2. 复旦大学附属中山医院呼吸科 上海 200032
2. Department of Respiratory Disease, Zhongshan Hospital, Fudan University, Shanghai 200032, China
淋巴管平滑肌瘤病(lymphangioleiomyomatosis, LAM)是一种罕见的肺部疾病, 主要发生在育龄期女性, 它可以散发或者与结节性硬化症(tuberous sclerosis complex, TSC)有关[1-2]。据最新统计LAM的发病率大约为3.35~7.76例/百万女性[3], 男性和儿童中罕有LAM的报道[4-6]。LAM的临床表现主要有进展性呼吸困难、复发性气胸、乳糜胸和咯血, 预后较差[7]。肺外表现有肾血管平滑肌脂肪瘤(renal angiomyolipoma, AML)和淋巴管平滑肌瘤(表现为淋巴结肿大和中轴淋巴管囊性肿块)[8]。LAM在胸部高分辨CT(high-resolution computed tomography, HRCT)上特征性表现为均匀分布的多发囊性病变, 囊泡的外形、大小、轮廓变化较大, 直径多在2~5 mm, 偶尔可以达到30 mm [9-10]。
LAM的诊断需要组织活检和/或结合病史与胸部HRCT。病理诊断依赖于特征性LAM细胞形态、免疫组化平滑肌肌动蛋白(smooth muscle actin, SAM)和人类黑色素瘤45抗体(HMB45)阳性。如无病理活检, 典型的临床和影像学表现并且伴有以下一项时也可以确诊, 如AML、乳糜胸或乳糜腹、淋巴管平滑肌瘤或淋巴结LAM受累和TSC[11]。由于LAM特征性的影像学表现, 有经验的临床医师依靠HRCT诊断LAM具有较高的准确性, 但是仅依靠HRCT作出临床诊断是不准确的[12]。许多疾病如肺朗格汉斯细胞增多症(pulmonary Langerhans cell histiocytosis, PLCH)、淋巴细胞间质性肺炎(lymphocytic interstitial pneumonia, LIP)、肺气肿、结缔组织病(Connective tissue diseases, CTD)、干燥综合征(Sjögren′s syndrome, SS)、系统性红斑狼疮(systemic lupus erythematosus, SLE)、结节病和Birt-Hogg Dubé(BHD)综合征等在影像上也有多发性囊泡改变, 需要与LAM进行鉴别诊断。
血管内皮生长因子-D(serum vascular endothelial growth factor-D, VEGF-D)是血管内皮生长因子家族的一员, 其在淋巴管内皮细胞增殖、迁移和诱导淋巴管形成中发挥重要作用[11]。最近国内外研究发现LAM患者中血清VEGF-D水平显著升高, 并提出将VEGF-D作为诊断LAM的生物标志物[13-16], 本研究将进一步探讨血清VEGF-D水平在LAM中的表达及其在PLD鉴别诊断中的意义。
资料和方法研究对象 选取2012年1月到2015年12月在复旦大学附属中山医院就诊的PLD患者。入选标准:胸部HRCT表现为双侧肺野广泛、多发囊泡性改变。LAM的诊断采用2010年欧洲呼吸协会制定的诊断标准[17]。健康志愿者为同期健康女性, 该研究经过复旦大学附属中山医院伦理委员会批准, 入选者自愿参加并签署知情同意书。
实验方法 患者入选后详细记录人口统计学资料、影像学检查资料、肺功能检查和病理检查结果。
胸部HRCT 采用GE Light Speed CVT 64层螺旋CT扫描机, 扫描方式:从肺尖覆盖至肺底, 扫描参数:120 kV, 150 mAs, 层厚5 mm, 层距5 mm, 运用HRCT薄层重建, 层厚1 mm。CT图像肺窗窗宽1 000~1 500 HU, 窗位-500 ~-400 HU, 纵隔窗窗宽350 HU, 窗位40 HU。每份HRCT结果均由复旦大学附属中山医院影像科2位医师评估。入选的患者均在本院进行至少1次HRCT检查。
