Vitamin D3, also referred as cholecalciferol, or the sunshine vitamin [1], is associated with a whole host of applications throughout the body.Research over the past decade suggested that vitamin D plays a much broader disease-fighting role [2-5].Currently, the evidence is anecdotal that vitamin D is an effective adjuvant for the treatment of other serious medicals [5-6].Pharmacological doses of vitamin D are effective in treating viral respiratory infections [7], and high doses are effective in treating vitamin D deficiency [8], multiple sclerosis [9], systemic lupus erythematosus[10] and so on.
Analytical method of vitamin D3 on the United States Pharmacopeia (USP) and the Chinese Pharmacopeia (ChP) used normal phase (NP) HPLC which is less widely used compared to reverse phase (RP) HPLC.Vitamin D3 is a fat-soluble secosteroid compound containing conjugated triene systems.It is liable to hydrolytic, oxidative, thermal and photolytic conditions.The related substances concerned in vitamin D3 system suitability assay on USP and ChP are pre-vitamin D3, trans-vitamin D3 and tachysterol (Fig 1).Two referred on both USP and ChP are pre-vitamin D3 from the thermal degradation of vitamin D3 and trans-vitamin D3 from ultraviolet (UV) degradation of pre-vitamin D3.The other one on ChP is tachysterol from UV degradation of vitamin D3 [11].The resolution both pharmacopoeias required is no less than 1 which is less than the common requirement, 1.5.This reveals the poor separation under NP-HPLC with V (hexane):V (pentanol)=997:3 as mobile phase.The more widely used RP-HPLC may be taken into consideration to achieve better separation.
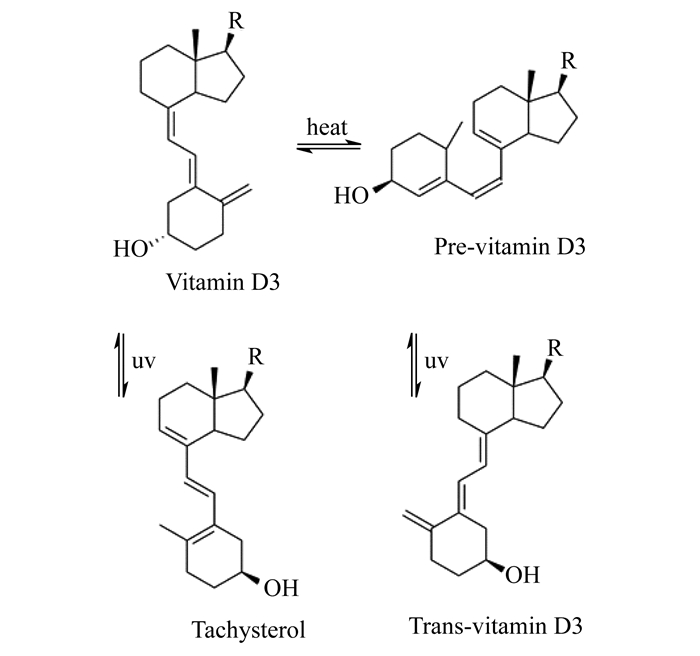
|
| R:-CHH3(CH2)3CH(CH3)2 Fig 1 Photochemical and thermal isomers of vitamin D3 |
Great majority of published HPLC-UV methods related to vitamin D3 focus on quantification in food, feed, pharmaceuticals, nutritional supplement [12], plasma [13], liver [14] and so on.They used NP or RP-HPLC with different mobile phase.In terms of impurities, there are several researches and they were mainly concerned about stability-indicating [15] or just followed the pharmacopoeia using NP-HPLC [16].All these studies did not simultaneously separate the three related substances referred on the USP and ChP.Moreover, these impurities studies also showed poor separation between pre-vitamin D3 and tachysterol [11], or trans-vitamin D3 and tachysterol [15].HPLC coupled to MS is not favorable because these three isomers show the similar fragmentation profile.
Especially, it is noted that oil is the matrix of vitamin D3 liquid preparation, either injection on ChP or vitamin D3 solution on USP.It is rather challenging to develop RP-HPLC methods for vitamin D3 preparations with oil matrix for both highly lipophilic nature which usually needs complex pretreatment like saponification with heating and alkali addition process [17].Assay with directly injection without any pretreatment is quite easy.However, it showed poor method accuracy because of great interference from oil matrix of vitamin D3 soft capsules.
