2. 复旦大学附属中山医院心脏超声诊断科 上海 200032
2. Department of Echocardiography, Zhongshan Hospital, Fudan University, Shanghai 200032, China
心肌肥大是一种较常见的原发性心脏病, 主要特点是心肌细胞体积增大及细胞内蛋白质合成增加, 这也是多种心血管类疾病共有的病理过程[1]。心肌肥大本质上是一种代偿性反应, 往往源于心脏超负荷, 临床经常表现为左室的异常肥大, 室壁增厚, 心室腔缩小, 可能发展为进行性心衰、致死性心律失常[2], 甚至是心源性猝死[3-4]。
去甲肾上腺素(norepinephrine, NE)作为一种刺激因子可以诱导心肌细胞肥大[5], 哌唑嗪(prazosin, PRA)对此具有一定的拮抗效果[6-7]。研究表明, NE可以通过心脏的肾上腺素能受体(adrenoceptor, AR)α和β[8], 进一步激活细胞内PLC、PKC、MAPK等信号通路, 进而使得心肌细胞的结构基因(如细胞骨架蛋白基因myosin、actin、titin等)过度或异常表达[9-11]。
目前临床上仍缺乏治疗心肌肥大的特效药物。β-AR是一种比较理想的药物靶点, 但是关于α受体的研究相对缺乏。由于人类心肌细胞在体外无法扩增, 来源稀少, 人类心脏疾病的体外细胞模型建立及大规模药物筛选一直无法开展。随着胚胎干细胞体外中胚层定向分化的技术成熟, 可大量获得人源性心肌细胞, 这为人类心血管疾病的研究与药物筛选提供了有力的工具[12-13]。本研究在人类胚胎干细胞(human embryonic stem cells, hESCs)的基础上大量获取人源性心肌细胞(hESCs-cardiomyocytes, hESCs-CMs), 进一步探索α受体拮抗剂PRA对NE诱导的心肌细胞肥大的影响, 为今后运用hESCs-CMs进行人类心肌肥大的建模及大规模药物筛选提供一定的实验基础。
材料和方法主要试剂与仪器 哌唑嗪盐酸盐(美国Sigma Aldrich公司), 重酒石酸去甲肾上腺素注射液(上海禾丰制药有限公司), DMEM/F12培养液、RPMI 1640培养液、DMEM培养液(美国Corning公司), mTeSR培养基(加拿大STEMCELL公司)、FBS、0.25%无EDTA胰酶(Trypsin)、B27添加剂、B27不含胰岛素添加剂、鼠来源的肌钙蛋白T(cardia troponin T, TNNT2)单克隆抗体, Alexa Fluor 488荧光标记的山羊抗小鼠IgG二抗、Trizol(美国Thermo Fisher公司), 兔来源的肌球蛋白轻链2(myosin light chain 2v, MLC2v)多克隆抗体、PE荧光标记的抗兔IgG二抗、细胞流式试剂盒、基质胶(美国BD公司), Ⅰ型胶原酶、IWR-1、DAPI(美国Sigma Aldrich公司), CHIR-99021(美国Selleckchem公司), 防荧光淬灭封片剂(美国Vector公司), Triton-X100 (北京鼎国公司), 山羊血清封闭工作液、4%多聚甲醛固定液(中国谷歌生物公司), 逆转录试剂、SYBR GREEN Realtime PCR Master Mix (日本TOYOBO公司), 荧光倒置显微镜(德国Leica公司), 实时定量PCR仪(瑞士Roche公司), 12孔细胞培养板、激光共聚焦皿(无锡耐思生物科技有限公司), Leica显微镜FelixGX细胞动缘检测系统, 流式细胞仪(美国BD公司)。