血清VEGF-D检测 患者初次就诊时采集静脉血3 mL。采用VEGF-D检测试剂盒(美国R&D Systems公司), 通过酶联免疫吸附试验检测患者和健康志愿者的血清VEGF-D水平。
肺功能检查 使用Type Masterscreen-PFT Jaeger (德国Hoechberg公司)肺功能仪进行检测。由复旦大学附属中山医院呼吸科技术人员负责操作。主要检测指标包括:第1秒用力肺活量(forced expiratory volume in 1 second, FEV1), 第1秒用力肺活量/预测值(FEV1%pred)和第1秒用力肺活量/用力肺活量(FEV1/FVC)。单次呼吸法检测肺弥散功能指标一氧化碳弥散率/肺容积(DLco/VA%)。
统计学处理 采用SPSS 17.0统计分析软件, 符合正态分布的计量资料以x±s表示, 非正态分布计量资料采用区间R表示。使用ROC曲线选择敏感性和特异性都达到最大时的最佳临界点。P < 0.05为差异有统计学意义。
结果一般资料 2012年1月到2015年12月本研究中心共入组确诊的LAM患者50例, OPLD患者34例, 健康志愿者20例。
50例LAM均为确诊患者, 其中33例通过病理确诊, 23例为肺活检, 5例为腹膜后肿物活检, 5例为AML活检。21例伴有乳糜胸, 2例伴有乳糜腹, 17例伴有AML, 3例伴有TSC。LAM患者首发症状年龄为19~60岁, 平均(39.3±8.7)岁。34例OPLD患者HRCT表现为多发囊泡样改变(囊泡改变为非LAM的典型表现), 其中1例为BHD, 1例为结节病, 4例为SS, 2例为SLE, 1例为类风湿性关节炎, 1例为抗JO-1抗体综合征, 2例为肺气肿, 余诊断尚不明确, 首发症状年龄为30~69岁, 平均(46.9±9.6)岁。20例健康对照者年龄为24~49岁, 平均(33.5±5.8)岁。3组的年龄差异无统计学意义。LAM组患者表现为阻塞性通气功能障碍和弥散功能障碍, 对照组和OPLD组通气功能和弥散功能基本正常(表 1)。
| (x±s) | |||
| Characteristics | Control | LAM | OPLD |
| Male/Female (n) | 0/20 | 0/50 | 2/34 |
| Age (y) | 33.5±5.8 | 39.3±8.7 | 46.9±9.6 |
| Chylothorax (n) | 0 | 21 | 0 |
| Chyloperitoneu (n) | 0 | 2 | 0 |
| AML (n) | 0 | 19 | 0 |
| VEGF-D (pg/mL) | 413.4±33.2 | 3 986.1±361.2 | 505.7±25.6 |
| FEV1 (L) | 2.52±0.15 | 1.64±0.13 | 2.27±0.19 |
| FEV1% pred (%) | 99.6±6.5 | 63.6±4.7 | 93.10±6.1 |
| FEV1/FVC (%) | 80.1±6.1 | 62.9±3.1 | 80.50±1.7 |
| DLco/VA% (%) | 98.7±6.5 | 53.8±4.8 | 96.1±5.7 |
3组血清VEGF-D水平比较 LAM组、健康对照组和OPLD组的血清VEGF-D水平分别为(3 986.1±361.2)、(413.4±33.2)和(505.7±25.6) pg/mL, LAM组的血清VEGF-D水平较健康对照组有显著升高, 差异有统计学意义(P < 0.001, 图 1A)。ROC曲线的AUC为0.995, 最佳临界值为692.5 pg/mL, 其敏感性和特异性分别为98%和100% (图 1B)。血清VEGF-D水平具有较高的敏感性和特异性, 因此可以将692.5 pg/mL作为诊断LAM的血清学指标。LAM组血清VEGF-D水平较OPLD组显著提高, 差异有统计学意义(P<0.001)。ROC曲线的AUC为0.991, 最佳临界值为901.0 pg/mL, 其敏感性和特异性分别为94%和100% (图 1C)。OPLD患者和健康对照者相比, 血清VEGF-D水平升高不明显(P=0.036)。