The aim of the present report was to study the retention behaviors of vitamin D3 and three isomers referred on USP and ChP in RP-HPLC system and develop an analysis method for vitamin D3 soft capsules (400 IU) with soybean oil matrix.For this purpose, methods using mobile phase with different composition were performed including acetonitrile, methanol, water and ethanol.Acetonitrile, methanol and water were widely used in previous research.Ethanol was found to show special effect on retention of vitamin D3 in oil matrix in the present work.Retention behaviors were studied on various chromatographic parameters such as resolution, asymmetry factor, retention time, etc.Furthermore, the study of retention behaviors was applied to content determination and impurities profile of challengeable vitamin D3 soft capsules.The present work can help to make more rigorous analysis strategy for vitamin D3 pharmaceuticals and provide reference for analysis of non-polar drugs with oil matrix in RP-HPLC system.
Materials and methodsChemicals and regents Acetonitrile and ethanol (HPLC grade) were purchased from Sigma-Aldrich(Shanghai, China).Methanol (HPLC grade) was purchased from Merck (Darmstadt, Germany).Deionized water was prepared by Millipore Elix® Advantage system (Millipore MA, USA).Vitamin D3 soft capsules (400 IU/softgel, 1 μg=40 IU) were purchased from Sinopharm Xingsha Pharmaceuticals (Xiamen) Co.Ltd (Xiamen, China).Standard of Vitamin D3, purity 99.8%, was purchased from National Institutes for Food and Drug Control of China (Beijing, China).
RP-HPLC conditions The HPLC separation was conducted on a HITACHI L-2000 series HPLC apparatus equipped with a L-2130 quaternary pump, a L-2200 auto sampler, a L-2300 column heater, and a L-2455 DAD.It was performed on an Agilent Zorbax Eclipse XDB-C18 column (4.6 mm×250 mm, 5 μm) at 25 ℃ with a flow rate of 1 mL/min.The detection wavelength was carried out at 265 nm.
Preparation of solutions For the preparation of the stock solution 1 (acetonitrile) and stock solution 2 (methanol), vitamin D3 was weighed accurately, dissolved in solution 1 and solution 2 respectively and diluted to 2.6×10-3 μmol/L subsequently.Working solutions at varied concentrations were prepared by an appropriate dilution of stock solution 1.Sample packaging and storage:preserved in hermetically sealed containers under nitrogen, stored at 4 ℃ and protected from light.
Forced degradation of vitamin D3 In our work, it was found that degradation of vitamin D3 in acetonitrile solution could only produce two isomers.Degradation vitamin D3 in methanol solution could achieved all three isomers.Thus, sample of vitamin D3 in methanol solution (stock solution 2) was chosen to be degraded.Degradation was performed by four samples each contained 2 mL stock solution 2.Sample 1 was heated in water bath for 1 h at 90 ℃ (thermal degradation).Sample 2 was exposed to 254 nm and 365 nm UV radiation for 5 min (photo degradation).Sample 3 (thermal and photo degradation) was heated in water bath for 1 h at 90 ℃and then exposed to 254 nm and 365 nm UV radiation for 5 min.Sample 4 was prepared with severer condition that heated in water bath for 1.5 h at 90 ℃ and exposed to 254 nm and 365 nm UV for 30 min subsequently.
Retention behaviors studies under various mobile phase Forced degradation samples (sample 3 and sample 4) achieved in 2.4 were applied to study the retention behaviors of vitamin D3 and three related substances by altering the mobile phase.100% acetonitrile, V (acetonitrile):V (methanol)=99:1, 98:2, 97:3, 90:10 and 80:20, V (acetonitrile):V (ethanol)=99:1, 98:2, 97:3, 90:10 and 80:20 and linear gradient from 0% (100% acetonitrile), 5%, 10% to 15% water were performed, respectively.Retention time, resolution, asymmetry factor and other HPLC parameters were taken into consideration.
Validation of content determination method Six different concentrations (0.26×10-4, 0.52×10-4, 0.78×10-4, 1.3×10-4, 2.6×10-4, 5.2×10-4μmol/L) of vitamin D3 were prepared by diluting stock solution 1 as described previously.The calibration plot was generated by replicate analysis (n=6) at all concentration levels and the linear relationship was evaluated using the least square method within Microsoft Excel® program.Both repeatability (within a day precision) and reproducibility (between days precision) were determined as follows:Six injections at each of the concentrations were injected within the same day for repeatability, and over a period of 3 days (6 injections/day)for reproducibility.Mean and relative standard deviation were calculated and used to judge accuracy and precision of the method.The LODs and LOQs were separately determined at an S/N of 3 and 10, respectively.The stability of the working solutions was tested at 4 ℃ for 24 h and room temperature for 24 h.