hESC系H7的培养及心肌的定向分化 将hESCs-H7用mTeSR培养液培养在6孔板中(包被有基质胶), 待细胞生长至密度为75%~85%, 将培养液替换为含有10 μmol/L CHIR99021的B27无胰岛素/RPMI 1640培养液, 48 h后去除CHIR99021, 替换为新的B27无胰岛素/RPMI 1640培养液, 24 h后换成含有5 μmol/L IWR-1的B27无胰岛素/RPMI 1640培养液, 48 h后去除IWR-1, 替换为新的B27无胰岛素/RPMI 1640培养液, 48 h后换为B27含胰岛素/RPMI 1640培养液, 隔天换液, 分化至8~10天可于显微镜下看见自主搏动的心肌细胞, 继续培养以进行后续实验。
心肌细胞分化效率的流式检测 取分化至25天的心肌细胞, 先用胶原酶Ⅰ于37 ℃消化30 min, DPBS洗去剩余的胶原酶Ⅰ后即加入0.25%不含EDTA的胰酶进行消化, 尽量将心肌细胞消化为单细胞。367×g离心2 min, 收集细胞沉淀, 使用试剂盒内固定液将收集的心肌细胞于4 ℃固定30 min, 离心(同上)弃上清, 使用1×BD洗涤缓冲液清洗细胞沉淀2次, 再用洗涤缓冲液将细胞重悬, 分为3组, 即空白对照、二抗阴性对照(只加二抗1:200稀释)和正常实验组(先加一抗1:100稀释, 后加二抗), 每组细胞密度约为1×106个/mL。一抗4 ℃孵育1 h, 洗去后加入二抗, 4 ℃避光孵育30 min, 洗去后PBS重悬细胞, 上机检测。
心肌细胞免疫荧光染色 取分化至25天的心肌细胞, 尽量消化为单细胞, 并接种在铺有无菌玻片的12孔板中, 使用10%FBS/DMEM培养液培养, 接种24 h后加入相应的药物处理, 培养5天, 每2天换液1次, 以保证心肌细胞完全铺展开。接种的心肌细胞分为3组(每组设4个复孔):对照组不含NE和Prazosin; NE组只加入20 μmol/L NE处理5天; PRA组, 先加入15 μmol/L PRA处理3 h, 再加入20 μmol/L NE共同处理5天。将3组细胞用4%多聚甲醛固定后, 染cTNT、荧光二抗及DAPI, 使用荧光显微镜进行拍摄, 计算每个视野的细胞个数并进行统计分析, 所有视野均随机采集。利用imageJ软件分析细胞大小形态, 首先测量采集图像右下角标尺的像素距离, 并修改相应的比例尺, 使用不规则图形工具圈出细胞轮廓进行测量分析, 同一条件下计算心肌细胞的相对面积并进行比较。
qRT-PCR检测心肌细胞肥大相关基因 心肌细胞消化后接种于铺有基质胶的12孔板中, 分组同上, 药物处理5天后收集细胞, 并用Trizol裂解细胞, 提取mRNA, 测定浓度, 反转录为cDNA进行PCR反应。以GAPDH作为内参基因, 引物序列参见表 1。
| Gene | Forward (5’-3’) | Reverse (5’-3’) |
| GAPDH | GGAGCGAGATCCCTCCAAAAT | GGCTGTTGTCATACTTCTCATGG |
| BNP | TTCCTGGGAGGTCGTTCCCAC | CATCTTCCTCCCAAAGCAGCC |
| TNNT2 | AAGAAGAAGATTCTGGCTGAGAG | ACTTTCTGGTTATCGTTGATCCT |
| Serca2 | CGAACCCTTGCCACTCATCT | CCAGTATTGCAGGTTCCAGGT |
| Mef2c | GATGCCATCAGTGAATCAAAGG | GTTGAAATGGCTGATGGATATCC |
| MYH6 | TCAGCTGGAGGCCAAAAGTAAAGGA | TTCTTGAGCTCTGAGCACTCGTCT |
