p in the diagnosis of breast cancer |
| Groups | Optimal cut-off (fmol/L) | Sensitivity (%) | Specificity (%) | AUC | 95%CI |
| miR-141 | > 6.664 | 80.2 | 88.0 | 0.904 | 0.855-0.941 |
| miR-195-5p | ≤3.525 | 96.8 | 84.0 | 0.912 | 0.865-0.948 |
| Combined detection | - | 88.1 | 92.0 | 0.964 | 0.928-0.985 |
miRNAs是一类小分子、非编码调控的RNAs, 由20~24个核苷酸组成, 在肿瘤的发生发展的过程中具有重要作用, 可能是肿瘤早期诊断、治疗和评估预后的重要标记物[4-5]。miRNAs在肿瘤中稳定表达, 并且在血液中具有较高的稳定性和特异性, 有无创和重复检测等特点[6], 因此血清miRNAs有望成为无创早期诊断肿瘤的新型肿瘤标记物。
本研究显示乳腺癌患者血清miR-141水平明显高于健康对照组和乳腺良性病变组, 并且发现手术后乳腺癌患者血清miR-141水平明显降低。同时发现, 乳腺癌患者血清miR-141表达水平与肿瘤直径和年龄无关系, 而与淋巴结转移、ER、PR、HER-2、TNM分期、分子分型和组织分型具有明显相关性; 通过多元回归分析发现, miR-141表达水平与淋巴结转移、PR、TNM分期和分子分型呈线性相关性, 说明血清miR-141水平能够反映乳腺的恶性程度, 与乳腺癌的预后有一定的相关性。miR-141在不同肿瘤中表达水平不同, 且表达方式也有差异, 与正常组织相比, 胃癌组织中miR-141表达水平明显降低, 并且与肿瘤分期呈负相关[7]; 在肝癌细胞中表达明显下降, 可抑制肝癌细胞的侵袭和转移[8]; 而在结肠癌患者中血清miR-141呈高表达[9], 并且认为血清中miR-141高表达说明结肠癌预后不良。在一项乳腺癌组织和乳腺正常标本中进行的miRNA表达谱分析发现, 包括miR-141基因在内共有7个基因表达上调和2个基因表达下调, 联合上述基因检测能够提高乳腺癌诊断的准确性[10]。本研究发现将miR-141质粒转染到乳腺癌后, 促进肿瘤细胞的增殖和迁移, 与文献报道的结果一致[11], 说明miR-141对乳腺癌细胞生长和转移具有重要临床意义, 推测miR-141可能是乳腺癌发生、发展和预后的重要指标。本研究还发现对于乳腺良性肿瘤鉴别乳腺癌, 当血清miR-141>6.664 fmol/L时, 灵敏度为80.2%, 特异性为88.0%, AUC为0.904, 对乳腺癌的诊断具有较高的特异性和灵敏度, 可能成为效能较高的诊断指标。
本研究发现乳腺癌患者血清miR-195-5p表达水平明显低于乳腺良性病变组和健康对照组, 且乳腺癌患者术后血清miR-195-5p水平较治疗前明显升高, 说明miR-195-5p是乳腺癌的保护因子。用miR-195-5p质粒转染乳腺癌细胞后, 发现miR-195-5p具有促进乳腺癌细胞增殖和迁移的作用, 说明miR-195-5p是反映乳腺癌生物学特性的指标。这一研究结果与文献报道相一致:肿瘤细胞miR-195-5p呈低表达, 上调miR-195-5p表达能够明显抑制细胞增殖[12-13]。本研究还发现乳腺癌患者血清miR-195-5p表达水平与年龄和肿瘤直径无相关性, 而与淋巴转移、肿瘤分期及肿瘤分级呈负相关, 与ER、PR和HER-2表达阳性呈正相关, 通过多元回归分析发现miR-195-5p表达水平与淋巴结转移、PR、TNM分期和分子分型呈线性相关, 说明miR-195-5p基因可能与抑癌基因类似。研究显示miR-195-5p在多种肿瘤中呈低表达:miR-195-5p能够抑制口腔鳞状细胞癌的侵袭和转移, 抑制肿瘤的生长[14-15]; miR-195-5p能够通过抑制血管内皮细胞因子的表达, 降低肿瘤细胞的侵袭和迁移[16]。
本研究发现在诊断乳腺良性疾病和乳腺癌方面, 患者血清miR-195-5p水平临界值为3.525 fmol/L, 当小于该临界值诊断乳腺癌的灵敏度为96.8%, 特异性为84.0%, AUC为0.912, 说明miR-195-5p诊断乳腺癌具有较高的特异性和灵敏度。
本研究发现血清miR-141和miR-195-5p联合检测诊断乳腺癌的灵敏度为88.1%, 特异性为92.0%, AUC为0.964, 联合检测的AUC明显优于miR-141和miR-195-5p单独检测, 而血清miR-141和miR-195-5p的AUC差异无统计学意义, 说明血清miR-141水平与miR-195-5p水平之间存在某种平衡关系, miR-141相当于促癌基因, 而miR-195-5p相当于抑癌基因。miR-141和miR-195-5p的来源仍不确定, 目前认为主要来自凋亡或坏死细胞, 细胞破坏后释放入血, 是反映乳腺癌细胞增殖和迁移的指标, 也是乳腺癌预后的指标, 同时可能是诊断乳腺癌的早期标记物。
综上所述, miR-141和miR-195-5p与乳腺癌细胞增殖和迁移具有密切联系, 对诊断乳腺癌具有重要的临床意义。本研究未与经典的钼靶、超声和MRI等影像学方法在诊断乳腺癌的效能方面进行比较, 需要在将来的研究中予以进一步阐明。
| [1] |
MILOSEVIC M, JANKOVIC D, MILENKOVIC A, et al. Early diagnosis and detection of breast cancer[J]. Technol Health Care, 2018, 26(4): 729-759.