Results and discussion100% acetonitrile as mobile phase was applied to challengeable soft capsules with soybean oil matrix which usually requires complex pretreatment.However, oil matrix was found to have great interference to the reproducibility of retention time and method accuracy.The recovery was only 80.55%-84.37% and retention time changed a lot in sequence with 100% acetonitrile as mobile phase.Remarkably, ethanol addition in acetonitrile was found to make significant improvement.This method using V (acetonitrile):V (ethanol)=90:10 as mobile phase by directly injection without any complex pretreatment is quite easy.Recovery rate was achieved between 98.07% and 103.23%, and the RSD was below 1.69%.The linear in the concentration range of (0.52-5.2)×10-4 μmol/L (R2>0.999).LOD and LOQ were found to be 0.2 and 1 μg/mL at an S/N of 3 and 8, respectively.The RSD of 4 concentrations (0.26×10-4, 0.78×10-4, 1.3×10-4, 5.2×10-4 μmol/L) tested intraday and interday were below 2.32%.The working solutions were shown to be stable in (RSD≤1.21%) over the tested period.
For impurities profile, it could be achieved using V (acetonitrile):V (water)=95:5 as mobile phase which can obviate interference from soybean oil matrix.
Forced degradation studies Fig 2 shows the chromatograms of vitamin D3 forced degradation sample 1-3 with 100% acetonitrile as mobile phase.Peak purity test complied for the peaks indicated no co-elution of degradation products.Peak 4 is the vitamin D3 parent peak.Peak 2 (Fig 2A) is pre-vitamin D3 according to thermal conversion property of vitamin D3 as mentioned before.Peak 1 (Fig 2B) is trans-vitamin D3 according to light conversion property of vitamin D3.Peak 3 (Fig 2C) is tachysterol according to light-thermal conversion property of vitamin D3.The maximum UV absorption wavelengths of pre-vitamin D3, vitamin D3, trans-vitamin D3 and tachysterol (Fig 3) were 260, 262, 276 and 280 nm respectively, which agreed with references [11, 15] Their molecular weight were all 385.4 ([M+H]+) confirmed by HPLC tandem MS.Sample 4 showed the same peaks with chromatogram of sample 3 and was used for better retention behaviors analysis.

|
| A:Thermal degradation; B:Photo degradation; C:Thermal and photo degradation.Peak 1:Trans-vitamin D3;Peak 2:Pre-vitamin D3;Peak 3:Tachysterol; Peak 4:Vitamin D3. Fig 2 Chromatograms of forced degradation samples |
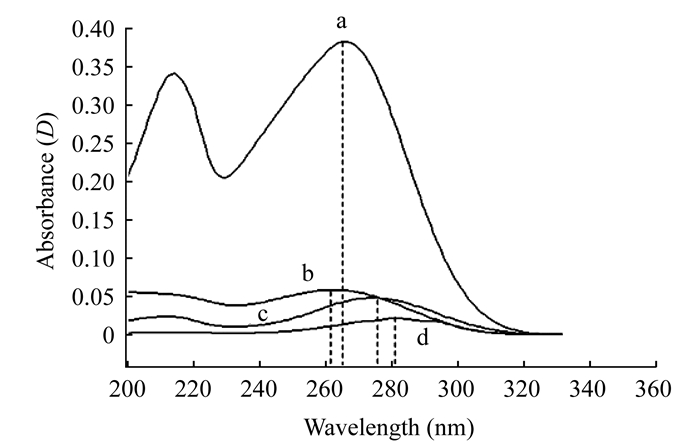
|
| a:Vitamin D3 (λmax=262 nm); b:Pre-vitamin D3 (λmax=260 nm); c:Trans-vitamin D3 (λmax=276 nm); d:Tachysterol (λmax=280 nm).λmax:Maximum absorption wavelength. Fig 3 UV absorption spectrums of vitamin D3 and its three isomers |
Retention behaviors studies under various mobile phase HPLC parameters of altering mobile phase were listed in Tab 1.Chromatograms were shown in Fig 4.As shown in Fig 4, the retention time gradually decreased as the proportion of ethanol or methanol to acetonitrile increased.Meanwhile, little addition of methanol and ethanol (1%) resulted in great resolution reduction between peak 2 (pre-vitamin D3) and peak 3 (tachysterol).When proportion of either methanol or ethanol up to 10%, peak 2 and 3 merged together totally into one peak (peak 2+3).Continuing up to 20%, peak 2, 3 and 4 merged together into one peak (peak 2+3+4).Addition of water in acetonitrile greatly increased retention time.Meanwhile, it also resulted in resolution reduction between peak 2 and peak 3.Peak tailing in Fig 4C-a and peak fronting of peak 2+3 in Fig 4C-b could be observed clearly.Continuing adding water until 15%, 4 peaks were separated (Fig 4C-c).The elution order changed from peak 1, 2, 3, 4 to peak 1, 3, 2, 4.This revealed that the relative position of tachysterol changed gradually from the back to the front of pre-vitamin D3 as the proportion of water increased, further showed the apparent different elution rate change among different peaks especially between peak 2 and 3.Unfortunately, the retention time was so long that it was not available in analysis.Using 100% ACN as mobile phase achieved the best separation and appropriate retention time (Fig 2C).