| MYH7 | TCGTGCCTGATGACAAACAGGAGT | ATACTCGGTCTCGGCAGTGACTTT |
心肌细胞收缩力检测 心肌细胞消化为单细胞后接种于铺有基质胶的激光共聚焦皿中, 分组同上, 接种48 h后加入药物再处理24 h, 用FelixGX细胞动缘检测系统测定并分析心肌细胞的收缩功能及跳动情况。
统计学处理 采用Graphpad Prism6进行数据处理和统计分析, 多组比较采用One-way ANOVA分析, 两组间比较采用t检验, P < 0.05为差异有统计学意义。所有实验结果均以x±s表示, 实验重复3次。
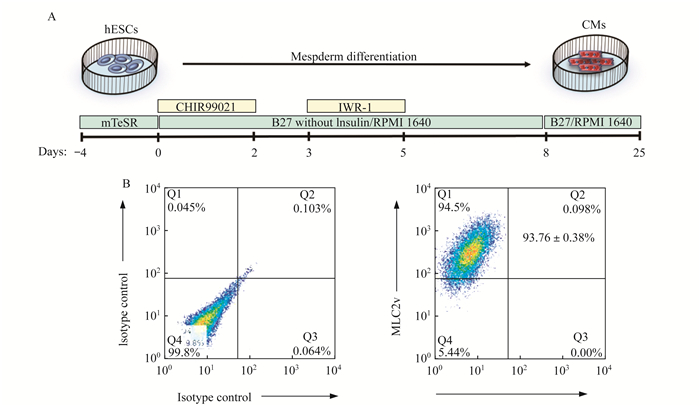
结果心肌细胞定向分化及分化效率检测 按图 1A描述的步骤对hESCs-H7进行中胚层定向诱导分化, 成功得到hESCs-CMs。收集分化至30天的心肌细胞进行流式检测, 检测指标为心室肌细胞所特有表达的蛋白—肌球蛋白轻链2(MLC2v), 结果发现MLC2v阳性细胞可达到90 %以上(图 1B), 说明我们所建立的分化体系相对成熟, 所分化出来的心肌细胞纯度较高, 绝大部分为心脏工作细胞——心室肌细胞。
|
| A:Schematic diagram of differentiation of hESCs to CMs.B:Representativeflow cytometry assay of MLC2v expression in CMs at D30 after differentiation.Over 90% cells were MLC2v positve CMs (n=5, 93.76%±0.38%).Isotype control was used as a negative control to set the gating parameters. 图 1 hESCs-CMs定向分化与流式检测 Fig 1 Differentiation and flow cytometry assay of hESCs-CMs |
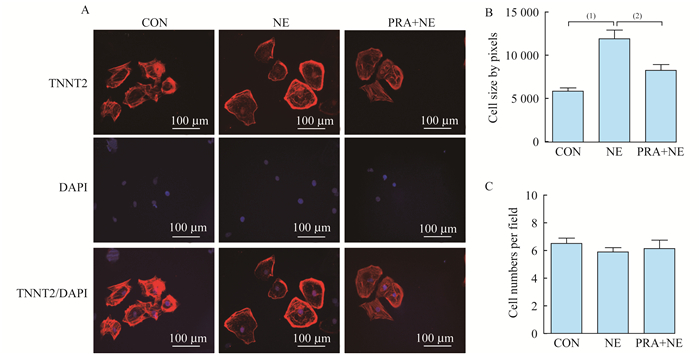
PRA与NE对心肌细胞面积的影响 对分化25天的心肌细胞用药物处理5天, 进行免疫荧光染色(图 2A), 用image J软件对细胞面积进行统计分析发现:与对照组相比, NE组心肌细胞面积有明显增大的趋势(P < 0.000 1);与NE组相比, PRA组心肌细胞面积减少(P < 0.01), 然而并未完全恢复到对照组水平, 说明PRA可以在一定程度上抑制NE诱导的心肌细胞面积增大(图 2B)。我们还统计了每个视野的细胞个数, 分析发现NE与PRA处理均未引起心肌细胞数目变化(图 2C)。
|
| A:Representative immunofluorescence staining of TNNT2 and DAPI at D30 after differentiation (scale bar:100 μm).CON:PBS; NE:20 μmol/L NE; PRA+NE:15 μmol/L PRA+20 μmol/L NE.B:Quantification of cell size by pixels (n>100);C:Quantification of cell numbers per field (n>30). (1) P < 0.000 1;(2) P < 0.01. 图 2 PRA处理前后hESCs-CMs的免疫荧光染色(×200) Fig 2 Immunofluorescence staining of hESCs-CMs before and after PRA treatment (×200) |