|
| A:Comparation of serum VEGF-D among control group, OPLD group and LAM group; B:ROC curve of LAM group to control group; C:ROC curve of LAM group to OPLD group. 图 1 3组患者的血清VEGF-D水平及ROC曲线 Fig 1 Serum VEGF-D and the ROC curve of the three groups |
PLD的影像学表现 LAM的HRCT的特征性改变是均匀分布的多个(>10)薄壁圆形界限清楚的含气囊性病变, 并且没有其他显著肺部受累, 如肺间质病变。OPLD的HRCT不典型, 表现为两肺分布的薄壁囊腔, 囊泡直径变化较大, 形状各不相同, 可为圆形、椭圆形和不规则形状(图 2)。41岁女性, 因反复咳嗽伴胸闷、气促就诊, 胸部CT提示两肺均匀分布多发、薄壁、圆形、界限清楚的囊腔, 胸腔镜肺活检病理明确诊断为LAM, 该患者血清VEGF-D水平为5 556 pg/mL (图 2A)。38岁女性, 因活动后气喘就诊, 胸部CT提示两肺均匀分布多发、薄壁、圆形、界限清楚的囊腔, 肺活检病理确诊为LAM, 该患者血清VEGF-D水平为4 562 pg/mL (图 2B)。46岁女性, 因反复气胸就诊, 通过基因检测诊断为BHD。该患者的胸部CT提示两肺分布薄壁囊腔, 囊泡直径变化较大, 形状各不相同, 可为圆形、椭圆形和不规则形状, 仍见气胸存在, 该患者血清VEGF-D水平为537 pg/mL (图 2C)。64岁女性, 因反复咳嗽伴眼干和口干就诊, 根据自身免疫指标检查、眼科检查和唇腺活检, 诊断为SS, 其胸部CT可见大小不等的囊腔, 形状不规则, 该患者血清VEGF-D水平为525 pg/mL (图 2D)。
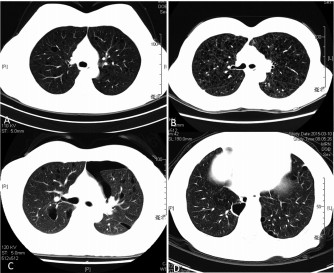
|
| A:A 41-year-old woman with repeated cough and shortness of breath was diagnosed as LAM.Chest CT showed multiple thin-walled circular cysts.B:A 38-year-old woman was diagnosed as LAM because of shortness of breath after exercise.Chest CT showed multiple thin-walled circular cysts with uniform distribution of the lungs.C:A 46-year-old woman diagnosed with BHD by genetic testing for repeated pneumothorax.Chest CT showed thin-walled cysts in both lungs.The diameter of the vesicles varied greatly with different shapes. It could be round, elliptical or irregular.D:A 64-year-old woman was diagnosed as dry syndrome.Chest CT showed different sizes and irregular shape of cysts. 图 2 LAM和OPLD的影像学表现 Fig 2 Imaging findings of LAM和OPLD |