| Mobile phase composition | Peak | RT (min) | Resolution | α | Asymmetry | k' |
| ACN | 1 | 26.04 | 1.02 | 9.54 | ||
| 2 | 30.20 | 5.30 | 1.16 | 1.02 | 11.23 | |
| 3 | 31.54 | 1.55 | 1.04 | 1.00 | 11.77 | |
| 4 | 35.16 | 3.73 | 1.11 | 1.36 | 13.23 | |
| V (ACN):V (EtOH)=99:1 | 1 | 25.64 | 0.88 | 9.51 | ||
| 2 | 29.12 | 3.27 | 1.14 | 10.93 | ||
| 3 | 29.97 | 0.78 | 1.03 | 11.28 | ||
| 4 | 32.52 | 2.14 | 1.08 | 0.88 | 12.33 | |
| V (ACN):V (EtOH)=98:2 | 1 | 25.15 | 0.96 | 9.27 | ||
| 2 | 28.29 | 3.62 | 1.12 | 10.55 | ||
| 3 | 28.83 | 0.62 | 1.02 | 10.77 | ||
| 4 | 30.85 | 2.14 | 1.07 | 0.96 | 11.59 | |
| V (ACN):V (EtOH)=97:3 | 1 | 24.72 | 0.99 | 9.05 | ||
| 2+3 | 27.95 | 2.92 | 1.13 | 0.80 | 10.36 | |
| 4 | 29.71 | 1.49 | 1.06 | 0.96 | 11.08 | |
| V (ACN):V (EtOH)=90:10 | 1 | 19.94 | 1.20 | 6.98 | ||
| 2+3 | 21.62 | 2.67 | 1.08 | 7.65 | ||
| 4 | 22.34 | 1.10 | 1.03 | 1.10 | 7.94 | |
| V (ACN):V (EtOH)=80:20 | 1 | 15.35 | 1.10 | 5.09 | ||
| 2+3+4 | 16.63 | 2.43 | 1.08 | 0.84 | 5.60 | |
| V (ACN):V (MeOH)=99:1 | 1 | 25.46 | 0.94 | 9.43 | ||
| 2 | 28.92 | 3.36 | 1.14 | 10.85 | ||
| 3 | 29.78 | 0.81 | 1.03 | 11.20 | ||
| 4 | 32.24 | 2.24 | 1.08 | 0.93 | 12.21 | |
| V (ACN):V (MeOH)=98:2 | 1 | 24.94 | 0.90 | 9.18 | ||
| 2+3 | 28.48 | 2.67 | 1.14 | 0.75 | 10.62 | |
| 4 | 30.41 | 1.34 | 1.07 | 0.90 | 11.41 | |
| V (ACN):V (MeOH)=97:3 | 1 | 24.61 | 0.93 | 9.00 | ||
| 2+3 | 27.69 | 2.64 | 1.13 | 0.87 | 10.26 | |
| 4 | 29.47 | 1.43 | 1.06 | 0.91 | 10.98 | |
| V (ACN):V (MeOH)=90:10 | 1 | 22.56 | 1.21 | 8.32 | ||
| 2+3 | 24.62 | 2.72 | 1.09 | 9.17 | ||
| 4 | 25.59 | 1.21 | 1.04 | 1.02 | 9.57 | |