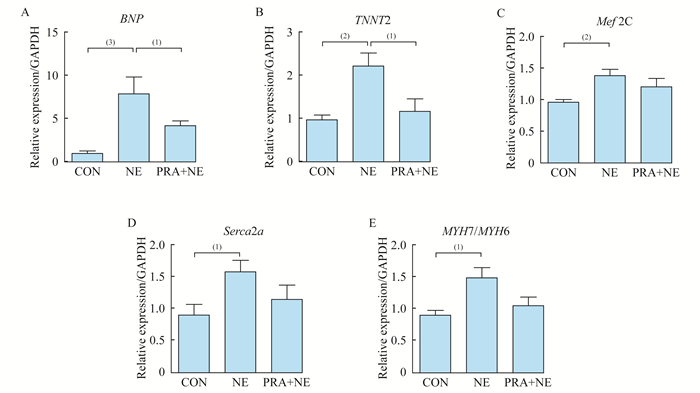
PRA与NE对心肌细胞肥大相关基因mRNA表达的影响 利用qRT-PCR方法检测PRA和NE处理5天后的心肌细胞肥大相关基因mRNA表达水平的变化, 我们发现, NE处理组肥大相关的标志B型钠尿肽(brain natriuretic peptide, BNP)较对照组表达明显增高(P < 0.001), 肌小节蛋白相关基因TNNT2、肌细胞增强因子2C(myocyte-specific enhancer factor 2c, Mef2C)、肌浆网Ca2+ATP酶2a (sarcoplasmic reticulum Ca+-ATPase 2a, Serca2a)及肌球蛋白重链7(myosin heavy cgain 7, MYH7)与MYH6比值表达均上升(P分别 < 0.01和 < 0.05);与NE处理组相比, 加入PRA后, BNP和TNNT2表达均下调(P < 0.05), Serca2a和MYH7/MYH6比值有下降趋势。这表明, PRA可以抑制NE诱导的hESCs-CMs肥大相关基因BNP、TNNT2表达的上调(图 3)。
|
| Cardiac hypertrophic-related genes’ mRNA levels in hESCs-CMs were analyzed by qRT-PCR, and normalized to GAPDH expression.CON:PBS; NE:20 μmol/L NE; PRA+NE:15 μmol/L PRA+20 μmol/L NE. (1)P < 0.05;(2)P < 0.01;(3)P < 0.001. 图 3 PRA处理前后心肌肥大相关基因表达 Fig 3 The expression of cardiac hypertrophy-associated genes before and after PRA treatment |
PRA与NE对心肌细胞收缩力及跳动频率的影响 我们检测了分化约30天的心肌细胞在药物处理24 h后的细胞收缩情况, NE处理后可以产生类似正性肌力的作用, 可明显提高心肌细胞的收缩力及搏动频率(P < 0.000 1);而PRA处理后, 心肌细胞的收缩强度及跳动频率均有所减弱(P < 0.000 1)。这表明PRA可以抑制NE引起的心肌细胞收缩力及跳动频率的增加(图 4)。

|
| A:Representative images showing the contraction force measuredby FelixGX detection system.B:Quantification of contractility in cardiomyocytes 24 h after PRA treatment (n>50).C:Quantification of beating frequency 24 h after PRA treatment.CON:PBS; NE:20 μmol/L NE; PRA+NE:15 μmol/L PRA+20 μmol/L NE. (1) P < 0.000 1. 图 4 PRA处理前后单个hESC-CM收缩力及跳动频率检测 Fig 4 The contractility and beating frequency detection of the single hESC-CM before and after PRA treatment |