LAM是一种罕见疾病, 于1937年由von Stossel首先报告, 1966年Cornog和Enterline将其命名[18]。近年来临床医师对该疾病的认识逐渐增加, 国内已有不少关于LAM的报告和影像学分析[19-22], 但国内对于VEGF-D的研究较少。VEGF-D也称为淋巴管生成因子, 可结合血管内皮细胞表面受体VEGFR-3/Flt-4, 调节血管和淋巴管的生理功能。Seyama等[13]发现血清VEGF-D水平在LAM患者中显著高于健康对照者, 并提出血清VEGF-D≥800 pg/mL结合影像学中典型的多发囊泡样改变可诊断LAM。Chang等[14]对LAM患者和健康对照者的VEGF-D、血管紧张素转化酶、基质蛋白酶-2和9分别进行检测, 只有VEGF-D在LAM患者中显著升高, 提示血清VEGF-D可作为LAM的辅助诊断指标, 有可能降低LAM的活检率。Xu等[15]发现血清VEGF-D≥800 pg/mL具有较高的敏感性和特异性, 如果将其加入LAM的诊断标准, 90%的疑诊LAM可确诊为LAM, 同时创伤性活检可降低到10%以下。2016年美国胸科协会/日本呼吸协会发表了关于LAM诊断和管理的文章, 指出血清VEGF-D≥800 pg/mL可以作为生物标志物诊断LAM, 避免有创性检查[16]。
从临床角度讲, LAM和OPLD的鉴别诊断价值要高于LAM和健康对照者。PLD是以两肺多发囊性气腔病变为特征的一组疾病, 其影像学均表现为多发囊泡性改变, 其病因主要包括LAM、PLCH、LIP、肺气肿、CTD、结节病和BHD综合征等, 其中LAM、PLCH是PLD最常见的病因。LAM特征性HRCT表现为均匀分布的多个(>10)大小不等的薄壁、圆形、界限清楚的含气囊性病变[10]。PLCH的HRCT表现为结节病变和囊泡性病变, 结节病变为早期主要表现, 囊泡性病变为晚期主要表现, 囊泡直径通常小于10 mm, 也可能高达20 mm, 主要表现为圆形、卵圆形或不规则形状囊腔。LIP的囊腔在两肺均匀分布, 形状各不相同, 通常较大, 但一般小于3 cm [23-24]。SS合并肺部病变时, 其重要的CT征象有磨玻璃样密度实变、结节和肺囊泡改变, 当HRCT出现肺内不规则线影和囊腔改变时, 肺部病理改变以不可逆的纤维化为主, 预示着肺部病变已达终末期[25]。BHD综合征为常染色体显性遗传病, HRCT囊性病变多为双侧分布的囊泡, 有时伴有隔膜, 囊泡变化较大, 可从数毫米到数厘米, 可为圆形、椭圆形和不规则形状, 下叶和纵膈旁多见[26]。
有经验的临床医师结合典型的临床表现、影像学表现以及肺外表现可以对LAM和OPLD进行鉴别诊断。当临床表现和HRCT不典型时, 肺活检是诊断和鉴别PLD的金标准。肺活检是有创性检查手段, 伴有较多并发症, 并非所有患者都能耐受, 此时血清VEGF-D检测就具有重要意义。近期的一些研究发现血清VEGF-D水平在LAM中显著高于OPLD, 并提出将血清VEGF-D≥800 pg/mL作为鉴别LAM和OPLD的指标, 其敏感性和特异性分别为90%~100%和73%~87%[15, 27-31]。
我们的研究发现, 与健康对照者相比, LAM患者的血清VEGF-D水平显著增加, 最佳临界值为692.5 pg/mL, 此时具有较高的敏感性和特异性。我们提出将692.5 pg/mL作为诊断LAM的参考标准。临床工作中, 我们参照指南诊断LAM, 对于没有病理的临床疑诊LAM患者, 血清VEGF-D水平的检测有助于减少活检率, 具有重要意义。LAM患者的血清VEGF-D水平显著高于OPLD, 所以我们提出将血清VEGF-D值901.0 pg/mL作为LAM和OPLD鉴别诊断的依据。
临床工作中, HRCT表现为多发囊泡性改变的疾病较难鉴别。VEGF-D作为一种有效的血清学生物标志物, 可以鉴别LAM和OPLD, 有助于减少有创性检查, 降低活检率, 具有重要临床意义。
| [1] |
OPRESCU N, MCCORMACK FX, BYRNES S, et al. Clinical predictors of mortality and cause of death in lymphangioleiomyomatosis:a population-based registry[J]. Lung, 2013, 191(1): 35-42.