| V (ACN):V (MeOH)=80:20 | 1 | 21.16 | 1.10 | 7.78 | ||
| 2+3+4 | 23.22 | 3.08 | 1.10 | 0.77 | 8.63 | |
| V (ACN):V (H2O)=95:5 | 1 | 43.52 | 1.07 | 18.34 | ||
| 2+3 | 48.29 | 3.91 | 1.11 | 1.10 | 20.46 | |
| 4 | 53.08 | 3.25 | 1.10 | 1.11 | 22.59 | |
| V (ACN):V (H2O)=90:10 | 1 | 58.71 | 1.10 | 17.40 | ||
| 2+3 | 63.23 | 3.45 | 1.08 | 0.92 | 18.82 | |
| 4 | 67.54 | 3.05 | 1.07 | 1.08 | 20.17 | |
| V (ACN):V (H2O)=85:15 | 1 | 87.56 | 25.53 | |||
| 3 | 91.20 | 2.70 | 1.04 | 26.64 | ||
| 2 | 93.09 | 1.27 | 1.02 | 1.11 | 27.21 | |
| 4 | 98.09 | 2.81 | 1.05 | 1.08 | 28.72 | |
| RT:Retention time; α:Separation factor; k':Capacity factor. | ||||||
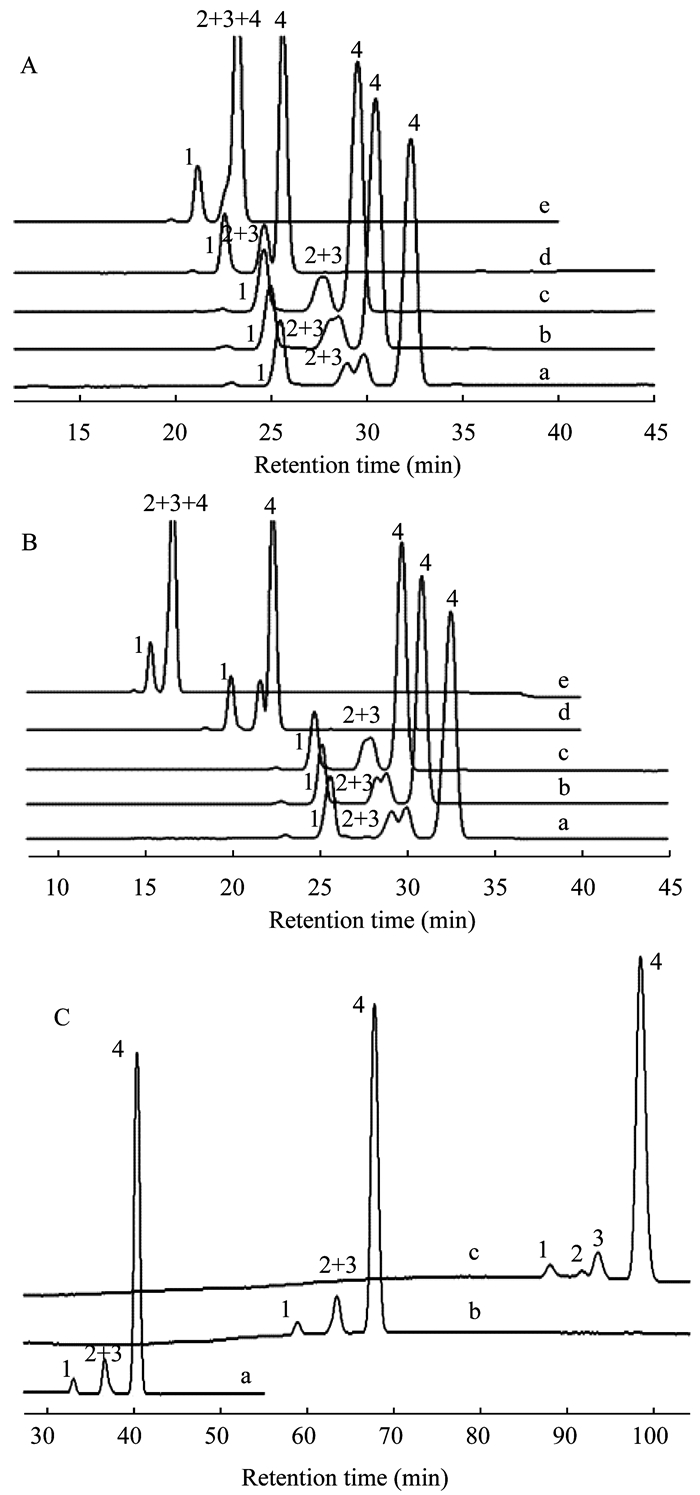
|
| A and B:a-e, V (ACH):V (MeOH)=99:1, 98:2, 97:3, 90:10 and 80:20;C:a-c, V (ACH):V (H2O)=95:5, 90:10 and 85:15.MeOH:Methanol; ACN:Acetonitrile; H2O:Water. Fig 4 Chromatograms of sample 4 with various proportions of methanol (A), ethanol (B) and water (C) in acetonitrile as mobile phase |
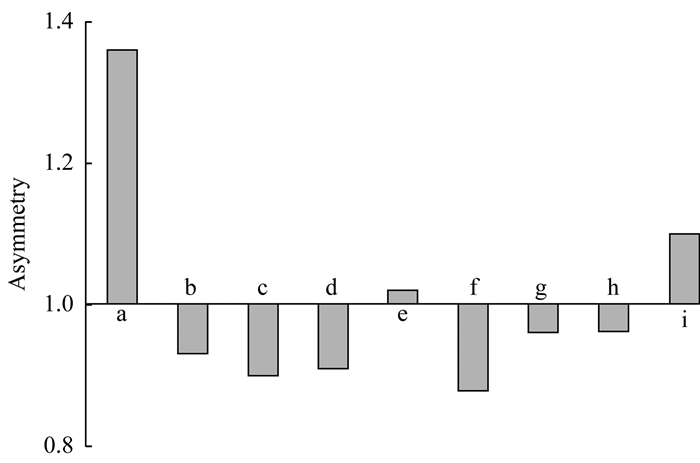
These results indicated that ethanol and methanol had stronger elution ability than acetonitrile.Ethanol was stronger than methanol for achieving shorter retention time at the same proportion (10% and 20%).Water was the weakest.Solubility was supposed to be the major concern in elution ability evaluation as vitamin D3 is very soluble in ethanol and freely soluble in methanol.Acetonitrile take the third place.Water in which vitamin D3 is insoluble had the weakest elution ability.Ethanol and methanol addition could not only increase elution ability but also improve peak shape as shown in Fig 5, especially for high concentration sample.