心肌肥大可分为生理性肥厚和病理性肥厚, 生理性肥厚常常见于孕妇、运动员等[14-15], 病理性肥大常常见于高血压、心梗、先心病等[16]。目前心肌肥大已是公认的导致猝死、心衰等心血管疾病的危险因素之一[17-18], 然而临床上尚无针对性治疗的药物, 因此探明发生机制、寻找药物靶点、开发特效药物已成为心血管领域的重要研究方向。
引起心肌肥大的因素通常分为3类:(1)机械性因素, 如压力负荷过度、容积超负荷等[19]; (2)神经体液内分泌性因素, 如加压素、血管紧张素Ⅱ、去甲肾上腺素等[20-21]; (3)遗传性因素, 如肌节蛋白突变引起的肥大型心肌病等[22]。NE是一种常用的刺激因子, 能导致心肌细胞肥大, 甚至是细胞的凋亡[23]。
在新生乳鼠的原代心肌细胞上, PRA可以抑制NE诱导的心肌肥大[24]。本研究中, 通过体外二维分化培养, 我们将hESCs-H7成功诱导分化为心肌细胞, 获得hESCs-CMs[25]。研究表明, hESCs-CMs无论是从基因表达的模式, 还是细胞电生理、收缩功能等多个方面, 都比鼠的心肌细胞更接近人类的心肌组织[26-28], 而我们在此基础上进行研究也更加接近于临床患者心肌肥大的治疗。我们发现NE可诱导hESCs-CMs肥大, 进一步肯定了NE促肥大的效果, 具体表现为心肌细胞面积增加, 肥大相关基因mRNA表达的上调, 心肌细胞收缩强度及跳动频率的增加, 而α受体拮抗剂PRA能够部分抑制细胞面积的增加, 使肥大相关基因BNP、TNNT2表达下调, 收缩力及跳动频率降低, 表明PRA能改善NE诱导的肥大效果。
心肌细胞分布有α受体和β受体, 研究表明这两种受体均参与心肌肥大的过程[29-31]。在心肌组织中, β受体表达相对较多, 包括β1和β2受体, 这些受体表达在心肌细胞膜的表面, 与心肌的收缩功能关系密切[32-33]。针对α受体, NE表现为强烈的兴奋作用, 而对β受体作用较弱, α受体的经典拮抗剂PRA能够抑制NE导致的收缩力增强, 表明心肌细胞的收缩功能在某种程度上也与α受体有关。
综上所述, 本研究表明PRA对hESCs-CMs可改善NE导致的心肌肥大, 为运用hESCs-CMs进行人类心肌肥大建模及大规模药物筛选提供一定的实验基础。而PRA发挥作用的具体机制以及α受体能否作为临床治疗心肌肥大的有效药物靶点还有待进一步的研究。
| [1] |
LORELL BH, CARABELLO BA. Left ventricular hypertrophy:pathogenesis, detection, and prognosis[J]. Circulation, 2000, 102(4): 470-479.
[DOI]
|
| [2] |
HUGHES SE. The pathology of hypertrophic cardiomyopathy[J]. Histopathology, 2004, 44(5): 412-427.
[DOI]
|
| [3] |
MARON BJ, MARON MS. Hypertrophic cardiomyopathy[J]. Lancet, 2013, 381(9862): 242-255.
[DOI]
|
| [4] |
CHEN YZ, QIAO SB, HU FH, et al. Left ventricular remodeling and fibrosis:sex differences and relationship with diastolic function in hypertrophic cardiomyopathy[J]. Eur J Radiol, 2015, 84(8): 1487-1492.