[DOI]
|
| [2] |
BHARALI D, JEBUR HB, BAISHYA D, et al. Expression analysis of serum microRNA-34a and microRNA-183 in hepatocellular carcinoma[J]. Asian Pac J Cancer Prev, 2018, 19(9): 2561-2568.
|
| [3] |
AISO T, OHTSUKA K, UEDA M, et al. Serum levels of candidate microRNA diagnostic markers differ among the stages of non-small-cell lung cancer[J]. Oncol Lett, 2018, 16(5): 6643-6651.
|
| [4] |
ZHOU Y, WANG X, ZHANG Y, et al. Circulating microRNA profile as a potential predictive biomarker for early diagnosis of spontaneous abortion in patients with subclinical hypothyroidism[J]. Front Endocrinol (Lausanne), 2018, 9: 128.
[DOI]
|
| [5] |
XIN L, GAO J, WANG D, et al. Novel blood-based microRNA biomarker panel for early diagnosis of chronic pancreatitis[J]. Sci Rep, 2017, 7: 40019.
[DOI]
|
| [6] |
LIU SS, CHAN K, CHU D, et al. Oncogenic microRNA signature for early diagnosis of cervical intraepithelial neoplasia and cancer[J]. Mol Oncol, 2018, 12(12): 2009-2022.
[DOI]
|
| [7] |
SHA M, LIN M, WANG J, et al. Long non-coding RNA MIAT promotes gastric cancer growth and metastasis through regulation of miR-141/DDX5 pathway[J]. J Exp Clin Cancer Res, 2018, 37(1): 58.
[DOI]
|
| [8] |
KIM MK, MOON YA, SONGC K, et al. Tumor-suppressing miR-141 gene complex-loaded tissue-adhesive glue for the locoregional treatment of hepatocellular carcinoma[J]. Theranostics, 2018, 8(14): 3891-3901.
[DOI]
|
| [9] |
DING L, YU LL, HAN N, et al. miR-141 promotes colon cancer cell proliferation by inhibiting MAP2K4[J]. Oncol Lett, 2017, 13(3): 1665-1671.
[DOI]
|
| [10] |
XIONG DD, LV J, WEI KL, et al. A nine-miRNA signature as a potential diagnostic marker for breast carcinoma:an integrated study of 1110 cases[J]. Oncol Rep, 2017, 37(6): 3297-3304.
[DOI]
|
| [11] |
ANTOLIN S, CALVO L, BLANCO-CALVOM, et al. Circulating miR-200c and miR-141 and outcomes in patients with breast cancer[J]. BMC Cancer, 2015, 15: 297.
[DOI]
|
| [12] |
FENG C, ZHANG L, SUN Y, et al. GDPD5, a target of miR-195-5p, is associated with metastasis and chemoresistance in colorectal cancer[J]. Biomed Pharmacother, 2018, 101: 945-952.
[DOI]
|
| [13] |
LI M, REN CX, ZHANG JM, et al. The effects of miR-195-5p/MMP14 on proliferation and invasion of cervical carcinoma cells through tnf signaling pathway based on bioinformatics analysis of microarray profiling[J]. Cell Physiol Biochem, 2018, 50(4): 1398-1413.
[DOI]
|
| [14] |
ZHOU S, YU L, XIONG M, et al. LncRNA SNHG12 promotes tumorigenesis and metastasis in osteosarcoma by upregulating Notch2 by sponging miR-195-5p[J]. Biochem Biophys Res Commun, 2018, 495(2): 1822-1832.
[DOI]
|
| [15] |
WANG T, REN Y, LIU R, et al. miR-195-5p suppresses the proliferation, migration, and invasion of oral squamous cell carcinoma by targeting TRIM14[J]. Biomed Res Int, 2017, 2017: 7378148.
[PubMed]
|
| [16] |
JIN Y, WANG M, HU H, et al. Overcoming stemness and chemoresistance in colorectal cancer through miR-195-5p-modulated inhibition of notch signaling[J]. Int J Biol Macromol, 2018, 117: 445-453.
[DOI]
|
 2019, Vol. 46
2019, Vol. 46