|
| a:100%ACN; b-e:V (ACN):V (MeOH)=99:1, 98:2, 97:3, 90:10, respectively; f-i:V (ACN):V (EtOH)=99:1, 98:2, 97:3, 90:10, respectively. Fig 5 Asymmetry of vitamin D3 peak using different mobile phases |
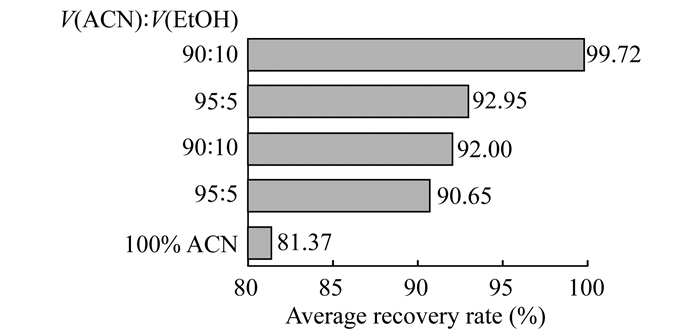
Application of retention behaviors in vitamin D3 soft capsules (400 IU) with soybean oil matrix Assay the content of vitamin D3 soft capsules with soybean oil matrix After studying the retention behaviors, we applied the results to assay content of vitamin D3 soft capsules by directly injecting without any pretreatment.However, the retention time changed a lot in sequence.The recovery of spiked sample was only 80.55%-84.37% using acetonitrile as mobile phase.The reason speculated was that part of vitamin D3 retained together with oil on the column for its relative low solubility in acetonitrile, which would be eluted later with oil during the elution process.This part of vitamin D3 that could not be eluted with others collectively resulted in poor method accuracy.Ethanol and methanol were chosen to add in acetonitrile as mobile phase for their good solubility for vitamin D3.V (acetonitrile):V (methanol)=95:5, 90:10 and V (acetonitrile):V (ethanol)=95:5, 90:10 were performed respectively.Vitamin D3 retention time was reproducible and recovery was found significantly improved (Fig 6).Ethanol does better than methanol may due to its excellent solubility to vitamin D3.The recovery rate between 98.07% and 103.23% can be achieved when V (acetonitrile):V (ethanol)=90:10 was used as mobile phase.