[DOI]
|
| [5] |
XIAO D, DASGUPTA C, CHEN M, et al. Inhibition of DNA methylation reverses norepinephrine-induced cardiac hypertrophy in rats[J]. Cardiovasc Res, 2014, 101(3): 373-382.
[DOI]
|
| [6] |
BRIEST W, HOMAGK L, RABLER B, et al. Norepinephrine-induced changes in cardiac transforming growth factor-β isoform expression pattern of female and male rats[J]. Hypertension, 2004, 44(4): 410-418.
[DOI]
|
| [7] |
LUO JD, XIE F, ZHANG WW, et al. Simvastatin inhibits noradrenaline-induced hypertrophy of cultured neonatal rat cardiomyocytes[J]. Br J Pharmacol, 2001, 132(1): 159-164.
[DOI]
|
| [8] |
AMIN JK, XIAO L, PIMENTAL DR, et al. Reactive oxygen species mediate alpha-adrenergic receptor-stimulated hypertrophy in adult rat ventricular myocytes[J]. J Mol Cell Cardiology, 2001, 33(1): 131-139.
[DOI]
|
| [9] |
MORISCO C, ZEBROWSKI D, CONDORELLI G, et al. The Akt-glycogen synthase kinase 3beta pathway regulates transcription of atrial natriuretic factor induced by beta-adrenergic receptor stimulation in cardiac myocytes[J]. J Biol Chem, 2000, 275(19): 14466-14475.
[DOI]
|
| [10] |
KEHAT I, DAVIS J, TIBURCY M, et al. Extracellular signal-regulated kinases 1 and 2 regulate the balance between eccentric and concentric cardiac growth[J]. Circ Res, 2011, 108(2): 176-183.
[DOI]
|
| [11] |
WANG X, FINEGAN KG, ROBINSON AC, et al. Activation of extracellular signal-regulated protein kinase 5 downregulates FasL upon osmotic stress[J]. Cell Death Differ, 2006, 13(12): 2099-2108.
[DOI]
|
| [12] |
MENASCHE P, VANNEAUX V, FABREGUETTES JR, et al. Towards a clinical use of human embryonic stem cell-derived cardiac progenitors:a translational experience[J]. Eur Heart J, 2015, 36(12): 743-750.
[DOI]
|
| [13] |
CHONG JJ, YANG X, DON CW, et al. Human embryonic-stem-cell-derived cardiomyocytes regenerate non-human primate hearts[J]. Nature, 2014, 510(7504): 273-277.
[DOI]
|
| [14] |
FAGARD RH. Impact of different sports and training on cardiac structure and function[J]. Cardiol Clin, 1997, 15(3): 397-412.
[DOI]
|
| [15] |
ZANATI BS, BORGES VM, MARTIN LC, et al. Disproportionate pregnancy-induced myocardial hypertrophy in women with essential hypertension[J]. Am J Hypertens, 2013, 26(6): 816-821.
[DOI]
|
| [16] |
SEGERS VF, De KEULENAER GW. Pathophysiology of diastolic dysfunction in chronic heart failure[J]. Future Cardiol, 2013, 9(5): 711-720.
[DOI]
|
| [17] |
YOU J, WU J, JIANG G, et al. Olmesartan attenuates cardiac remodeling through DLL4/Notch1 pathway activation in pressure overload mice[J]. J Cardiovasc Pharmacol, 2013, 61(2): 142-151.
[DOI]
|
| [18] |
DUAN Q, NI L, WANG P, et al. Deregulation of XBP1 expression contributes to myocardial vascular endothelial growth factor-a expression and angiogenesis during cardiac hypertrophy in vivo[J]. Aging Cell, 2016, 15(4): 625-633.
[DOI]
|
| [19] |
HUNTER JJ, CHIEN KR. Signaling pathways for cardiac hypertrophy and failure[J]. N Engl J Med, 1999, 341(17): 1276-1283.