[DOI]
|
| [2] |
FRANZ DN, BISSLER JJ, MCCORMACK, et al. Tuberous sclerosis complex:neurological, renal and pulmonary manifestations[J]. Neuropediatrics, 2010, 41(5): 199-208.
[DOI]
|
| [3] |
HARKNETT EC, CHANG WYC, BYRNES S, et al. Use of variability in national and regional data to estimate the prevalence of lymphangioleiomyomatosis[J]. QJM, 2011, 104(11): 971-979.
[DOI]
|
| [4] |
AUBRY MC, MYERS JL, RYU JH, et al. Pulmonary lymphangioleiomyomatosis in a man[J]. Am J Respir Crit Care Med, 2000, 162(2 Pt 1): 749-752.
[URI]
|
| [5] |
SCHIAVINA M, DI SCIOSCIO V, CONTINI P, et al. Pulmonary lymphangioleiomyomatosis in a karyotypically normal man without tuberous sclerosis complex[J]. Am J Respir Crit Care Med, 2007, 176(1): 96-98.
[DOI]
|
| [6] |
WAKIDA K, WATANABE Y, KUMASAKA T, et al. Lymphangioleiomyomatosis in a male[J]. Ann Thorac Surg, 2015, 100(3): 1105-1107.
[DOI]
|
| [7] |
TAVEIRA-DASILVA AM, MOSS J. Clinical features, epidemiology, and therapy of lymphangioleiomyomatosis[J]. Clin Epidemiol, 2015, 7: 249-257.
[URI]
|
| [8] |
AVILA NA, KELLY JA, CHU SC, et al. Lymphangioleiomyomatosis:abdominopelvic CT and US findings[J]. Radiology, 2000, 216(1): 147-153.
[DOI]
|
| [9] |
AVILA NA, CHEN CC, CHU SC, et al. Pulmonary lymphangioleiomyomatosis:correlation of ventilation-perfusion scintigraphy, chest radiography, and CT with pulmonary function tests[J]. Radiology, 2000, 214(2): 441-446.
[URI]
|
| [10] |
AVILA NA, DWYER AJ, MURPHY-JOHNSON DV, et al. Lymphangioleiomyomatosis: correlation of qualitative and quantitative thin-section CT with pulmonary function tests and assessment of dependence on pleurodesis[J]. Radiology, 2002, 223(1): 189-197.
[DOI]
|
| [11] |
TOIVANEN PL, NIEMINEN T, VⅡTANEN L, et al. Novel vascular endothelial growth factor-D variants with increased biological activity[J]. J Biol Chem, 2009, 284(23): 16037-16048.
[DOI]
|
| [12] |
GUPTA N, MERAJ R, TANASE D, et al. Accuracy of chest highresolution computed tomography in diagnosing diffuse cystic lung diseases[J]. Eur Respir J, 2015, 46(4): 1196-1199.
[DOI]
|
| [13] |
SEYAMA K, KUMASAKA T, SOUMA S, et al. Vascular endothelial growth factor-D is increased in serum of patients with lymphangioleiomyomatosis[J]. Lymphat Res Biol, 2006, 4(3): 143-152.
[DOI]
|
| [14] |
CHANG WY, CANE JL, BLAKEY JD, et al. Clinical utility of diagnostic guidelines and putative biomarkers in lymphangioleiomyomatosis[J]. Respir Res, 2012, 13(1): 34.
[DOI]
|
| [15] |
XU KF, ZHANG P, TIAN X, et al. The role of vascular endothelial growth factor-D in diagnosis of lymphangioleiomyomatosis[J]. Respir Med, 2013, 107(2): 263-268.
[DOI]
|
| [16] |
FRANCIS X.MCCORMACK, NISHANT GUPTA, et al. Official American Thoracic Society/Japanese Respiratory Society clinical practice guidelines:lymphangioleiomyomatosis diagnosis and management[J]. Am J Respir Crit Care Med, 2016, 194(6): 748-761.