|
| Fig 6 Effect of ethanol or methanol addition in acetonitrile as mobile phase on recovery of vitamin D3 soft capsules with oil matrix (n=6) |
Impurities profile of vitamin D3 soft capsules with soybean oil matrix Considering the low dosage of this soft capsules (400 IU, 0.96×10-4 μmol/L), we extracted vitamin D3 by ethanol [18-19], which is very soluble for vitamin D3 but very slightly soluble for soybean oil.Then it was concentrated by using rotary evaporator at 25 ℃ and resolved in acetonitrile.The final enriched sample was used for analysis.
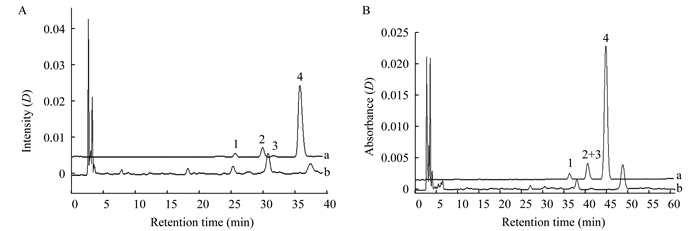
Baseline separation among vitamin D3 and its three isomers could be achieved with acetonitrile as mobile phase.However, when it was applied to impurities profile of vitamin D3 soft capsules enriched sample, there were 2 components peaks in blank matrix overlapped with peaks of impurities (Fig 7A).According to the previous studies of retention behaviors, water addition in acetonitrile as mobile phase was tried.V (acetonitrile):V (water)=95:5 was found to obviate the interference (Fig 7B).The peak 2+3 of pre-vitamin D3 and tachysterol can be considered together in impurities limit assay.Due to the unsatisfactory poor separation between pre-vitamin D3 and tachysterol of this method, many ways had been tried to resolve interference from blank matrix, such as using a detection wavelength that matrix interference is negligible at this wavelength, or useing other pretreatments like lipase hydrolysis and solid phase extraction (SPE).All these methods were not available.Using V (acetonitrile):V (water)=95:5 as mobile phase is the best way known up to now.
|
| A:100% acetonitrile; B:V (acetonitrile):V (H2O)=95:5.a:Degradation sample (sample 3);b:Blank matrix of vitamin D3 soft capsule. Fig 7 Chromatograms of degradation sample and blank matrix with 100% acetonitrile and V (acetonitrile):V (H2O)=95:5 as mobile phase |
Concluding remarks In this research, it was clearly studied that how different mobile phase compositions including acetonitrile, methanol, water and ethanol influenced the retention behaviors of vitamin D3 and its three isomers in RP-HPLC system.The proposed quantification method for vitamin D3 soft capsules with soybean oil matrix is quite easy by directly injection without any complex pretreatment.
Based on the above study, it was suggested that for vitamin D3 pharmaceuticals content determination, it is better to choose acetonitrile with appropriate addition of methanol or ethanol as mobile phase for shorter retention time.The parent peak (vitamin D3) should be separated from impurities as methanol and ethanol could reduce resolution.Ethanol addition is especially, recommended for better method accuracy when analyzing liquid preparation with oil matrix.For impurities profile, one hundred percent acetonitrile as mobile phase is recommended for good separation.However, if excipients interfere analysis, little water addition can be taken into consideration.Importantly, our work can also provide reference for the analysis of other non-polar drug with oily matrix in RP-HPLC system.
Conflict of interest The authors have declared no conflicts of interest.
| [1] |
BARTOLUCCI G, GIOCALIERE E, BOSCARO F, et al. Vitamin D3 quantification in a cod liver oil-based supplement[J]. J Pharm Biomed Anal, 2011, 55(1): 64-70.
[DOI]
|
| [2] |
GARLAND CF, GARLAND FC, GORHAM ED, et al. The role of vitamin D in cancer prevention[J]. Am J Public Health, 2006, 96(2): 252-261.
[DOI]
|
| [3] |
SCARANTI M, JÚNIOR GDE C, HOFF AO. Vitamin D and cancer:does it really matter?[J]. CurrOpinOncol, 2016, 28(3): 205-209.