[DOI]
|
| [20] |
PELLIEUX C, SAUTHIER T, AUBERT JF, et al. Angiotensin Ⅱ-induced cardiac hypertrophy is associated with different mitogen-activated protein kinase activation in normotensive and hypertensive mice[J]. J Hypertens, 2000, 18(9): 1307-1317.
[DOI]
|
| [21] |
YANG F, LIU Z, WANG Y, et al. Hydrogen sulfide endothelin-induced myocardial hypertrophy in rats and the mechanism involved[J]. Cell Biochem Biophys, 2014, 70(3): 1683-1686.
[DOI]
|
| [22] |
PASQUALE F, SYRRIS P, KASKI JP, et al. Long-term outcomes in hypertrophic cardiomyopathy caused by mutations in the cardiac troponin T gene[J]. Circ Cardiovasc Genet, 2012, 5(1): 10-17.
[DOI]
|
| [23] |
徐旖旎, 王羿, 柏帅, 等. 环维黄杨星D对高交感活性诱导大鼠实验性心肌损伤氧化应激及能量代谢的影响[J]. 中药材, 2014, 37(7): 1213-1217. [URI]
|
| [24] |
BISHOPRIC NH, SIMPSON PC, ORDAHL CP. Induction of the skeletal alpha-actin gene in alpha 1-adrenoceptor-mediated hypertrophy of rat cardiac myocytes[J]. J Clin Invest, 1987, 80(4): 1194-1199.
[DOI]
|
| [25] |
LIAN X, BAO X, ZILBERTER M, et al. Chemically defined, albumin-free human cardiomyocyte generation[J]. Nat Methods, 2015, 12(7): 595-596.
[DOI]
|
| [26] |
SHIBA Y, FERNANDES S, ZHU WZ, et al. Human ES-cell-derived cardiomyocytes electrically couple and suppress arrhythmias in injured hearts[J]. Nature, 2012, 489(7415): 322-325.
[DOI]
|
| [27] |
LUNDY SD, ZHU WZ, REGNIER M, et al. Structural and functional maturation of cardiomyocytes derived from human pluripotent stem cells[J]. Stem Cells Dev, 2013, 22(14): 1991-2002.
[DOI]
|
| [28] |
BLAZESKI A, ZHU R, HUNTER DW, et al. Electrophysiological and contractile function of cardiomyocytes derived from human embryonic stem cells[J]. Prog Biophys Mol Biol, 2012, 110(2-3): 178-195.
[DOI]
|
| [29] |
COTECCHIA S, DEL VC, COLELLA M, et al. The alpha1-adrenergic receptors in cardiac hypertrophy:signaling mechanisms and functional implications[J]. Cell Signal, 2015, 27(10): 1984-1993.
[DOI]
|
| [30] |
PAPAY RS, SHI T, PIASCIK MT, et al. alpha(1)A-adrenergic receptors regulate cardiac hypertrophy in vivo through interleukin-6 secretion[J]. Mol Pharmacol, 2013, 83(5): 939-948.
[DOI]
|
| [31] |
VIDAL M, WIELAND T, LOHSE MJ, et al. beta-Adrenergic receptor stimulation causes cardiac hypertrophy via a Gbetagamma/Erk-dependent pathway[J]. Cardiovasc Res, 2012, 96(2): 255-264.
[DOI]
|
| [32] |
COWLEY PM, WANG G, CHANG AN, et al. The alpha1A-adrenergic receptor subtype mediates increased contraction of failing right ventricular myocardium[J]. Am J Physiol Heart Circ Physiol, 2015, 309(5): H888-H896.
[URI]
|
| [33] |
FU Q, KIM S, SOTO D, et al. A long lasting beta1 adrenergic receptor stimulation of cAMP/protein kinase A (PKA) signal in cardiac myocytes[J]. J Biol Chem, 2014, 289(21): 14771-14781.
[DOI]
|
 2019, Vol. 46
2019, Vol. 46