[DOI]
|
| [17] |
JOHNSON SR, CORDIER JF, LAZOR R, et al. European Respiratory Society guidelines for the diagnosis and management of lymphangioleiomyomatosis[J]. Eur Respir J, 2010, 35(1): 14-26.
[DOI]
|
| [18] |
CORNOG JL JR, ENTERLINE HT. Lymphangiomyoma, a benign lesion of chyliferous lymphatics synonymous with lymphangiopericytoma[J]. Cancer, 1966, 19(12): 1909-1930.
[DOI]
|
| [19] |
金美玲, 蔡映云, 张志凤. 肺淋巴管平滑肌瘤病[J]. 中华结核和呼吸杂志, 2000, 23(6): 355-257. [DOI]
|
| [20] |
叶伶, 金美玲, 白春学. 120例肺淋巴管平滑肌瘤病临床分析[J]. 临床内科杂志, 2010, 27(12): 932-934. [URI]
|
| [21] |
YE L, JIN M, BAI C. Clinical analysis of patients with pulmonary lymphangioleiomyomatosis (PLAM) in mainland China[J]. Respir Med, 2010, 104(10): 1521-1526.
[DOI]
|
| [22] |
赵金勇, 曹世文. 高分辨CT对肺淋巴管平滑肌瘤病与特发性肺间质纤维化的鉴别诊断意义[J]. 医学影像学杂志, 2016, 26(2): 359-361. [URI]
|
| [23] |
FERREIRA FRANCISCO FA, SOARES SOUZA AJR, ZANETTI G, et al. Multiple cystic lung disease[J]. Eur Respir Rev, 2015, 24(138): 552-564.
[DOI]
|
| [24] |
DAL SASSO AA, BELÉM LC, ZANETTI G, et al. Birt-Hogg-Dubé syndrome.State-of-the-art review with emphasis on pulmonary involvement[J]. Respir Med, 2015, 109(3): 289-296.
[DOI]
|
| [25] |
姜传武, 范传波, 陆学超, 等. 胸部CT诊断干燥综合征1例[J]. 中国中西医结合影像学杂志, 2010, 8(5): 476-477. [DOI]
|
| [26] |
MATSUYAMA N, ASHIZAWA K, OKIMOTO T, et al. Pulmonary lesions associated with Sjögren's syndrome:radiographic and CT findings[J]. Br J Radiol, 2003, 76(912): 880-884.
[DOI]
|
| [27] |
YOUNG LR, INOUE Y, MCCORMACK FX. Diagnostic potential of serum VEGF-D for lymphangioleiomyomatosis[J]. N Engl J Med, 2008, 358(2): 199-200.
[DOI]
|
| [28] |
RADZIKOWSKA E, JAGUS' P, SKOCZYLAS A, et al. Role of serum vascular endothelial growth factor D in discrimination of patients with polycystic lung diseases[J]. Pol Arch Med Wewn, 2013, 123(10): 533-538.
[URI]
|
| [29] |
ELZBIETA RADZIKOWSKA, PAULINA JAGUS, MAŁGORZATA SOBIECKA, et al. Correlation of serum vascular endothelial growth factor-D concentration with clinical presentation and course of lymphangioleiomyomatosis[J]. Respir Med, 2015, 109(11): 1469-1475.
[DOI]
|
| [30] |
YOUNG LR, VANDYKE R, GULLEMAN PM, et al. Serum vascular endothelial growth factor D prospectively distinguishes lymphangioleiomyomatosis from other diseases[J]. Chest, 2010, 138(3): 674-681.
[DOI]
|
| [31] |
MOU Y, YE L, WANG J, et al. Diagnostic and treatment monitoring potential of serum vascular endothelial growth factor-D in lymphangioleiomyomatosis[J]. Lymphology, 2016, 49(3): 140-149.
[URI]
|
 2019, Vol. 46
2019, Vol. 46