[URI]
|
| [4] |
SWAMI S, KRISHNAN AV, WILLIAMS J, et al. Vitamin D mitigates the adverse effects of obesity on breast cancer in mice[J]. Endocr Relat Cancer, 2016, 23(4): 251-264.
[DOI]
|
| [5] |
DOU R, NG K, GIOVANNUCCI EL, et al. Vitamin D and colorectal cancer:molecular, epidemiological and clinical evidence[J]. Br J Nutr, 2016, 115(9): 1-18.
[URI]
|
| [6] |
CANNELL JJ, HOLLIS BW. Use of vitamin D in clinical practice[J]. Altern Med Rev, 2008, 13(1): 6-20.
[URI]
|
| [7] |
CANNELL JJ, VIETH R, UMHAU JC, et al. Epidemic influenza and vitamin D[J]. Epidemiol Infect, 2006, 134(6): 1129-1140.
[DOI]
|
| [8] |
DIAMOND TH, HO KW, ROHL PG, et al. Annual intramuscular injection of a megadose of cholecalciferol for treatment of vitamin D deficiency:efficacy and safety data[J]. Med JAust, 2005, 183(1): 10-12.
[URI]
|
| [9] |
KIMBALL SM, URSELL MR, O′CONNOR P, et al. Safety of vitamin D3 in adults with multiple sclerosis[J]. Am J ClinNutr, 2007, 86(3): 645-651.
[URI]
|
| [10] |
ABOU-RAYA A, ABOU-RAYA S, HELMII M. The effect of vitamin D supplementation on inflammatory and hemostatic markers and disease activity in patients with systemic lupus erythematosus:a randomized placebo-controlled trial[J]. J Rheumatol, 2013, 40(3): 265-272.
[DOI]
|
| [11] |
BENMOUSSA A, DELAURENT C, LACOUT JL, et al. Determination of cholecalciferol and related substances by calcium phosphate hydroxyapatite and calcium phosphate fluoroapatite high-performance liquid chromatography[J]. J Chromatogr A, 1996, 731(1): 153-160.
[URI]
|
| [12] |
SCHADT HS, GÖSSL R, SEIBEL N, et al. Quantification of vitamin D3 in feed, food, and pharmaceuticals using high-performance liquid chromatography/tandem mass spectrometry[J]. J AOAC Int, 2012, 95(5): 1487-1494.
[DOI]
|
| [13] |
ALBARHANI AA, COLLIER F, GREAVES RF, et al. Vitamins D and A can be successfully measured by LC-MS/MS in cord blood diluted plasma[J]. Clin Biochem, 2015, 48(16-17): 1105-1112.
[DOI]
|
| [14] |
BURILD A, FRANDSEN HL, POULSEN M, et al. Quantification of physiological levels of vitamin D-3 and 25-hydroxyvitamin D-3 in porcine fat and liver in subgram sample sizes[J]. J Sep Sci, 2014, 37(19): 2659-2663.
[DOI]
|
| [15] |
TEMOVA Ž, ROŠKAR R. Stability-indicating HPLC-UV method for vitamin D3 determination in solutions, nutritional supplements and pharmaceuticals[J]. J Chromatogr Sci, 2016, 54(7): 1180-1186.
[DOI]
|
| [16] |
PETRITZ E, TRITTHART T, WINTERSTEIGER R. Determination of phylloquinone and cholecalciferol encapsulated in granulates formed by melt extrusion[J]. J Biochem Biophys Methods, 2006, 69(1): 101-112.
[URI]
|
| [17] |
PERALES S. Review:determination of vitamin D in dairy products by high performance liquid chromatography[J]. Food Sci TechnolInt, 2005, 11(6): 451-462.
[DOI]
|
| [18] |
GOMIS DB, FERNÁNDEZ MP, GUTIÉRREZ ALVAREZ MD. Simultaneous determination of fat-soluble vitamins and provitamins in milk by microcolumn liquid chromatography[J]. J Chromatogr A, 2000, 891(1): 109-114.
[DOI]
|
| [19] |
GOMIS DB, FERNÁNDEZ MP, ALVAREZ M DG. Simultaneous determination of fat-soluble vitamins and provitamins in dairy products by liquid chromatography with a narrow-bore column[J]. Analyst, 2000, 125(3): 427-431.
[DOI]
|
 2019, Vol. 46
2019, Vol. 46


